Synthesis, dispersion and lubrication potential of basal plane functionalized alkylated graphene nanosheets
Abstract
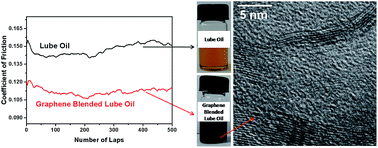
A single step facile approach for grafting of long alkyl chains in the basal plane of graphene oxide and simultaneous reduction of oxygen functionalities to restore the graphitic characteristics, is reported. Chemical and structural features of the synthesized dual-layer alkylated graphene are elucidated by infrared, 13C solid state nuclear magnetic resonance, X-ray diffraction, and high-resolution transmission electron microscopy analyses. The van der Waals interaction between the octadecyl chains grafted on graphene and the alkyl chains of lube oils provided long-term dispersion stability to the alkylated graphene. The 0.02 mg mL−1 alkylated graphene as an optimized concentration in the lube oil, decreased both friction and wear significantly under the sliding contacts between steel tribo-pairs. Micro-Raman results demonstrate the deposition of graphene nanosheets on the tribo-interfaces under the sheared contact, and reduced the friction and protects the surfaces against undesirable wear.

 Please wait while we load your content...
Please wait while we load your content...