Na3B4O7X (X = Cl, Br): two new borate halides with a 1D Na-X (X = Cl, Br) chain formed by the face-sharing XNa6 octahedra†
Chunyan Baiab,
Shujuan Han*a,
Shilie Pan*a,
Bingbing Zhangab,
Yun Yanga,
Lin Liab and
Zhihua Yanga
aKey Laboratory of Functional Materials and Devices for Special Environments of CAS, Xinjiang Key Laboratory of Electronic Information Materials and Devices, Xinjiang Technical Institute of Physics & Chemistry of CAS, 40-1 South Beijing Road, Urumqi 830011, China. E-mail: hansj@ms.xjb.ac.cn; slpan@ms.xjb.ac.cn; Fax: +86-991-3838957; Tel: +86-991-3674558
bUniversity of Chinese Academy of Sciences, Beijing 100049, China
First published on 14th January 2015
Abstract
Two new sodium borate halides, namely, Na3B4O7X (X = Cl, Br), have been successfully synthesized through high-temperature solid state reactions. Single-crystal X-ray diffraction analysis reveals that both of them crystallize in the same space group P6522 of the hexagonal system. In their crystal structures, the B4O9 groups link with each other by oxygen-sharing to form a three dimensional (3D) framework containing channels viewing along the [001] direction, in which the 1D helical [Na3X] chain formed by the face-sharing XNa6 octahedra resides. To the best of our knowledge, the 1D helical [Na3X] chain has not been found in other alkali metal borate halides. From another perspective, the two compounds can also be regarded as NaX salt inclusion borates. In addition, infrared (IR) spectroscopy and bond valence sum (BVS) calculations are used to verify the validity of the structures. Differential scanning calorimetry (DSC) and powder X-ray diffraction (XRD) results prove that both of them are incongruently melting compounds. The electronic structures have been calculated by the first-principle method based on density-functional theory.
Introduction
Borates,1–8 owing to the possible 3- or 4-fold coordination of boron atoms, form a great number of compounds having diverse structures. The diverse structures give them interesting and excellent properties for technical applications, which has attracted the attention of material scientists. After much research effort, many excellent borate crystals have been obtained, such as α-BaB2O4 (α-BBO),9 β-BaB2O4 (β-BBO),10 KBe2BO3F2 (KBBF).11Studies of alkali metal borates have also produced a large family of compounds with outstanding physical properties, such as LiB3O5 (LBO),12 CsB3O5 (CBO),13 CsLiB6O10 (CLBO),14 Li4Cs3B7O24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 and Li6Rb5B11O22,16 the crystals of which have wide range transparency. In addition, it is well-known that combination of halogen with borate can widen band gaps.17 In our group, by introducing halogen atoms into borate, a series of new crystals consisting of similar XM6 (X = Cl, Br; M = alkali metals) octahedra have been successfully synthesized, such as K3B6O10X,18 Na3B6O10X,19 K3−xNaxB6O10Br (x = 0.13, 0.67, 1.30, 2.20)20 and RbNa2B6O10X.19a Interestingly, although these crystals have similar stoichiometry and contain the B6O13 groups and XM6 (X = Cl, Br; M = alkali metals) octahedra, their properties have great difference owing to the different XM6 (X = Cl, Br; M = alkali metals) octahedra. Inspired by the above work, we believe that it is significant to continue an investigation of alkali-metal borate halides containing XM6 octahedra. After extensive efforts, we successfully synthesized two new alkali metal borate halides Na3B4O7X (X = Cl, Br) containing 1D helical [Na3X] chains of face-sharing XNa6 octahedra, which have not been found in other alkali metal borate halides. From another perspective, the two compounds can also be regarded as NaX salt inclusion borates. This paper describes the syntheses, crystal and electronic structures and optical properties of Na3B4O7X (X = Cl, Br). These results give a hint of the very extensive crystal chemistry of alkali-metal borate halides containing XM6 (X = Cl, Br; M = alkali metals) octahedra system, which continues to provide a rich source of new compounds.
15 and Li6Rb5B11O22,16 the crystals of which have wide range transparency. In addition, it is well-known that combination of halogen with borate can widen band gaps.17 In our group, by introducing halogen atoms into borate, a series of new crystals consisting of similar XM6 (X = Cl, Br; M = alkali metals) octahedra have been successfully synthesized, such as K3B6O10X,18 Na3B6O10X,19 K3−xNaxB6O10Br (x = 0.13, 0.67, 1.30, 2.20)20 and RbNa2B6O10X.19a Interestingly, although these crystals have similar stoichiometry and contain the B6O13 groups and XM6 (X = Cl, Br; M = alkali metals) octahedra, their properties have great difference owing to the different XM6 (X = Cl, Br; M = alkali metals) octahedra. Inspired by the above work, we believe that it is significant to continue an investigation of alkali-metal borate halides containing XM6 octahedra. After extensive efforts, we successfully synthesized two new alkali metal borate halides Na3B4O7X (X = Cl, Br) containing 1D helical [Na3X] chains of face-sharing XNa6 octahedra, which have not been found in other alkali metal borate halides. From another perspective, the two compounds can also be regarded as NaX salt inclusion borates. This paper describes the syntheses, crystal and electronic structures and optical properties of Na3B4O7X (X = Cl, Br). These results give a hint of the very extensive crystal chemistry of alkali-metal borate halides containing XM6 (X = Cl, Br; M = alkali metals) octahedra system, which continues to provide a rich source of new compounds.
Experimental
Reagents
NaCl (Tianjin BaiShi Chemical Reagent Co., 99.0%), NaBr (Tianjin HongYan Chemical Reagent Co., Ltd., 99.0%), Na2CO3 (Tianjin HongYan Chemical Co., Ltd., 99.5%) and H3BO3 (Tianjin HongYan Chemical Co., Ltd., 99.5%) were used as received.Crystal growth
Single crystals of Na3B4O7Cl (X = Cl, Br) were grown by spontaneous crystallization. A mixture of 0.424 g (4.00 mmol) of Na2CO3, 0.234 g (4.00 mmol) of NaCl (or 0.411 g (4.00 mmol) of NaBr), and 0.7420 g (12.00 mmol) of H3BO3 was thoroughly ground for Na3B4O7Cl (X = Cl, Br). The mixture was then placed in a platinum crucible that was placed into a vertical, programmable-temperature furnace. The crucible was gradually heated to 750 °C in air, held for 12 h until the solution became transparent and clear. The homogenized solution was cooled rapidly to 700 °C and then slowly cooled to 600 °C at a rate of 2 °C h−1, followed by rapid cooling to room temperature. The crystals were separated mechanically from the crucible for the further characterization by single-crystal X-ray measurements.Structure determination
The single crystals of Na3B4O7X (X = Cl, Br) with dimensions 0.245 mm × 0.126 mm × 0.109 mm and 0.200 mm × 0.172 mm × 0.091 mm were selected for the structure determination, respectively. Their crystal structures were determined by single-crystal XRD on an APEX II CCD diffractometer using monochromatic Mo Kα radiation (λ = 0.71073 Å) at 293(2) K and integrated with the SAINT program.21 Numerical absorption corrections were carried out using the SCALE program for area detector. All calculations were performed with programs from the SHELXTL crystallographic software package.22 All atoms were refined using full matrix least-squares techniques; final least-squares refinement is on Fo2 with data having Fo2 ≥ 2σ(Fo2) (Fo: observed structure factors, σ: standard uncertainty). The structures were checked for missing symmetry elements by the program PLATON.23 Crystal data and structure refinement information are presented in Table 1. The final refined atomic positions, isotropic thermal parameters and bond valence sums (BVS)24 of each atom are summarized in Table 2. Selected bond lengths (in Å) and angles (in degree) are listed in Tables S1 and S2 in the ESI.†| Empirical formula | Na3B4O7Cl | Na3B4O7Br |
|---|---|---|
| a R1 = Σ‖Fo| − |Fc‖/Σ|Fo| and wR2 = [Σw(Fo2 − Fc2)2/ΣwFo4]1/2 for Fo2 > 2σ(Fo2). | ||
| Formula weight | 259.66 | 304.12 |
| Crystal system | Hexagonal | Hexagonal |
| Space group, Z | P6522, 6 | P6522, 6 |
| Unit cell dimensions | a = 8.031(1) Å | a = 8.146(5) Å |
| c = 20.369(3) Å | c = 20.24(3) Å | |
| Volume | 1137.7(2) Å3 | 1163.1(18) Å3 |
| Density (calculated) | 2.274 Mg m−3 | 2.605 Mg m−3 |
| Theta range for data collection | 2.93° to 24.96° | 2.89° to 27.50° |
| Limiting indices | −9 ≤ h ≤ 8, −7 ≤ k ≤ 9, −24 ≤ l ≤ 20 | −9 ≤ h ≤ 10, −7 ≤ k ≤ 10, −23 ≤ l ≤ 26 |
| Reflections collected/unique | 5755/671 [R(int) = 0.0408] | 7065/896 [R(int) = 0.0502] |
| Completeness to theta | 100% | 99.8% |
| Goodness-of-fit on F2 | 1.184 | 1.100 |
| Final R indices [Fo2 > 2σ(Fo2)]a | R1 = 0.0609, wR2 = 0.1809 | R1 = 0.0482, wR2 = 0.1304 |
| R indices (all data)a | R1 = 0.0636, wR2 = 0.1822 | R1 = 0.0548, wR2 = 0.1343 |
| Absolute structure parameter | 0.0(4) | 0.06(3) |
| Extinction coefficient | 0.031(10) | 0.005(3) |
| Largest diff. peak and hole | 0.819 and −0.356 e·Å−3 | 0.938 and −0.883 e·Å−3 |
| Atom | wyck | x | y | z | Ua (eq.) | BVSb,c |
|---|---|---|---|---|---|---|
| a U (eq.) is defined as the one-third of the trace of the orthogonalized Uij tensor.b Bond valences calculated with the program Bond Valence Calculator Version 2.00, C. Hormillosa, S. Healy, T. Stephen, McMaster University, 1993.c Valence sums calculated with the formula: Si = exp[(R0 − Ri)/B], where Si = valence of bond “i” and B = 0.37. | ||||||
| Na3B4O7Cl | ||||||
| Na(1) | 12c | 0.5438(5) | 0.7980(5) | 0.5576(2) | 0.049(1) | 0.838 |
| Na(2) | 6a | 0 | 0.8478(6) | 0.6667(0) | 0.034(1) | 0.979 |
| B(1) | 12c | 0.5134(11) | 0.3563(10) | 0.5298(3) | 0.017(2) | 3.072 |
| B(2) | 12c | 0.8631(10) | 0.5462(11) | 0.5479(4) | 0.018(2) | 3.088 |
| O(1) | 6b | 0.5131(4) | 0.4869(4) | 0.5833(0) | 0.017(1) | 1.906 |
| O(2) | 12c | 0.7133(7) | 0.4383(7) | 0.5050(2) | 0.023(1) | 2.048 |
| O(3) | 12c | 0.4374(7) | 0.1675(6) | 0.5546(2) | 0.022(1) | 2.105 |
| O(4) | 12c | 0.3994(8) | 0.3529(7) | 0.4734(2) | 0.024(1) | 2.057 |
| Cl(1) | 6b | 0.9023(2) | 0.0977(2) | 0.5833(0) | 0.049(1) | 0.693 |
| Na3B4O7Br | ||||||
| Na(1) | 12c | 0.4667(4) | 0.2071(3) | 0.0555(1) | 0.034(1) | 0.926 |
| Na(2) | 6a | 0.8454(5) | 0 | 0 | 0.035(1) | 0.961 |
| B(1) | 12c | 0.1428(7) | 0.4548(8) | 0.0477(3) | 0.010(1) | 3.058 |
| B(2) | 12c | 0.4878(8) | 0.6284(7) | 0.0296(2) | 0.008(1) | 3.043 |
| O(1) | 6b | 0.4896(3) | 0.5104(3) | 0.0833(0) | 0.011(1) | 1.933 |
| O(2) | 12c | 0.2895(5) | 0.5581(5) | 0.0054(2) | 0.013(1) | 1.904 |
| O(3) | 12c | 0.5635(6) | 0.8252(5) | 0.0536(2) | 0.014(1) | 2.032 |
| O(4) | 12c | 0.5991(5) | 0.6398(6) | 0.9719(8) | 0.016(1) | 1.995 |
| Br(1) | 6b | 0.1055(1) | 0.8945(1) | 0.0833(0) | 0.042(1) | 0.890 |
Solid-state synthesis
The polycrystalline powders of Na3B4O7X (X = Cl, Br) were synthesized by high temperature solid-state reactions. The stoichiometric mixtures of Na2CO3, NaX (X = Cl, Br) and H3BO3 at a molar ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 for Na3B4O7X (X = Cl, Br) were ground well and packed into Pt crucibles, respectively. The raw materials were heated to 400 °C to decompose the carbonate and eliminate the water, then the compounds were gradually heated up to 650 °C for Na3B4O7X (X = Cl, Br) and kept at this temperature for 72 h. During the sintering steps, the samples were cooled to room temperature and ground. The powders of Na3B4O7X (X = Cl, Br) were obtained. The powder X-ray diffraction (XRD) data were collected at room temperature in the angular range of 2θ = 10–70° with a scan step width of 0.02° and a fixed counting time of 1 s per step by using an automated Bruker D2 X-ray diffractometer with Cu-Kα radiation (λ = 1.5418 Å). The diffraction patterns are well agreeable with the calculated ones (Fig. 1).
4 for Na3B4O7X (X = Cl, Br) were ground well and packed into Pt crucibles, respectively. The raw materials were heated to 400 °C to decompose the carbonate and eliminate the water, then the compounds were gradually heated up to 650 °C for Na3B4O7X (X = Cl, Br) and kept at this temperature for 72 h. During the sintering steps, the samples were cooled to room temperature and ground. The powders of Na3B4O7X (X = Cl, Br) were obtained. The powder X-ray diffraction (XRD) data were collected at room temperature in the angular range of 2θ = 10–70° with a scan step width of 0.02° and a fixed counting time of 1 s per step by using an automated Bruker D2 X-ray diffractometer with Cu-Kα radiation (λ = 1.5418 Å). The diffraction patterns are well agreeable with the calculated ones (Fig. 1).
 | ||
| Fig. 1 Powder XRD patterns of calculated, before melting and after melting for (a) Na3B4O7Cl, (b) Na3B4O7Br. | ||
IR spectroscopy
IR spectra of Na3B4O7X (X = Cl, Br) were recorded on a Shimadzu IR Affinity-1 Fourier transform infrared spectrometer in the 400–4000 cm−1 range. The samples were mixed thoroughly with dried KBr, and the characteristic absorption peaks were shown in Fig. S1 in the ESI.†UV-Vis-NIR diffuse reflectance spectra
Optical diffuse reflectance spectra of Na3B4O7X (X = Cl, Br) were measured at room temperature with a Shimadzu SolidSpec-3700DUV spectrophotometer. Data were collected in the wavelength range 190–2600 nm. Also, reflectance spectra were converted to absorbance with the Kubelka–Munk function.25,26Thermal analysis
The melting behaviors of Na3B4O7X (X = Cl, Br) were carried out on NETZSCH STA 449C simultaneous analyzer instrument in an atmosphere of flowing N2. The sample heated from 25 °C to 1000 °C at a rate of 10 °C min−1.Elemental analysis
Elemental analysis of single crystal was measured by a VISTA-PRO CCD simultaneous ICP-OES. The crystal samples were dissolved in nitric acid. The elemental analysis results are listed in Table S3.†Theoretical calculations
The electronic structures of the title crystals, including the band structure and full/partial density of states (DOS/PDOS), were performed by a plane-wave pseudopotential package employed in CASTEP.27 Norm-conserving pseudopotentials (NCP)28 were used. The exchange–correlation functional was Perdew–Burke–Emzerhoff (PBE) functional within the generalized gradient approximation (GGA).29 The plane-wave energy cutoff was set at 750 eV. The numerical integration of the Brillouin zone was performed using a 4 × 4 × 2 Monkhorst–Pack k-point sampling.Results and discussion
Crystal structures
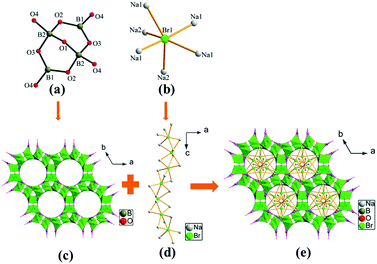
Na3B4O7X (X = Cl, Br) have similar 3D framework, except that the ClNa6 group is replaced by BrNa6 group. Hence only the structure of Na3B4O7Br will be discussed in detail as a representation. X-ray analysis reveals that Na3B4O7Br crystallizes in the chiral space group P6522 of the hexagonal system. In the asymmetric unit of Na3B4O7Br, there are two unique Na atoms, two unique B atoms, four unique O atoms and one unique Br atom (Table 2).The structure of Na3B4O7Br is illustrated in Fig. 2. The structure consists of the B4O9 groups and BrNa6 polyhedra as structure building units (Fig. 2(a) and (b)). Interestingly, a notable feature in the structure is the 1D helical [Na3Br] chain formed by the face-sharing BrNa6 polyhedra, which has not been found in other alkali metal borate halides. In general, XM6 octahedron (A = alkali metals; X = halogen) exists in the structure as the 3D framework, such as K3B6O10X,18 Na3B6O10X,19 K3−xNaxB6O10Br (x = 0.13, 0.67, 1.30, 2.20),20 RbNa2B6O10X.19a The B4O9 group is built of two BO4 tetrahedra (T) and two BO3 triangles (Δ) through corner-sharing, which is the typical tetraborate block with notation 4:[2Δ + 2T] introduced by Christ and Clark.30 Each B4O9 group is connected by sharing its four terminal oxygens to form a 3D network with channels pointing along the c axis (Fig. 2(c) and S3 in the ESI†), where the [Na3Br] chains are filled. The tetraborate B4O9 group is also found in many anhydrous as well as hydrous borates. It can be found that the B4O9 groups can exist as isolated groups or further linked with each other to form 3D frameworks. For example, it can exist as, an isolated [B4O5(OH)4]2− group in Borax Na2B4O5(OH)4·8H2O,31 as a [B4O7(OH)2]2− group with a [MnB4O7(OH2)]2− chain in Roweite Ca2Mn2(OH)4B4O7(OH)2,32 as an isolated B4O9 group in Na3GaB4O9,33 Cs2GeB4O9 (ref. 34) and as a 3D [B4O9]2− network in Li2B4O7 (ref. 35) and LiNaB4O7.36
The Na atoms have one kind of coordinated environment: both Na(1) and Na(2) atoms are coordinated by four O atoms and two Br atoms (Fig. S2 in the ESI†). The Na–O bond distances have a wide region varying from 2.282(5) Å to 2.928(5) Å, and Na–Br distances vary from 2.822(3) Å to 3.346(4) Å. The B atoms have two kinds of coordinated environments: the B(1) atoms are coordinated by three O atoms to form BO3 triangles while the B(2) atoms are coordinated to four O atoms to form BO4 tetrahedra. The B–O distances range from 1.356(6) Å to 1.501(6) Å. All of the bond lengths are consistent with those observed in other compounds.19,20 The results of bond valence calculations (Na, 0.926–0.961; B, 3.043–3058; Br, 0.890) indicate that the Na, B, and Br atoms are in oxidation states of +1, +3, and −1, respectively.24,37
The difference between the two crystal structures is mainly in the size and the coordination environment of the Cl and Br atoms. In Na3B4O7Cl, the Na–Cl bond lengths are range from 2.725(3) Å to 3.402(5) Å, while in Na3B4O7Br, the Na–Br bond lengths are range from 2.822(3) Å to 3.346(4) Å.
It is of some interest to find that the Na(1) and Cl (Br) atoms have large thermal factors in the two crystals during the refinement (Table 2), either indicating diffuse positioned atoms or site deficiencies. Such phenomenon has been observed in some salt inclusion borates, such as Ba4Ga2B8O18Cl2·NaCl,38 Ba4(BO3)3(SiO4)·Ba3X (X = Cl, Br).39 From the bond length calculation, in Na3B4O7Cl, the Na(1)–O distances are in the range of 2.406(6) Å to 2.940(6) Å with an average length of 2.558(2) Å. It is found that the Na(1)–O bond length of 2.558(2) Å is significantly longer than the sum of their ionic radii (2.40 Å), whereas that of Na(1)–Cl (2.725(3) Å) is much shorter than the sum (2.83 Å). Such observation may indicate that, Na(1) and Cl are more or less forms of the NaCl molecule, and because it is weakly bonded to the rest of the lattice with a bond valence sum for Na(1) of only +0.84 and that for Cl of −0.69. In the same way, the average bond length of the Na(1)–O bonds in Na3B4O7Br is 2.553(7) Å, which is longer than the sum of their ionic radii (2.40 Å), whereas that of Na(1)–Br (2.822(3) Å) is much shorter than the sum (2.98 Å), which further indicate that weak interaction between NaBr and the rest of the lattice. Therefore, from another perspective, Na3B4O7X (X = Cl, Br) also can be regarded as salt inclusion borates.40
IR spectra
Fig. S1 in the ESI† presents the IR spectra of Na3B4O7X (X = Cl, Br) and they are similar. The assignments of IR spectra of the two compounds are listed in Table S4 in the ESI.† Referring to the literatures,41,42 the peaks at 1346–1338 cm−1 and 950 cm−1 can be assigned to the asymmetric stretching and symmetric stretching vibrations of BO3, respectively. The peaks located at 1140 and 862–753 cm−1 arise from the asymmetric stretching and symmetric stretching vibrations of BO4, respectively. The peaks observed in the regions of 663–654 cm−1 are attributed to the out-of-plane bending of BO3. The peaks at 520 cm−1 characterize the bending of BO3 and BO4. The peaks observed in the regions of 475–465 cm−1 are attributed to the bending of BO4. The IR spectra further confirm the existence of the BO3 triangles and BO4 tetrahedra, which is consistent with the results obtained from the single crystal X-ray structural analyses.UV-Vis-NIR diffuse reflectance spectroscopy
The UV-Vis-NIR diffuse reflectance spectra of Na3B4O7X (X = Cl, Br) in the region 190–2600 nm are shown in Fig. S4 in the ESI.† It is clear that the two compounds have no obvious absorption from 340 nm to 2600 nm, but the absorption sharply increases below 340 nm and the cut-off edges for the two compounds are below 200 nm.Thermal analysis
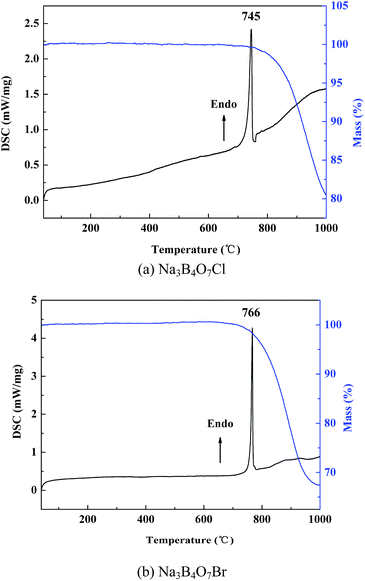
The DSC/TG curves of the two crystals are shown in Fig. 3. It can be seen that there is one endothermic peak at 745 and 766 °C on the DSC curves for Na3B4O7Cl, and Na3B4O7Br, respectively, along with weight loss on the TG curves. Analysis of the powder XRD pattern of the solidified melt reveal that the entire solid product exhibits a diffraction pattern different from that of the initial pure powder for the two compounds, as shown in Fig. 1. It demonstrates that the two compounds are incongruently melting compounds.Theoretical calculations
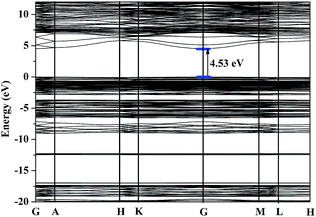
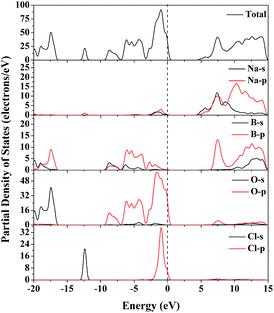
The electronic band structure of Na3B4O7Cl calculated by GGA method is plotted along high symmetry k-points in Fig. 4 (the electronic band structure of Na3B4O7Br is in Fig. S5 in the ESI†). All the two crystals are direct band gap materials with the calculated band gap of 4.53 (Na3B4O7Cl) and 4.16 eV (Na3B4O7Br), which are relatively smaller than experimental optical gaps (>6.2 eV) due to a typical disadvantage in density functional theory (DFT) calculations.The PDOS of Na3B4O7Cl can be divided into four major distinct regions, as can be seen from Fig. 5. The lowest part located from −23.0 eV to −16.0 eV contains contributions mainly from the O2p orbital, with a small amount of B2s and B2p orbitals. The next region, around −12.5 eV, is contributed by Cl3s orbital with very sharp and narrow shape. The third region extends in a wide range from −9.5 eV to VBM, that is, the Fermi level. This band mostly originates from the O2p, B2p and Cl3p states, with some mixing of B2s state. For the lowest conduction band, the main components are the Na2p, Nas, and B2p states. The PDOS of Na3B4O7Br (Fig. S6 in the ESI†) is similar to that of Na3B6O10Cl due to the two crystals having the similar building units and chemical environments.
Conclusion
This study is a continuation of systematic investigation of the alkali-metal borate halides containing XM6 (X = Cl, Br; M = alkali metals) octahedra system. In this system, two new alkali metal borate halides Na3B4O7X (X = Cl, Br) have been reported for the first time. The compounds exhibit a 3D structure consisting of XNa6 (X = Cl, Br) polyhedra and B4O9 groups. The B4O9 group is connected by sharing its four terminal oxygens to form a 3D network with channels pointing along the c axis, where the 1D helical [Na3X] (X = Cl, Br) chains are filled. In addition, the two compounds also can be regarded as NaX salt inclusion borates. The DSC/TG curves show that Na3B4O7X (X = Cl, Br) melt incongruently. The formation of Na3B4O7X (X = Cl, Br) provides another example that alkali-metal borate halides containing XM6 (X = Cl, Br; M = alkali metals) octahedra system possess very extensive crystal chemistry, which continues to provide a rich source of new compounds. In the future, the research on the alkali-metal borate halides containing XM6 (X = Cl, Br; M = alkali metals) octahedra system will be further expanded.Acknowledgements
This work is supported by the “National Natural Science Foundation of China” (Grant nos 51302307, U1303193 51425206, U1129301, 51172277), the Western Light of Chinese Academy of Sciences (Grant no. XBBS201220), the High-level Professional and Technical Personnel of Autonomous region, the National Key Basic Research Program of China (Grant no. 2014CB648400), Major Program of Xinjiang Uygur Autonomous Region of China during the 12th Five-Year Plan Period (Grant no. 201130111).Notes and references
- (a) P. Becker, Adv. Mater., 1998, 10, 979 CrossRef CAS; (b) P. Becker and R. Z. Frohlich, Zeitschrift für Naturforschung B., 2004, 59, 256 CAS; (c) C. T. Chen, B. C. Wu, A. D. Jiang and G. M. You, Sci. Sin., Ser. B, 1985, 28, 235 Search PubMed; (d) H. W. Huang, J. Y. Yao, Z. S. Lin, X. Y. Wang, R. He, W. J Yao, N. X. Zhai and C. T. Chen, Angew. Chem., Int. Ed., 2011, 50, 9141 CrossRef CAS PubMed.
- (a) P. S. Halasyamani and K. R. Poeppelmeier, Chem. Mater., 1998, 10, 2753 CrossRef CAS; (b) D. A. Keszler, A. Akella, K. I. Schaoers and T. Alekel III, Mater. Res. Soc. Symp. Proc., 1994, 329, 15 CrossRef CAS PubMed; (c) S. C. Wang, N. Ye, W. Li and D. Zhao, J. Am. Chem. Soc., 2010, 132, 8779 CrossRef CAS PubMed; (d) S. C. Wang and N. Ye, J. Am. Chem. Soc., 2011, 133, 11458 CrossRef CAS PubMed.
- (a) N. Ye, W. R. Zeng, J. Jiang, B. C. Wu, C. T. Chen, B. H. Feng and X. L. Zhang, J. Opt. Soc. Am. B, 2000, 17, 764 CrossRef CAS; (b) H. W. Yu, S. L. Pan, H. P. Wu, W. W. Zhao, F. F. Zhang, H. Y. Li and Z. H. Yang, J. Mater. Chem., 2012, 22, 2105 RSC; (c) M. Zhang, S. L. Pan, J. Han, Y. Yang, L. Cui and Z. X. Zhou, J. Alloys Compd., 2011, 509, 6696 CrossRef CAS PubMed; (d) H. W. Huang, J. Y. Yao, Z. S. Lin, X. Y. Wang, R. He, W. J. Yao, N. X. Zhai and C. Y. Chen, Chem. Mater., 2011, 23, 5457 CrossRef CAS.
- (a) H. X. Zhang, J. Zhang, S. T. Zheng and G. Y. Yang, Cryst. Growth Des., 2005, 5, 157 CrossRef CAS; (b) G. Z. Liu, S. T. Zheng and G. Y. Yang, Inorg. Chem. Commun., 2007, 10, 84 CrossRef CAS PubMed; (c) B. Petermüller, L. L. Petschnig, K. Wurst, G. Heymann and H. Huppertz, Inorg. Chem., 2014, 53, 9722 CrossRef PubMed; (d) H. Yang, C. L. Hu, J. L. Song and J. G. Mao, RSC Adv., 2014, 4, 45258 RSC.
- (a) S. L. Pan, J. P. Smit, B. Watkins, M. R. Marvel, C. L. Stern and K. R. Poeppelmeier, J. Am. Chem. Soc., 2006, 128, 11631 CrossRef CAS PubMed; (b) A. Q. Jiao, H. P. Wu, S. L. Pan, H. W. Yu and C. Lei, J. Alloys Compd., 2014, 588, 514 CrossRef CAS PubMed; (c) H. P. Wu, H. W. Yu, S. L. Pan, Z. J. Huang, Z. H. Yang, X. Su and K. R. Poeppelmeier, Angew. Chem., Int. Ed., 2013, 52, 3406 CrossRef CAS PubMed; (d) L. L. Liu, X. Su, Y. Yang, S. L. Pan, X. Y. Dong, S. J. Han, M. Zhang, J. Kang and Z. H. Yang, Dalton Trans., 2014, 8905 RSC.
- (a) T. Pilz and M. Jansen, Z. Anorg. Allg. Chem., 2011, 637, 1 CrossRef; (b) T. Sasaki, Y. Mori, M. Yoshimura, Y. K. Yap and T. Kamimura, Mater. Sci. Eng., R, 2000, 30, 1 CrossRef; (c) T. S. Ortner, K. Wurst, L. Perfler, M. Tribus and H. Huppertz, J. Solid State Chem., 2015, 221, 66 CrossRef CAS PubMed; (d) Z. S. Lin, L. Bai, J. Liu, M. H. Lee, J. Xu, X. Y. Wang and C. T. Chen, J. Appl. Phys., 2011, 107, 073721 CrossRef PubMed; (e) Z. S. Lin, L. F. Xu, L. J. Liu, J. Xu, M. H. Lee and C. T. Chen, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 82, 035124 CrossRef.
- (a) Z. Wang, M. Zhang, S. L. Pan, Y. Wang, H. Zhang and Z. H. Chen, Dalton Trans., 2014, 2828 RSC; (b) S. L. Pan, Y. C. Wu, P. Z. Fu, G. C. Zhang, Z. H. Li, C. X. Du and C. T. Chen, Chem. Mater., 2003, 15, 2218 CrossRef CAS; (c) Z. Wang, M. Zhang, X. Su, S. L. Pan, Z. H. Yang, H. Zhang and L. Liu, Chem.–Eur. J., 2015, 21, 1414 CrossRef CAS PubMed; (d) X. W. Zhang, H. W. Yu, H. P. Wu, S. L. Pan, A. Q. Jiao, B. B. Zhang and Z. H. Yang, RSC Adv., 2014, 1, 13195 RSC.
- (a) X. A. Chen, J. L. Zuo, X. A. Chang, Y. H. Zhao, H. G. Zang and W. Q. Xiao, J. Solid State Chem., 2006, 179, 3191 CrossRef CAS PubMed; (b) X. A. Chen, F. P. Song, X. A. Chang, H. G. Zang and W. Q. Xiao, J. Solid State Chem., 2009, 182, 3091 CrossRef CAS PubMed; (c) W. J. Yao, T. Xu, X. X. Jiang, H. W. Huang, X. Y. Wang, Z. S. Lin and C. T. Chen, Dalton Trans., 2014, 9998 RSC; (d) S. L. Pan, B. Watkins, J. P. Smit, M. R. Marvel, I. Saratovsky and K. R. Poeppelmeier, Inorg. Chem., 2007, 46, 3851 CrossRef CAS PubMed.
- Y. Z. Huang, L. M. Wu, X. T. Wu, L. H. Li, L. Chen and Y. F. Zhang, J. Am. Chem. Soc., 2010, 132, 12788 CrossRef CAS PubMed.
- C. T. Chen and G. Z. Liu, Annu. Rev. Mater. Sci., 1986, 16, 136 CrossRef.
- (a) C. T. Chen, J. H. Lu, T. Togashi, T. Suganuma, T. Sekikawa, S. Watanabe, Z. Y. Xu and J. Y. Wang, Opt. Lett., 2002, 27, 637 CrossRef CAS; (b) B. C. Wu, D. Y. Tang, N. Ye and C. T. Chen, Opt. Mater., 1996, 5, 105 CrossRef CAS; (c) C. T. Chen, G. L. Wang, X. Y. Wang and Z. Y. Xu, Appl. Phys. B, 2009, 97, 9 CrossRef CAS.
- C. Chen, Y. Wu, A. Jiang, G. You, R. Li and S. Lin, J. Opt. Soc. Am. B, 1989, 6, 616 CrossRef CAS.
- Y. Wu, T. Sasaki, S. Nakai, A. Yokotani, H. Tang and C. Chen, Appl. Phys. Lett., 1993, 62, 2614 CrossRef CAS PubMed.
- Y. Mori, I. Kuroda, S. Nakajima, T. Sasaki and S. Nakai, Appl. Phys. Lett., 1995, 67, 1818 CrossRef CAS PubMed.
- Y. Yang, S. L. Pan, H. Y. Li, J. Han, Z. H. Chen, W. W. Zhao and Z. X. Zhou, Inorg. Chem., 2011, 50, 2415 CrossRef CAS PubMed.
- Y. Yang, S. L. Pan, J. Han, X. L. Hou, Z. X. Zhou, W. W. Zhao, Z. H. Chen and M. Zhang, Cryst. Growth Des., 2011, 11, 3912 CAS.
- J. G. M. Jesudurai, K. Prabha, P. D. Christy, J. Madhavan and P. Sagayaraj, Spectrochim. Acta, Part A, 2008, 71, 1371 CrossRef PubMed.
- (a) H. P. Wu, S. L. Pan, K. R. Poeppelmeier, H. Y. Li, D. Z. Jia, Z. H. Chen, X. Y. Fan, Y. Yang, J. M. Rondinelli and H. S. Luo, J. Am. Chem. Soc., 2011, 133, 7786 CrossRef CAS PubMed; (b) H. P. Wu, S. L. Pan, H. W. Yu, D. Z. Jia, A. M. Chang, H. Y. Li and X. Huang, CrystEngComm, 2012, 14, 799 RSC; (c) M. Zhang, S. L. Pan, X. Y. Fan, Z. X. Zhou, K. R. Poeppelmeier and Y. Yang, CrystEngComm, 2011, 13, 2899 RSC.
- (a) C. Y. Bai, H. W. Yu, S. J. Han, S. L. Pan, B. B. Zhang, Y. Wang, H. P. Wu and Z. H. Yang, Inorg. Chem., 2014, 53, 11213 CrossRef CAS PubMed; (b) Z. H. Chen, S. L. Pan, X. Y. Dong, Z. H. Yang, M. Zhang and X. Su, Inorg. Chim. Acta, 2013, 406, 205 CrossRef CAS PubMed.
- S. J. Han, Y. Wang, S. L. Pan, X. Y. Dong, H. P. Wu, J. Han, Y. Yang, H. W. Yu and C. Y. Bai, Cryst. Growth Des., 2014, 14, 1794 CAS.
- SAINT, Version 7.60 A, Bruker Analytical X-ray Instruments, Inc., Madison, WI, 2008 Search PubMed.
- G. M. Sheldrick, SHELXTL, Version 6.14, Bruker Analytical X-ray Instruments, Inc., Madison, WI, 2003 Search PubMed.
- A. L. Spek, J. Appl. Crystallogr., 2003, 36, 7 CrossRef CAS.
- N. E. Brese and M. O'Keeffe, Acta Crystallogr., Sect. B: Struct. Sci., 1991, 47, 192 CrossRef.
- J. Tauc, Mater. Res. Bull., 1970, 5, 721 CrossRef CAS.
- (a) H. W. Yu, S. L. Pan, H. P. Wu, Z. H. Yang, L. Y. Dong, X. Su, B. B. Zhang and H. Y. Li, Cryst. Growth Des., 2013, 13, 3514 CrossRef CAS; (b) Y. J. Shi, S. L. Pan, X. Y. Dong, Y. Wang, M. Zhang, F. F. Zhang and Z. X. Zhou, Inorg. Chem., 2012, 51, 10870 CrossRef CAS PubMed; (c) L. Zhou, S. L. Pan, X. Y. Dong, H. W. Yu, H. P. Wu, F. F. Zhang and Z. X. Zhou, CrystEngComm, 2013, 15, 3412 RSC.
- S. J. Clark, M. D. Segall, C. J. Pickard, P. J. Hasnip, M. J. Probert, K. Rrfson and M. C. Payne, Z. Kristallogr., 2005, 220, 567 CrossRef CAS.
- (a) M.-H. Lee, Ph.D. Thesis, The University of Cambridge, 1996; (b) J. Lin, A. Qteish, M. Payne and V. Heine, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47, 4174 CrossRef; (c) A. M. Rappe, K. M. Rabe, E. Kaxiras and J. D. Joannopoulos, Phys. Rev. B: Condens. Matter Mater. Phys., 1990, 41, 1227 CrossRef.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS.
- (a) J. D. Grice, P. C. Burns and F. C. Hawthorne, Can. Mineral., 1999, 37, 731 CAS; (b) P. C. Burns, J. D. Grice and F. C. Hawthorne, Can. Mineral., 1995, 33, 1131 CAS.
- H. A. Levy and G. C. Lisensky, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1978, 34, 3502 Search PubMed.
- P. B. Moore and T. Araki, Am. Mineral., 1974, 59, 60 CAS.
- P. Becker and R. Froehlich, Z. Kristallogr., 2001, 216, 31 CrossRef CAS.
- X. Xu, C. L. Hu, F. Kong, J. L. Zhang, J. G. Mao and J. L. Sun, Inorg. Chem., 2013, 52, 5831 CrossRef CAS PubMed.
- J. Krogh-Moe, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1968, 24, 179 CrossRef CAS.
- M. Maczka, A. Waśkowska, A. Majchrowskib, J. Żmija, J. Hanuza, G. A. Petersond and D. A. Keszler, J. Solid State Chem., 2007, 180, 410 CrossRef CAS PubMed.
- I. D. Brown and D. Altermatt, Acta Crystallogr., Sect. B: Struct. Sci., 1985, 41, 244 CrossRef.
- X. X. Lin, F. F. Zhang, S. L. Pan, H. W. Yu, F. Y. Zhang, X. Y. Dong, S. J. Han, L. Y. Dong, C. Y. Bai and Z. Wang, J. Mater. Chem. C, 2014, 2, 4257 RSC.
- R. K. Li and Y. Yu, Inorg. Chem., 2006, 45, 6840 CrossRef CAS PubMed.
- (a) J. P. West and S.-J. Hwu, J. Solid State Chem., 2012, 195, 101 CrossRef CAS PubMed; (b) X. Mo and S.-J. Hwu, Inorg. Chem., 2003, 42, 3978 CrossRef CAS PubMed; (c) Q. Huang and S.-J. Hwu, Inorg. Chem., 2003, 42, 655 CrossRef CAS PubMed.
- W. W. Zhao, S. L. Pan, J. Han, J. Y. Yao, Y. Yang, J. J. Li, M. Zhang, L. H. Zhang and Y. Hang, J. Solid State Chem., 2011, 184, 2849 CrossRef CAS PubMed.
- Y. J. Wang, S. L. Pan, X. L. Tian, Z. X. Zhou, G. Liu, J. Wang and D. Z. Jia, Inorg. Chem., 2009, 48, 7800 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Selected bond lengths (Å) and angles (degree); the element analysis of Na3B4O7X (X = Cl, Br); the assignment of infrared spectra; Infrared spectra; the coordination environments of the cations in Na3B4O7Br; the boron–oxygen framework in the crystal structure of Na3B4O7Br in bc plane; UV-Vis-NIR diffuse-reflectance spectra; calculated band structure of Na3B4O7Br; the total and partial densities of states of Na3B4O7Br. CCDC 1039806 and 1039807. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra16639f |
| This journal is © The Royal Society of Chemistry 2015 |