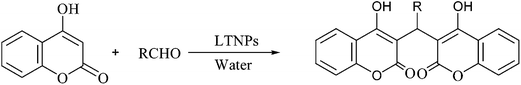
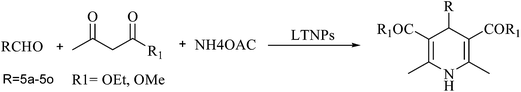L-Tyrosine loaded nanoparticles: an efficient catalyst for the synthesis of dicoumarols and Hantzsch 1,4-dihydropyridines†
Anamika Khaskela,
Pranjit Barman*a and
Utpal Janab
aDepartment of Chemistry, National Institute of Technology, Silchar 788010, Assam, India. E-mail: barmanpranjit@yahoo.co.in; Fax: +91 3842 224797
bSchool of Pharmacy, Chouksey Engineering College, Lal Khadan, NH-49, Bilaspur 495004, India
First published on 19th January 2015
Abstract
Environmentally benign L-tyrosine loaded nanoparticles are fabricated and characterized by PCS, TEM, FT-IR and AFM studies. A novel straightforward green approach was applied for the synthesis of dicoumarols and Hantzsch 1,4-dihydropyridines using this catalyst. The structures and purity of these compounds were confirmed by FT-IR and NMR (1H, 13C and DEPT). The flexible and swelling properties of the polymer coating increase L-tyrosine dispersion and its high catalytic activity in organic reactions.
Introduction
Implementation of several transformations through a multi-component reaction strategy is highly compatible with the goal of green chemistry.1 Use of environmentally benign solvents like water2 and solvent-free reactions3 offers a powerful and green protocol from both synthetic and economical points of view. It has several advantages like reduced pollution, simplicity of processing and low cost, which is useful to the industry as well as for the environment.Dicoumarols and their derivatives are of interest because of their anticoagulant (Antivitamin K activity), spasmolytic and rodenticidal activities.4 Chemically, it is designated as 3,3′-methylenebis[4-hydroxycoumarin]. Dicoumarol and its synthetic derivative warfarin sodium (Coumadin) help to decrease metastases in animal model.5 Recently, Xian-Xing Luo and his co-workers reported the anti-bacterial activity of pyridine substituted dicoumarols.6 Similarly, Hantzsch 1,4-dihydropyridines (1,4-DHPs) have remarkable pharmacological efficiency.7 The ferrocene moiety is known to play an important role in organometallic drugs because of its high affinity towards amino acids, proteins, DNA and carbohydrates.8 Therefore, the attempts to modify the conditions of Hantzsch reaction are still of growing importance. Dicoumarols have been synthesized using various catalysts such as Zn(proline)2,9 molecular iodine,10 sulphated titania,11 SDS,12 MnCl2,13 Biodegradable Task-specific ionic liquid,14 nano silica chloride15 etc. For Hantzsch's 1,4-DHPs synthesis Several modified methods have been reported nowadays for example such as free nano Fe2O3,16 Zn(L-proline)2,17 PPh3,18 L-proline under ultrasound condition19 etc. Furthermore, ionic liquid such as glycine nitrate,20 Ni2+ containing ionic liquid,21a Silica functionalized sulphonic acid coated with ionic liquid,21b enzymes such as Baker's yeast,22 lipase B23 demonstrated catalytic activity in 1,4-DHPs synthesis. Some heterogeneous catalysts have also been reported such as sulfonic acid supported γ-Fe2O3,24 cellulose,25 alginic acid.26
Two recent studies have established L-tyrosine as an efficient organocatalyst in multi-component reactions for green synthesis.27 Thitherto, there was no report of any catalytic activity of this amino acid. Researchers in the past few years have investigated the pharmacological importance L-tyrosine loaded nanoparticles (LTNPs). It has been found to increase the antitumoural activity of direct electric current in a metastatic melanoma cell model.28 Beside, L-tyrosine polyphosphate nanoparticles are also used in gene therapy.29a So these studies reveal that encapsulating L-tyrosine inside nano-sized polymeric coating possibly increases its pharmacological activity. Consequently, the next question that arises is whether this encapsulation of L-tyrosine affects its ability to catalyze organic reactions and if yes, in which direction. The fact that L-proline functionalized polymeric nanoreactors have been used as catalyst in Aldol reaction,29b makes the above question more pertinent. With this aim, LTNPs have been used as catalyst for preparation of dicoumarols and Hantzsch 1,4-DHPs following a fully green synthetic method. To the best of our knowledge, this is the first report of LTNPs acting as catalyst in any organic reaction. The products are fully characterized by FT-IR, 1H NMR, 13C NMR, DEPT NMR and also by comparison with authentic samples (Scheme 1).
Results and discussions
LTNPs were prepared with polymer Eudragit® RS100 using the solvent evaporation (single emulsion) technique with slight modification as previously reported by Jana et al.30 Eudragit is the co-polymer of poly(ethylacrylate, methyl-methacrylate and chlorotrimethyl-ammonioethyl methacrylate) containing quaternary ammonium group.30 The ammonium groups are present as salts and make the polymers permeable. This polymer has high permeability and pH independent swelling properties. The prepared nanoparticles (ESI-1,† for details) are characterized by particle size analysis, TEM (average size 50 nm), FT-IR and AFM studies (ESI-2†) (Scheme 2).The C-3 position of 4-hydroxycoumarin (1) ring is highly reactive as it is flanked by electron donating hydroxyl group C-4 and electron withdrawing carbonyl group at C-2. As the carbon–carbon double bond and lone pair of electrons present on the oxygen atom of the OH group is in conjugation, this make coumarin ring very convenient at position 3 to react with carbonyl carbon of the aldehydes. In Scheme 3 we have mentioned the plausible mechanism for the synthesis of various dicoumarols using LTNPs as catalyst. At the outset, we tried the condensation of 4-methoxy benzaldehyde and 4-hydroxy coumarin as a model reaction to synthesize the dicoumarol (3a) using LTNPs catalyst in water at 70 °C in a water bath. A broad range of dicoumarols (3a–3j) have been synthesized. In a comparative study a large number of catalysts were used to show the effectiveness of LTNPs over other catalysts (Table 1). Increment in the amount of LTNPs catalyst to 0.05 g did not show any improvement in the yield (Table 1, Entry-7) whereas, the yield was found to be lesser when the catalyst loading reduced to 0.01 g (Table 1, Entry-8).
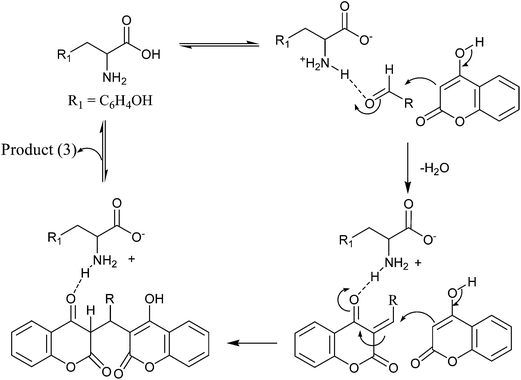 | ||
| Scheme 3 Plausible mechanism for the synthesis of dicoumarols using LNTPs (LTNPs dispersed through Eudragit coating). | ||
| Entry | Catalyst | Catalyst loading (g) | Time | Yieldb (%) |
|---|---|---|---|---|
a Reaction temperature: 70 °C; molar ratio of aldehydes![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4-hydroxycoumarin: 1 4-hydroxycoumarin: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.b Isolated yields. 2.b Isolated yields. |
||||
| 1 | PTS | 0.02 | No reaction | NA |
| 2 | Glycine | 0.02 | 1.5 h | 65 |
| 3 | L-Proline | 0.02 | 2 h | 50 |
| 4 | L-Serine | 0.02 | 20 min | 72 |
| 5 | L-Tyrosine | 0.02 | 15 min | 75 |
| 6 | LTNPs | 0.02 | 5 min | 90 |
| 7 | LTNPs | 0.05 | 5 min | 90 |
| 8 | LTNPs | 0.01 | 15 min | 78 |
The next aim was to look for a suitable medium in which the chosen catalyst, i.e. LTNPs provides high yield in short reaction time. Both protic as well as aprotic solvents were tried and the former type proved to be way more effective (Table 2) both in terms of product yield and reaction time.
Among the three protic solvents tried, an interesting trend was observed which made our choice of reaction medium very easy and straightforward. With increment in the dielectric constant of solvents the catalyst was found to give higher yield in lesser time, and when the yield to time ratio was plotted against the dielectric constants of the solvents, it showed a monotonic relationship between the two (Fig. 1).
 | ||
| Fig. 1 Relative efficiency of three protic solvents as reaction medium for dicoumarol (3a) synthesis using LTNPs. | ||
The highest yield (90%) in shortest period (5 minutes) was obtained for the solvent with highest dielectric constant among the three solvents tried, water. Considering the undeniable importance of water as solvent in organic reactions when the primary goal is search for a green synthetic protocol, it was promptly chosen as the solvent for dicoumarol synthesis. A range of dicoumarols were synthesized using the various aldehydes and 4-hydroxycoumarin. The results are summarized in Table 3.
Regardless of substitution (electron withdrawing and electron donating) of the aromatic aldehydes, the products were obtained in good yields. Similar results were also obtained with the heterocyclic aldehydes. The excellent catalytic performance of this catalyst prompted us to explore its further applications toward the synthesis of various substituted 1,4-DHPs under solvent-free condition at room temperature stirring.
Here too, a number of different catalysts were tried and LTNPs was easily the best of the lot in terms of both yields and time (Table 4).
Once the reaction condition was established, the scope of this reaction was investigated with various aldehydes and β-ketoesters yielding a variety of highly functionalized 1,4-DHPs (Table 5, 5a–5o). In Scheme 4 we have mentioned the plausible mechanism for the synthesis of various 1,4-DHPs using LTNPs as catalyst.
| Entry | Aldehyde | 1,3-Diketone | Product | Timeb (min) | Yieldc (%) |
|---|---|---|---|---|---|
a Reaction conditions – aldehyde![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1,3-diketone 1,3-diketone![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ammonium acetate = 1 ammonium acetate = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; catalyst loading = 0.02 g.b Reaction progress monitored by TLC.c Isolated yields. 1; catalyst loading = 0.02 g.b Reaction progress monitored by TLC.c Isolated yields. |
|||||
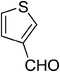
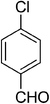
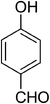
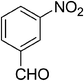
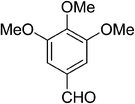
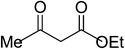
| 1 | 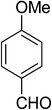 |
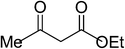 |
5a | 10 | 91 |
| 2 | 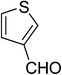 |
 |
5b | 15 | 85 |
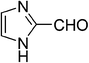
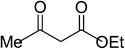
| 3 | 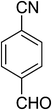 |
 |
5c | 5 | 89 |
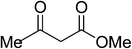
| 4 | Ferrocene-3-carboxaldehdye | 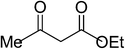 |
5d | 15 | 91 |
| 5 | Ferrocene-3-carboxaldehyde | 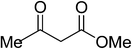 |
5e | 15 | 90 |
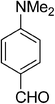
| 6 | 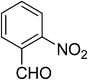 |
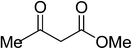 |
5f | 10 | 91 |
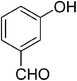
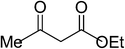
| 7 | 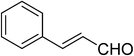 |
 |
5g | 12 | 94 |
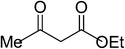
| 8 | 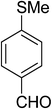 |
 |
5h | 15 | 85 |
| 9 | 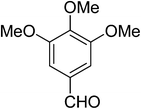 |
 |
5i | 10 | 87 |
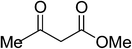
| 10 | 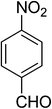 |
 |
5j | 5 | 93 |
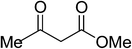
| 11 | 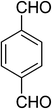 |
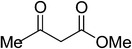 |
5k | 10 | 89 |
| 12 | 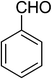 |
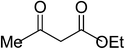 |
5l | 7 | 92 |
| 13 | 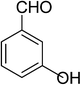 |
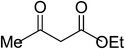 |
5m | 10 | 90 |
| 14 | 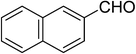 |
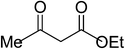 |
5n | 15 | 86 |
| 15 | 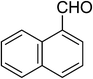 |
 |
5o | 15 | 85 |
 | ||
| Scheme 4 Plausible mechanism for the synthesis of 1,4-DHPs using LNTPs (LTNPs dispersed through Eudragit coating). | ||
When reaction was carried out with ferrocenyl aldehydes to synthesize their corresponding 1,4-DHPs using free L-tyrosine as catalyst, it resulted in moderate yields (72%) in relatively longer time (25 min). Replacing it with LTNPs showed striking improvements in both yield and time (5d and 5e). Owing to the potential of ferrocene moiety to be used in drugs, a fast, easy and green method for synthesizing 1,4-DHPs possessing ferrocenyl side chains can lead up to much easier synthetic protocols for large scale production of important drug molecules. The reduced particle size and improved surface area of the L-tyrosine loaded polymeric nanoparticulate system increase the penetrability of L-tyrosine in the reaction mixture which in turns gives the high yields. Moreover the polymer Eudragit® RS100 has the ability of swelling, which represents the good material for L-tyrosine dispersion.
Experimental
All commercially available chemicals were obtained from Merck and Aldrich, and used without further purifications. Melting points were uncorrected. IR spectra were recorded as KBr pellets on a Perkin Elmer 782 spectrophotometer. The 1H NMR and 13C NMR spectra in DMSO were run on a Bruker AM-300L instrument operating at 300 MHz and 75 MHz respectively.a. Materials
L-Tyrosine and Lutrol® F-68 (Poloxamer 188) were obtained from Sigma, Mumbai. Eudragit® RS100 (Evonik Industries AG, Germany) was obtained from Sandoz Ltd. Mumbai. Distilled-deionized water was prepared with Milli-Q plus System (Elix 10, Millipore corp. India). All other chemicals used were of the highest available grade.b. Catalyst preparation
The L-tyrosine loaded-polymeric nanoparticles were prepared with polymer Eudragit® RS100 using the solvent evaporation (single emulsion) technique with slight modification.30 In brief, the polymeric solution was prepared by adding 100 mg of Eudragit® RS100 in the mixture of methanol and acetone (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 v/v) at room temperature. Weighed quantity of L-tyrosine (equivalent to 10% w/w dry weight of polymer) was dissolved in 1(N) HCl and added to the polymeric solution. The resultant solution was poured into 25 ml of aqueous phase containing 1% (w/v) of poloxamer-188 with a constant flow rate of 1 ml min−1. The mixture was then homogenized using a probe homogenizer (VIRTIS, Cyclone IQ, USA), at constant agitation speeds of 10
80 v/v) at room temperature. Weighed quantity of L-tyrosine (equivalent to 10% w/w dry weight of polymer) was dissolved in 1(N) HCl and added to the polymeric solution. The resultant solution was poured into 25 ml of aqueous phase containing 1% (w/v) of poloxamer-188 with a constant flow rate of 1 ml min−1. The mixture was then homogenized using a probe homogenizer (VIRTIS, Cyclone IQ, USA), at constant agitation speeds of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm in an ice bath. The formed emulsion was kept at room temperature under gentle stirring for 24 h to evaporate the organic solvents. The prepared polymeric nanoparticles were centrifuged at 18
000 rpm in an ice bath. The formed emulsion was kept at room temperature under gentle stirring for 24 h to evaporate the organic solvents. The prepared polymeric nanoparticles were centrifuged at 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, for 15 min (Sorvall Ultracentrifuge, USA). The nanoparticle was recovered and freeze dried for 2 days (−80 °C and <10 mm mercury pressure, Freezone 6lt, Labconco Corp., MO) to get powdered nanoparticles and stored in freeze.
000 rpm, for 15 min (Sorvall Ultracentrifuge, USA). The nanoparticle was recovered and freeze dried for 2 days (−80 °C and <10 mm mercury pressure, Freezone 6lt, Labconco Corp., MO) to get powdered nanoparticles and stored in freeze.
c. Characterization of nanoparticles
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio before examination. One drop of sample was placed for one minute on a copper grid coated with a formvar carbon film. The excess of sample was wicked away with the aid of filter paper. The sample was then ready for analysis by TEM.
1 ratio before examination. One drop of sample was placed for one minute on a copper grid coated with a formvar carbon film. The excess of sample was wicked away with the aid of filter paper. The sample was then ready for analysis by TEM.Conclusions
In summary, we have demonstrated that LTNPs as an organocatalyst in dicoumarols and 1,4-DHPs synthesis expecting advantages in (i) L-tyrosine can easily disperse through the flexible thin polymer coating (ii) increment in surface area (iii) easy separation of the catalyst (iv) fully green methodology and (v) very small catalyst loading is sufficient for reactions. Henceforth, this new catalyst works well in various organic transformations under green methodology.Acknowledgements
AK thanks NIT Silchar for financial support and Dr Biman Bandyopadhyay, IEM, Kolkata for many helpful discussions.References
- (a) B. B. Toure and D. G. Hall, Chem. Rev., 2009, 109, 4439 CrossRef CAS PubMed; (b) J. D. Sunderhaus and S. F. Martin, Chem.–Eur. J., 2009, 15, 1300 CrossRef CAS PubMed.
- (a) C. J. Li and T. H. Chan, Organic Reactions in Aqueous Media, John Wiley & Sons, New York, 1997 Search PubMed; (b) Organic Synthesis in Water, ed. P. A. Grieco, Blackie Academic and Professional, London, 1998 Search PubMed; (c) U. M. Lindstrom, Chem. Rev., 2002, 102, 2751 CrossRef PubMed.
- (a) B. C. Ranu and K. Chattopadhyay, in Eco-Friendly Synthesis of Fine Chemicals, ed. R. Ballini, Royal Society of Chemistry, Cambridge, UK, 2009, ch. 5 Search PubMed; (b) R. A. Sheldon, Green Chem., 2005, 7, 267 RSC; (c) T. Chatterjee, S. Bhadra and B. C. Ranu, Green Chem., 2011, 13, 1837 RSC; (d) T. Erdmenger, C. G. Sanchez, J. Vitz, R. Hoogenboom and U. S. Schubert, Chem. Soc. Rev., 2010, 39, 3317 RSC.
- (a) A. Stahhman, M. Ikawa and K. P. Link, US Pat., 2427578, 1947Chem. Abstr., 1948, 42, P603h; (b) E. Boschetti, D. Molho, and L. Fontaine, S. African 68 01, 383, 02 August 1968; E. Boschetti, D. Molho and L. Fontaine, Fr Appl. 20, November 1967, p. 15; (c) LIPHA (Lyonnaise Industrielle Pharmaceutique), Brit. 1,252,088 (Cl. C07d, A61k, A01n), 03 Nov 1968; 11.
- G. F. Smith, B. L. Neubauer, J. L. Sundboom, K. L. Best, R. L. Goode, L. R. Tanzer, R. L. Merriman, J. D. Frank and R. G. Herrmann, Thromb. Res., 1988, 50, 163 CrossRef CAS.
- J. Li, Z. Hou, G. H. Chen, F. Li, Y. Zhou, X. Y. Xue, Z. P. Li, M. Jia, Z. D. Zhang, M. K. Li and X. X. Luo, Org. Biomol. Chem., 2014, 12, 5528 CAS.
- G. M. Reddy, M. Shiradkar and A. K. Chakravarthy, Curr. Org. Chem., 2007, 11, 484 Search PubMed.
- (a) C. S. Allardyce, A. Dorcier, C. Scolaro and P. J. Dyson, Appl. Organomet. Chem., 2005, 19, 1 CrossRef CAS; (b) D. R. van Staveren and N. Metzler-Nolte, Chem. Rev., 2004, 104, 5931 CrossRef CAS PubMed.
- Z. N. Siddiqui and F. Farooq, Catal. Sci. Technol., 2011, 1, 810 Search PubMed.
- M. Kidwai, V. Bansal, P. Mothsra, S. Saxena, R. K. Somvanshi, S. Dey and T. P. Singh, J. Mol. Catal. A: Chem., 2007, 268, 76 CrossRef CAS PubMed.
- B. Karmakar, A. Nayek and J. Banerji, Tetrahedron Lett., 2012, 53, 4343 CrossRef CAS PubMed.
- H. Mehrabi and H. Abusaidi, J. Iran. Chem. Soc., 2010, 7, 890 CrossRef CAS.
- J. N. Sangshetti, N. D. Kokare and D. B. Shinde, Green Chem. Lett. Rev., 2009, 2, 233 CrossRef CAS.
- A. Tzani, A. Douka, A. Paradopoulos, E. A. Pavlatou, E. Voutsas and A. Detsi, ACS Sustainable Chem. Eng., 2013, 1, 1180 CrossRef CAS.
- R. Karimian, F. Piri, A. A. Safari and S. J. Davarpanah, J Nanostruct. Chem., 2013, 3, 52 CrossRef.
- N. Koukabi, E. Kolvari, A. Khazaei, M. A. Zolfigol, B. Shirmardi-Shaghasemic and H. R. Khavasid, Chem. Commun., 2011, 47, 9230 RSC.
- V. Sivamurugan, R. S. Kumar, M. Palanichamy and V. Murugesan, J. Heterocycl. Chem., 2005, 42, 969 CrossRef CAS.
- A. Debache, W. Ghalem, R. Boulcina, A. Belfaitah, S. Rhouati and B. Carboni, Tetrahedron Lett., 2009, 50, 5248 CrossRef CAS PubMed.
- S. Guo and Y. Yuan, Chin. J. Chem., 2010, 28, 811 CrossRef CAS.
- R. Kumar, N. H. Andhare, A. Shard, Richaa and A. K. Sinha, RSC Adv., 2014, 4, 19111 RSC.
- (a) J. Safari and Z. Zarnegar, RSC Adv., 2013, 3, 26094 RSC; (b) P. Sharma and M. Gupta, Green Chem., 2015 10.1039/c4gc00923a.
- J. H. Lee, Tetrahedron Lett., 2005, 46, 7329 CrossRef CAS PubMed.
- J. L. Wang, B. K. Liu, C. Yin, Q. Wu and X. F. Lin, Tetrahedron, 2011, 67, 2689 CrossRef CAS PubMed.
- N. Koukabi, E. Kolvari, M. A. Zolfigol, A. Khazaei, B. S. Shaghasemi and B. Fasahati, Adv. Synth. Catal., 2012, 354, 2001 CrossRef CAS.
- J. Safari, S. H. Banitaba and S. D. Khalili, J. Mol. Catal. A: Chem., 2011, 335, 46 CrossRef CAS PubMed.
- M. G. Dekamin, S. Ilkhanizadeh, Z. Latifidoost, H. Daemi, Z. Karimi and M. Barikani, RSC Adv., 2014, 4, 56658 RSC.
- (a) A. Khaskel, P. Gogoi, P. Barman and B. Bandyopadhyay, RSC Adv., 2014, 4, 35559 RSC.
- V. Campos, C. Teixeira, V. Veiga, E. R. Junior and C. Holandino, Int. J. Nanomedicine, 2010, 5, 961 CAS; G. Thirupathi, M. Venkatanarayana, P. K. Dubey and Y. Bharathi Kumari, Der. Phama. Chemica., 2012, 4, 1897 Search PubMed.
- (a) A. J. Ditto, J. J. Reho, K. N. Shah, J. A. Smolen, J. H. Holda, R. J. Ramirez and Y. H. Yun, Mol. Pharm., 2013, 10, 1836 CrossRef CAS PubMed; (b) A. Lu, P. Cotanda, J. P. Patterson, D. A. Longbottom and R. K. O'Reilly, Chem. Commun., 2012, 48, 9699 RSC.
- U. Jana, A. K. Mohanty, P. K. Manna and G. P. Mohanta, Colloids Surf., B, 2014, 113, 269 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra16627b |
| This journal is © The Royal Society of Chemistry 2015 |