Sustainable synthesis of hollow Cu-loaded poly(m-phenylenediamine) particles and their application for arsenic removal†
Shuo Dai‡
a,
Bing Peng‡ab,
Liyuan Zhang‡a,
Liyuan Chaiab,
Ting Wanga,
Yun Menga,
Xiaorui Lia,
Haiying Wang*ab and
Jian Luoc
aSchool of Metallurgy and Environment, Central South University, Changsha 410071, China. E-mail: haiyw25@yahoo.com; zhang_livyl@csu.edu.cn; Fax: +86-0731-88710171; Tel: +86-0731-88836804
bNational Research Centre for Heavy Metal Pollution Prevention & Control, Changsha 410017, China. E-mail: haiyw25@163.com
cSchool of Civil and Environmental Engineering, Georgia Institute of Technology, Atlanta, GA 30332-0355, USA
First published on 20th March 2015
Abstract
A new Cu-catalyzed air oxidation method was successfully developed to prepare Cu-loaded poly(m-phenylenediamine) (PmPD) with monomer conversion rates close to 100%. The polymerization process was examined by the in situ tracking of open-circuit potential (OCP) and pH. The product was characterized with scanning electron microscopy (SEM), transmission electron microscopy (TEM), Fourier transform infrared spectroscopy (FTIR), X-ray photoelectron spectroscopy (XPS), X-ray diffraction (XRD). The results show that the Cu catalytic effect in the air oxidation was responsible for the great enhancement of monomers conversion rates. On increasing Cu addition, PmPD particles tended to form hollow microstructures, which may result from a dehydration effect. Furthermore, the as-obtained samples have superior arsenic(V) removal performance in aqueous solution. The adsorption equilibrium can be rapidly reached within 10 min. Higher loadings of Cu particles exhibited an improved arsenic(V) removal capability of 27.4 mg g−1, much higher than in other reports. The adsorption behavior can be well described by Freundlich and pseudo-second-order models. The related possible mechanisms have been explored carefully.
Introduction
As a diamine derivative of polyaniline, poly(phenylenediamine) has many important applications in sensors, catalysis, electrodes and actuators, etc.1–8 In particular, widespread attention has been attracted to investigate the adsorption performance of poly(phenylenediamine) over recent decades due to its superior redox reversibility and chelation ability,9–12 and it can be used for removing various metals ions from water, such as Cr(VI),13 Hg(II)14 and Ag(I).15 Among the three isomers of poly(phenylenediamine)s, poly(m-phenylenediamine) (PmPD) as an adsorbent possesses some important virtues: (i) it is insoluble in common solvent, (ii) has high water permeability, (iii) can be produced in high yields, (vi) and its synthesis doesn't involve high temperature or acidic solvents. Many developments of poly(m-phenylenediamine) have been made by Li and our groups, in terms of molecular structures,16 monomer conversion efficiency17 and morphology.15 The improved nanoscale particles exhibit superior adsorption capability of Ag(I) (2359.3 mg g−1)15 and Orange G (387.6 mg g−1).17Generally, the conventional chemically oxidative polymerization is the most accepted method for synthesizing polyaniline and its derivatives, due to its mass production and simple processes. However, introducing a large amounts of costly oxidants (e.g. persulfate) is the prerequisite to initiate the polymerization, which definitely give rise to concentrated salt water (e.g., Na2SO4)18 with plenty of unreacted monomers or even toxic oligomers dissolved in bulk solution after polymerization.19,20 To prevent the mass production of sulfates, adopting green oxidants is another effective strategy, such as H2O2 or O2. Nevertheless, the synthesis yields in previous reports remained quite low (generally not exceeding 40%), which meant more abundant toxic organic chemicals left in the bulk system.21–23 To date, it is still a challenge problem to improve polymerization efficiency of mPD.
In this context, it is reasonable to conclude that a sustainable synthesis of conjugated polymers, including the poly(m-phenylenediamine), must involve two indispensable elements: (i) side-products from oxidant decomposition should be totally harmless and (ii) ultrahigh conversion rate that leaves nearly no toxic organic chemicals in polymerization solution. Unfortunately, seldom synthesis has fulfilled these two requirements.
In present research, we developed a sustainable synthesis of poly(m-phenylenediamine) microparticles with hollow structures via Cu-catalyzed air oxidation. The conversion efficiency of monomer in this research can reach close to 100%, which is higher than the highest yield reported (93.1%).24 More profoundly, the nanoscaled Cu-loaded PmPD particle is a hopeful material in arsenic removal in aqueous solution. This study is the first to present detailed investigations of As(V) removal performance and mechanisms by Cu-loaded PmPD particles.
Experimental
Materials
The chemicals were of analytical grade. CuCl2·2H2O and Na3AsO4·12H2O were purchased from Sinopharm Chemical (Shanghai, China), while the m-phenylenediamine (mPD) was from Acros (Belgium). Organic solvents, such as ethanol (EtOH), dimethylformamide (DMF), N-methyl-2-pyrrolidone (NMP) and dimethyl sulfoxide (DMSO) were from Hui-Hong Chemical (Hunan, China).Preparation of poly(m-phenylenediamine) particles
Typical synthetic processes of poly(m-phenylenediamine) (PmPD) hollow sub-microparticles were as follows: 3.0 g mPD (monomer) was dissolved into 100 mL aqueous solution to form the monomer solution in 250 mL single-neck flat bottom flask. The monomer solution was pre-warmed to 30 °C and kept stirring by an IKA RCT basic magnetic stirrer at a speed of 1000 r s−1. Then 10 mL of solution with a specific concentration of Cu2+ (its temperature was also 30 °C) was added to the monomer solution in one pot. And air was introduced into the mixture as soon as the addition of Cu2+ solution. And air velocity was 40 L min−1 by air pump and rubber hose was used to connect reaction system with air pump (ESI-1†). Then, the reaction system was kept on stirring with air for 24 hours. The equipment and synthesis sketch map is illustrated in ESI-2.† The resulting mixture was separated by filtration and rinsed with to remove the impurity by using sand cored funnel (G5) which pore size was about 1.5–2.5 μm. During filtration, used 50 mL distilled water to wash the reaction vessel and 50 mL to rinse PmPD particles, and then took this 100 mL filtrate and the initial filtrate of 100 mL, a total of 200 mL, to measure total organic carbon (TOC) and Cu mass. The product was dried at 60 °C in air for more than 12 h. The PmPD particles was named as PmPD-Cux (x: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, 1
0.5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25, 1
0.25, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.125 and 1
0.125 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1), where x corresponds to the mPD
0.1), where x corresponds to the mPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu2+ molar ratio and y corresponds to the reaction time (h), respectively. And then, the conversion rate Y (%) of monomers in polymerization can be calculated according to the following equation:
Cu2+ molar ratio and y corresponds to the reaction time (h), respectively. And then, the conversion rate Y (%) of monomers in polymerization can be calculated according to the following equation:
 | (1) |
Characterization
Fourier transform infrared spectroscopy (FTIR) of products was collected via Nicolet IS10 infrared spectrometer in the range of 4000–1000 cm−1 with resolution of 4 cm−1. Raman (LABRAM-HR 800, 514 nm He–Ne laser excitation) were applied to study the molecular structure of PmPD. The Atomic Absorption Spectrometry (WFX-200 AAS) was applied to test the Cu content in the filtrate. Before detection, the PmPD particles or solution was digested by a mixed solution of HCl and HNO3.25 JSM-6360 scanning electron microscopy (SEM) and TECNAI G2 transmission electron microscopy (TEM) were used to get the morphologies of the PmPD particles, with accelerating voltages of 20 kV and 120 kV, respectively. The X-ray diffraction (XRD) pattern was collected on a D/Max 2500 VB+X X-ray diffractometer using Cu (40 kV, 300 mA) radiation. X-ray photoelectron spectroscopy (XPS) measurements were carried out on a Thermo Fisher Scientific K-Alpha 1063 using Al Kα X-ray as the excitation source. The Inductively Coupled Plasma Atomic Emission Spectrometry (ICP-AES, Intrepid II XSP Radial) was used to detect the arsenic in the solution after adsorption. TOC was measured by TOC-VCPH.The solubility of products was investigated semiquantitatively as follows: 10 mg of PmPD particles was added into 10 mL of the solvent and the solution was shaken for 24 h at room temperature. After filtration, the filtrate was collected for calculation.26
Adsorption experiment
In the batch experiments, 20 mg of PmPD particles was added into 40 mL of As(V) aqueous solution with pH 5 ± 0.2 at 35 °C and shaken for a specific time (0–12 h). The solution pH was adjusted with HCl and NaOH solution. After filtration, the arsenic concentration in the filtrate was measured by ICP-AES.The adsorption isotherm was obtained by varying the initial arsenic concentrations and stirring for 12 h (concentration range: 0–80 mg L−1 for As(V)). The equilibrium adsorption capacity (qe) (mg g−1) for arsenic was calculated according to the following eqn (2). The Langmuir (3) and Freundlich (4) models were used to study the isotherm adsorption behavior of the PmPD particles.
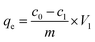 | (2) |
 | (3) |
 | (4) |
The adsorption kinetics was investigated with the initial As(V) concentration of 3.7 mg L−1 at pH of 5.0 and adsorbents dose of 0.5 g L−1. The solution was allowed to shake with the adsorbent at 35 °C for a special time (10–720 min).
The pseudo-first (5) and -second (6) order rate equations were used to test the kinetic process of adsorption:
 | (5) |
 | (6) |
The initial pH of the solution was controlled by HCl or NaOH from 3 to 9 for research the effect of pH on adsorption. And Cl−, SO42−, NO3−, PO43− were investigated as the competing anions and the molar proportion of competing anions to AsO43− was controlled from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 4
1 to 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.
Results and discussion
Efficiency of Cu-catalyzed air oxidation
The polymerization of mPDs in Cu-catalyzed air oxidation system was investigated. As shown in Fig. 1, Cu addition has a significant effect on the polymerization efficiency. Without Cu addition, no obvious solid particles could be filtered out after the 24 h polymerization finished, except that the bulk solution became brown. This is quite consistent with the previous reports,21,22 indicating that the oxidation ability of pure air oxidation system cannot effectively afford the polymerization of mPD. However, when Cu amount increased to nmPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nCu = 1
nCu = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1, the monomers conversion rates rapidly rise to 49%. Especially, when Cu amount further increased to nmPD
0.1, the monomers conversion rates rapidly rise to 49%. Especially, when Cu amount further increased to nmPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nCu = 1
nCu = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25 and more, the conversion rates continued to grow rapidly and reached close to 100%, this is far more than previous reports stated up to our knowledge (e.g. 93%).24 Especially, the obtained filtrates (before distilled water rinsed) appeared to be transparent, clean without any obvious suspended solids. The TOC remaining in the filtrates were below <10 mg L−1 (in ESI-3†), which is compliant with the discharge standard of the United States Environmental Protection Agency.27
0.25 and more, the conversion rates continued to grow rapidly and reached close to 100%, this is far more than previous reports stated up to our knowledge (e.g. 93%).24 Especially, the obtained filtrates (before distilled water rinsed) appeared to be transparent, clean without any obvious suspended solids. The TOC remaining in the filtrates were below <10 mg L−1 (in ESI-3†), which is compliant with the discharge standard of the United States Environmental Protection Agency.27
Of course, large amounts of Cu (65–878 mg) were still remained in the filtrates after polymerization (ESI-3†). To test the recyclability of these filtrates including residual Cu (TOC < 10 mg L−1, which is within the EPA limits), a comparable experiment was designed by directly adding certain amount of Cu and mPD into the said filtrates to make up nmPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nCu = 1
nCu = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5. The other procedures were the same as the above-mentioned method. Actually, the monomers conversion rate can also reach to 98.97%. That strongly demonstrated that the filtrates can certainly be re-used for the PmPD polymerization (ESI-2†) without any other treatments. Effective utilization of the obtained filtrates can greatly improve process cost and substantially prevent secondary pollutions to the maximum during the PmPD synthesis.
0.5. The other procedures were the same as the above-mentioned method. Actually, the monomers conversion rate can also reach to 98.97%. That strongly demonstrated that the filtrates can certainly be re-used for the PmPD polymerization (ESI-2†) without any other treatments. Effective utilization of the obtained filtrates can greatly improve process cost and substantially prevent secondary pollutions to the maximum during the PmPD synthesis.
To explore the polymerization process, open-circuit potential (OCP) and pH of the reaction system were monitored in situ, as shown in Fig. 2. As soon as the Cu2+ solution added into monomer solution, the polymerization OCP suddenly rose to more than 150 mV which was ascribed to the oxidizability and coordination of Cu2+.28–30 The Cu2+ could readily react with the monomers to form the complexes and the Cu2+ was reduced to the corresponding Cu+, which has been proven by our previous study.17,24 It can explain the OCP dropped sharply after finish adding the Cu2+ solution. As reflected by the color, the polymerization system rapidly changed from clean transparent to dark brown with lots of solids suspended. From Fig. 2(a), the higher solution potentials apparently can be obtained with the increased Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPD ratios after air introducing. Moreover, the increased Cu amounts also decreased the final solution pH (Fig. 2(b)) which was resulted from H+ release from mPD during the oxidation. It clearly demonstrates that the air oxidation was activated significantly through employing Cu2+. Especially, the solution potential with air introducing gradually declined to a relatively constant value. That is to say, the oxidation reaction remains steady in the solution. However, the exact mechanism is still unclear, yet it is apparently a consequence of catalytic air oxidation via Cu coordination with monomers. During the polymerization, the reduction of Cu2+ to Cu+ triggered the oxidation of monomer and polymerization, while the O2 in air could easily oxidize the Cu+ to Cu2+ again.22 This reversible process promised a dynamic balance for the oxidation.
mPD ratios after air introducing. Moreover, the increased Cu amounts also decreased the final solution pH (Fig. 2(b)) which was resulted from H+ release from mPD during the oxidation. It clearly demonstrates that the air oxidation was activated significantly through employing Cu2+. Especially, the solution potential with air introducing gradually declined to a relatively constant value. That is to say, the oxidation reaction remains steady in the solution. However, the exact mechanism is still unclear, yet it is apparently a consequence of catalytic air oxidation via Cu coordination with monomers. During the polymerization, the reduction of Cu2+ to Cu+ triggered the oxidation of monomer and polymerization, while the O2 in air could easily oxidize the Cu+ to Cu2+ again.22 This reversible process promised a dynamic balance for the oxidation.
Taken together, the presented Cu-catalyzed air oxidation provides a significant strategy for the sustainable and high efficient synthesis of PmPD particles.
Morphology of PmPD particles
The morphology of PmPD particles was measured by the SEM and TEM (Fig. 3 and ESI-4†). Obviously, amount of Cu addition have a significant influence on the micro-morphology of PmPD particles. As shown in Fig. 3(g) and (h), the product synthesized in nmPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nCu = 1
nCu = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.125 just consisted of abundant irregular solid particles. However, great variation emerged by increasing Cu content. When mPD/Cu ratio became 1
0.125 just consisted of abundant irregular solid particles. However, great variation emerged by increasing Cu content. When mPD/Cu ratio became 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, a lot of distinguishable solid microspheres appeared actually in the product, though their surfaces are very rough (Fig. 3(c) and (d)). It is noted that some hollow nanostructures could be identified apparently in Fig. 3(d). Especially, in a further experiment, the obtained PmPD-Cu1:1 particles were mainly composed of nanosized smooth spheres with the diameters of 300–500 nm (Fig. 3(a) and (b)). More importantly, the microspheres are hollow with a wall thickness of about 60–80 nm, as confirmed by the TEM image. Up to our knowledge, that is a new sustainable alternate to constitute hollow structure of PmPD particles.
0.5, a lot of distinguishable solid microspheres appeared actually in the product, though their surfaces are very rough (Fig. 3(c) and (d)). It is noted that some hollow nanostructures could be identified apparently in Fig. 3(d). Especially, in a further experiment, the obtained PmPD-Cu1:1 particles were mainly composed of nanosized smooth spheres with the diameters of 300–500 nm (Fig. 3(a) and (b)). More importantly, the microspheres are hollow with a wall thickness of about 60–80 nm, as confirmed by the TEM image. Up to our knowledge, that is a new sustainable alternate to constitute hollow structure of PmPD particles.
 | ||
| Fig. 3 SEM and TEM images: (a and b) PmPD-Cu1:1; (c and d) PmPD-Cu1:0.5; (e and f) PmPD-Cu1:0.25 and (g and h) PmPD-Cu1:0.125 (reaction time was 24 hours). | ||
To give insight to these morphologies variations, some samples were separated from the reaction solution with mPD/Cu ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 before air introduction and after reaction for 6 h, and then characterized using TEM. On account of the reaction of the Cu and monomer was very strong and fast. When Cu2+ solution was added, the color of solution suddenly turns to black from transparent and particulate matter could be found immediately. ESI-4a† was obtained by separate the solution in this time; primarily consisted of hollow nanorods with the length of 400–500 nm, the diameters of ∼100 nm and wall thickness of 20–30 nm. When the reaction time reached 6 h, the aggregates greatly changed into nanosized spheres with the increased diameters of 200–400 nm and wall thickness of 60–80 nm (in ESI-4b†), just similar to the final product (Fig. 3(a) and (b)). More importantly, a fission phenomenon among these aggregates was evidently observed (in ESI-4b†), which showed a tendency to split the nanorods into the nanospheres. It strongly demonstrated that the initial micromorphology of the synthesized particles definitely possessed the hollow rod-like nanomorphology, which finally was changed into the hollow nanospheres.
1 before air introduction and after reaction for 6 h, and then characterized using TEM. On account of the reaction of the Cu and monomer was very strong and fast. When Cu2+ solution was added, the color of solution suddenly turns to black from transparent and particulate matter could be found immediately. ESI-4a† was obtained by separate the solution in this time; primarily consisted of hollow nanorods with the length of 400–500 nm, the diameters of ∼100 nm and wall thickness of 20–30 nm. When the reaction time reached 6 h, the aggregates greatly changed into nanosized spheres with the increased diameters of 200–400 nm and wall thickness of 60–80 nm (in ESI-4b†), just similar to the final product (Fig. 3(a) and (b)). More importantly, a fission phenomenon among these aggregates was evidently observed (in ESI-4b†), which showed a tendency to split the nanorods into the nanospheres. It strongly demonstrated that the initial micromorphology of the synthesized particles definitely possessed the hollow rod-like nanomorphology, which finally was changed into the hollow nanospheres.
The self-formation mechanism of the hollow structures can be described as follows. When in contact with Cu2+, mPD instantly reacted to generate the positively charged complex, which would increase the molecules amphiphilicity to constitute micelles in water. As higher amount of Cu(II) salt was added into the system, the counter ion concentration also increased, which can compress the electrical double layer and reduce charge repulsion, allowing the micelles to come closer to each other.31–33 At the same time, the increased ionic strength can cause the molecules' polar groups undergo more ‘‘dehydration’’ to enhance their hydrophobic character, which will greatly reduce monomers exchange between the micelles and the bulk phase.34 Hence, the effects worked together to drive micellar molecules to be rearranged to form enlarged stable micellar cluster or bilayer aggregates, just like the obtained nanorods as ESI-4.†34 When air was introduced into the bulk solution, the oxidation chain-propagation will principally takes place around the surface of the aggregates to make them growing up.32 However, when the reaction reached to a certain extent, the dynamic equilibrium cannot be maintained between the grown aggregates and polymer molecules. The nanorods will spontaneously split into nanosized spheres by fission. Particularly, if the total Cu addition is not enough, low ionic strength will go against keeping hollow aggregates stable and finally formed irregular solid particles.
Structural characterization
The molecular structures of PmPD-Cux (x: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, 1
0.5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25, 1
0.25, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.125, 1
0.125, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1) were investigated by FTIR (Fig. 4). As seen in Fig. 4, the FTIR spectra of these four polymers were similar. The broad absorption centered between 3500 and 3000 cm−1 should be due to the stretching mode of –NH–.35–37 The peak at ∼1620 cm−1 was associated with phenazine and quinoid imine. And the peak at ∼1500 cm−1 was attributed to benzenoid amine structures.38–40 Meanwhile, the peak at ∼1250 cm−1 corresponded to the C–N stretching mode in the PmPD.41 Moreover, it was found that the relative content of the two peaks changed obviously with the variation of the Cu/monomer molar ratio. With the increase of Cu2+, the peak at ∼1620 cm−1 turned to stronger which indicated that content of phenazine and quinoid structure were increased. Generally speaking, Cu/monomer molar ratio was an effective factor on the variations of oxidation state.
0.1) were investigated by FTIR (Fig. 4). As seen in Fig. 4, the FTIR spectra of these four polymers were similar. The broad absorption centered between 3500 and 3000 cm−1 should be due to the stretching mode of –NH–.35–37 The peak at ∼1620 cm−1 was associated with phenazine and quinoid imine. And the peak at ∼1500 cm−1 was attributed to benzenoid amine structures.38–40 Meanwhile, the peak at ∼1250 cm−1 corresponded to the C–N stretching mode in the PmPD.41 Moreover, it was found that the relative content of the two peaks changed obviously with the variation of the Cu/monomer molar ratio. With the increase of Cu2+, the peak at ∼1620 cm−1 turned to stronger which indicated that content of phenazine and quinoid structure were increased. Generally speaking, Cu/monomer molar ratio was an effective factor on the variations of oxidation state.
Except for FTIR, Raman spectra were also used to analyses the structures of obtained PmPD particles in ESI-5.† The peak at ∼1573 cm−1 was attributed to quinoid. While the two peaks at ∼1330 and ∼1410 cm−1 were attributed to C–N+ and phenazine structure, respectively.42 It was found that the relative intensity of phenazine increased with the enhancement of Cu2+ amount. The increase of phenazine structure should be resulted from the oxidation of linear benzenoid structure.40 This indirectly verifies the increased oxidation state of PmPD with promoting the Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPD ratio, which was in line with the analysis of FTIR. On the other hand, the C–N+ was caused by the chelation of imine groups by Cu2+.
mPD ratio, which was in line with the analysis of FTIR. On the other hand, the C–N+ was caused by the chelation of imine groups by Cu2+.
Instead of FTIR and Raman, the empirical composition, functional groups on the surfaces, chemical state and electronic state of the elements within the PmPD particles were quantitatively determined by XPS technique. Fig. 5 was the typical survey spectra from 0 to 1400 eV of the samples. It clearly indicates that the PmPD particles with Cu loaded are basically made up of carbon, oxygen, nitrogen, copper and a small amount of chlorine. With the decrease of the initial dosage of Cu2+, the peak of Cu became weaker.
 | ||
| Fig. 5 Wide energy range surface spectra of PmPD-Cu1:1; PmPD-Cu1:0.5 PmPD-Cu1:0.25 and PmPD-Cu1:0.125. | ||
Here, as shown in Fig. 6(a)–(d), the XPS spectra of N element of various PmPD particles were investigated to study the relative molar contents of N-containing segments. The peak at 399.2 eV was associated with the neutral –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) in both quinoid imine and phenazine while the one at 400.2 eV was due to the –NH– in the benzenoid amine units. Beside these, there was a weak peak at 401.2 eV assigned to –N+
in both quinoid imine and phenazine while the one at 400.2 eV was due to the –NH– in the benzenoid amine units. Beside these, there was a weak peak at 401.2 eV assigned to –N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) .43–45 With the increase of Cu2+, the area of –N
.43–45 With the increase of Cu2+, the area of –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) and –N+
and –N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) increased, indicating the enhancement of oxidation state. This was in agreement with the analysis of FTIR and Raman. The presence of –N+
increased, indicating the enhancement of oxidation state. This was in agreement with the analysis of FTIR and Raman. The presence of –N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) prove that Cu2+ was chelated on the PmPD, which was in agreement with literatures.9,44 Based on XPS analysis, it was interesting to see that the oxidation state of PmPD increased with the increase of Cu/monomer molar ratio.
prove that Cu2+ was chelated on the PmPD, which was in agreement with literatures.9,44 Based on XPS analysis, it was interesting to see that the oxidation state of PmPD increased with the increase of Cu/monomer molar ratio.
 | ||
| Fig. 6 Deconvolution results of N 1s (a–d) and Cu 2p (e–h) XPS spectra of PmPD-Cu1:1 (a–e), PmPD-Cu1:0.5 (b–f), PmPD-Cu1:0.25 (c–g) and PmPD-Cu1:0.125. | ||
The XPS spectra of Cu element of PmPD particles were also shown in Fig. 6(e)–(h). Obviously, copper existed in two oxidation states on the PmPD particles, Cu2+ and Cu+.46–49 Cu+ contents tended to rise with the increase of Cu/mPD ratio. The variation of Cu+ contents proved that Cu2+ was involved in the mPD oxidation polymerization. It was the reduction of Cu2+ to Cu+ achieving the oxidation and the increased amount of Cu+ was possibly a direct reason for the increased oxidation state of PmPD. Therefore, it could be further correlated to the increase of conversion rate.
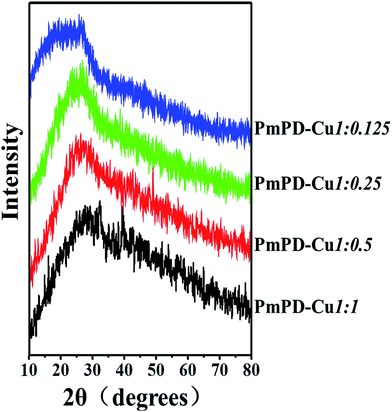
The supermolecular structure of PmPD particles was characterized by XRD (Fig. 7). As shown in Fig. 7, there was one broad peak in these five patterns located at ∼25 to 28°, which was a typical characteristic for amorphous structure.50 Generally speaking, an amorphous structure was conducive to the penetration and then adsorption of ions onto the macromolecules due to the loose and disordered piles of the polymeric chains in the amorphous structure.9,26,50 No peak observed in the range of 14–68°, suggesting that Cu, Cu2O and CuO did not exist51,52 which indicating that Cu2+ was chelated to PmPD consistent with XPS analysis.
In addition, the solubility of PmPD particles in H2O, DMF, EtOH, NMP and DMSO is listed in Table 1. As seen, the solubility of the five PmPD particles in H2O, DMF, EtOH, NMP and DMSO is very poor. The insoluble or slightly soluble property is suitable as adsorbent.
Arsenic adsorption ability of PmPD particles
 | ||
| Fig. 8 Effects of initial concentration of PmPD particles (T = 35 °C; adsorbent doses = 0.5 g L−1; pH = 5 ± 0.2). | ||
As shown in Fig. 8, when initial As(V) concentration is 70 mg L−1, PmPD-Cu0 exhibited a certain arsenic adsorbance as low as about 8 mg g−1. As the Cu/mPD ratios rose, the adsorbance of PmPD particles loaded with Cu significantly increased. The maximal adsorbance (about 27.4 mg g−1) occurred at Cu/mPD ratios 0.25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 4 times as much as that of PmPD-Cu0. Subsequently, further enhancing Cu/mPD ratio had no great impacts upon the As(V) adsorption of PmPD particles with just a slight decrease to about 23–24 mg g−1.
1, 4 times as much as that of PmPD-Cu0. Subsequently, further enhancing Cu/mPD ratio had no great impacts upon the As(V) adsorption of PmPD particles with just a slight decrease to about 23–24 mg g−1.
It is indicative that Cu played a crucial role in the arsenic removal of PmPD particles. Consequently, PmPD-Cu1:0.25 is more preferable adsorbent for As(V) removal.
To give a deeper analysis on the isotherm adsorption, math models, e.g., Langmuir and Freundlich were used to fit the data above. Results are given in ESI-6.† It summarizes the correlation coefficients (R2) of Langmuir and Freundlich isotherms of PmPD-Cu particles. The Freundlich can better describe the adsorption since the correlation efficiency of Freundlich is all much higher than that of Langmuir. This suggests that PmPD particles adsorption of arsenic is a multicomponent adsorption isotherm which is derived based on the assumption that an exponential distribution of adsorption energies exists for each component.53 The maximal capacity reached 27.4 mg g−1 for PmPD-Cu1:0.25. To demonstrate the advantage of the PmPD with Cu loaded, a comparison between them and some similar kind of reported adsorbents is provided in Table 2.54–57 It is obvious that the Cu loaded PmPD particles have a superior As removal performance in aqueous solution.
| Adsorbent sample | pH | Removal capacity for As(V) (mg g−1) | Reference |
|---|---|---|---|
| PmPD-Cu1:1 | 5 | 24.51 | This work |
| PmPD-Cu1:0.5 | 5 | 24.60 | This work |
| PmPD-Cu1:0.25 | 5 | 28.13 | This work |
| PmPD-Cu1:0.125 | 5 | 16.63 | This work |
| PmPD-Cu1:0.1 | 5 | 13.12 | This work |
| PmPD-Cu0 | 5 | 11.24 | This work |
| Fe3O4 particles | 5 | 7.23 | 54 |
| Ni/NixB nanoparticles coated resin | 6 | 17.8 | 56 |
| CTS-g-PA | 7.2 | 6.56 | 55 |
| Acidithiobacillus ferrooxidans BY-3 | 4.0 | 0.333 | 57 |
 | ||
| Fig. 9 Effects of time on the adsorption performance of PmPD-Cu1:0.25 particles (T = 35 °C; adsorbent doses = 0.5 g L−1; pH = 5 ± 0.2). | ||
For a further analysis, the pseudo-first-order and -second-order adsorption models were used to fit the above data and results are listed in ESI-7.† It summarizes the kinetic parameters and the R2. As can be seen, the pseudo-second-order kinetic model (>0.99) is better for describing the adsorption process, because of its higher correlation efficiency than the pseudo-first-order kinetic model (<0.97). The initial adsorption rate of arsenic onto the PmPD particles is in the range from 0.74 to 2.38 mg g−1 min−1. Noticeably, the establishment of the pseudo-second-order model is based on the fact that the interaction force between adsorbent and adsorbate is chemisorption process.9 That is to say, arsenic removal of PmPD particles is probably a chemical adsorption process.
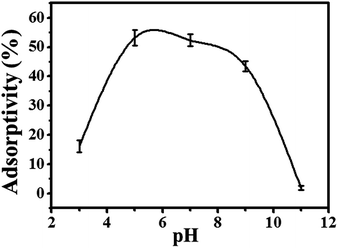 | ||
| Fig. 10 Effect of pH on the adsorption performance of PmPD-Cu1:0.25 particles (T = 35 °C; adsorbent doses = 20 mg L−1). | ||
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– and
N– and ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+– increased correspondingly.
N+– increased correspondingly.As is known to all, –NH– of conjugated polymers can be oxidized readily and then hydrogen left, which was finally transformed to ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–.44 That is to say, 0.92% of –NH– was oxidized by As(V) form to
N–.44 That is to say, 0.92% of –NH– was oxidized by As(V) form to ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–. On the other hand, the electron density of
N–. On the other hand, the electron density of ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– is much higher than that of –NH– due to its different molecular configuration,59,60 which allows it to interact with As(III) cations through coordination. The increased amount of
N– is much higher than that of –NH– due to its different molecular configuration,59,60 which allows it to interact with As(III) cations through coordination. The increased amount of ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+– after adsorption also strongly verifies this chelation between As(III) and
N+– after adsorption also strongly verifies this chelation between As(III) and ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–. It should be noted that part of the
N–. It should be noted that part of the ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– in PmPD before adsorption has been already chelated by Cu ions to generate
N– in PmPD before adsorption has been already chelated by Cu ions to generate ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+– which can interact with negative ionic As(V) through static manner. Based on the discussion of XPS, the corresponding interaction was illustrated in Scheme 1.44,61,62
N+– which can interact with negative ionic As(V) through static manner. Based on the discussion of XPS, the corresponding interaction was illustrated in Scheme 1.44,61,62
As soon as the addition of PmPD-Cu nanoparticles into the As(V) solution, abundant As(V) was adsorbed by ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+– components via electrostatic attraction. Meanwhile, the redox reaction between part of As(V) and nearby imine group (–NH–) occurred which produced the
N+– components via electrostatic attraction. Meanwhile, the redox reaction between part of As(V) and nearby imine group (–NH–) occurred which produced the ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– and As(III). Therein, As(III) can readily tend to interacts with
N– and As(III). Therein, As(III) can readily tend to interacts with ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– through coordination.
N– through coordination.
Conclusions
In this study, a sustainable synthesis approach of Cu-loaded PmPD particles with hollow morphologies was successfully proposed based on Cu-catalysed air oxidation. The maximal conversion rate is nearly 100%, much higher than other previous reports. The TOC in filtrate is less than 10 mg L−1 and can be directly re-used for air oxidation to eliminate secondary pollution. This work clearly indicates the importance of Cu in the air oxidation to prepare PmPD particles by facilitating the electron transfer from ligand to O2. In addition, the increase of Cu/mPD ratio tends to definitely constitute hollow morphology of PmPD particles. The explanation can be attributed to dehydration effect, which is beneficial for forming more stable and enlarged amphiphilic aggregates and finally form the hollow nanosized particles. Moreover, the Cu loaded PmPD particles show ability toward As(V) removal in aqueous solution with a maximal adsorbance of 27.4 mg g−1. The adsorption behaviour can be well described by Freundlich and pseudo-second-order model. Altogether, by using air as a costless green oxidant, the presented idea provides a significant green strategy for high efficiency and mass production of PmPD particles. The obtained Cu-loaded PmPD particles is a promising adsorbent for As removal in environmental protection and is potentially capable of being used in many fields, as a new composite materials, such as biosensors, catalysis and anticorrosion, etc.Conflict of interest
The authors declare no competing financial interest.Acknowledgements
This work was supported by National Natural Science Foundation of China (51304251, 51374237); Special Program on Environmental Protection for Public Welfare (201509050); Shanghai Tongji Gao Tingyao Environmental Science & Technology Development Foundation and Young Scholarship Award for Doctoral Candidate Issued by Ministry of Education (1343-76140000018).Notes and references
- Q. F. Lü, M. R. Huang and X. G. Li, Chem.–Eur. J., 2007, 13, 6009–6018 CrossRef PubMed.
- M. R. Huang, Y. B. Ding and X. G. Li, Analyst, 2013, 138, 3820–3829 RSC.
- M. R. Huang, X. W. Rao, X. G. Li and Y. B. Ding, Talanta, 2011, 85, 1575–1584 CrossRef CAS PubMed.
- J. Huang, S. Li, M. Ge, L. Wang, T. Xing, X. Liu, G. Chen, S. S. Al-Theyab, K.-Q. Zhang, T. Chen and Y. Lai, J. Mater. Chem. A, 2015, 3, 2825–2832 CAS.
- M. R. Huang, H. J. Lu and X. G. Li, J. Colloid Interface Sci., 2007, 313, 72–79 CrossRef CAS PubMed.
- F. Lü, X. Feng, L. Liu and S. Wang, Chin. Sci. Bull., 2013, 58, 4039–4044 CrossRef.
- X. G. Li, W. Duan, M. R. Huang and L. N. J. Rodriguez, React. Funct. Polym., 2005, 62, 261–270 CrossRef CAS PubMed.
- X. G. Li, W. Duan, M. R. Huang, Y. L. Yang, D. Y. Zhao and Q. Z. Dong, Polymer, 2003, 44, 5579–5595 CrossRef CAS.
- X. G. Li, X. L. Ma, J. Sun and M. R. Huang, Langmuir, 2009, 25, 1675–1684 CrossRef CAS PubMed.
- J. J. Wang, J. Jiang, B. Hu and S. H. Yu, Adv. Funct. Mater., 2008, 18, 1105–1111 CrossRef CAS.
- M. R. Huang, H. J. Lu, W. D. Song and X. G. Li, Soft Mater., 2010, 8, 149–163 CrossRef CAS.
- L. Zhang, T. Wang, H. Wang, Y. Meng, W. Yu and L. Chai, Chem. Commun., 2013, 49, 9974–9976 RSC.
- W. Yu, L. Chai, L. Zhang and H. Wang, Trans. Nonferrous Met. Soc. China, 2013, 23, 3490–3498 CrossRef CAS.
- L. Zhou, Y. Wang, Z. Liu and Q. Huang, J. Hazard. Mater., 2009, 161, 995–1002 CrossRef CAS PubMed.
- Z. Su, L. Zhang, L. Chai, H. Wang, W. Yu, T. Wang and J. Yang, New J. Chem., 2014, 38, 3984–3991 RSC.
- X. G. Li, R. Liu and M. R. Huang, Chem. Mater., 2005, 17, 5411–5419 CrossRef CAS.
- L. Zhang, H. Wang, W. Yu, Z. Su, L. Chai, J. Li and Y. Shi, J. Mater. Chem., 2012, 22, 18244 RSC.
- M. R. Huang, H. J. Lu and X. G. Li, J. Mater. Chem., 2012, 22, 17685–17699 RSC.
- Z. Chen, C. Della Pina, E. Falletta and M. Rossi, J. Catal., 2009, 267, 93–96 CrossRef CAS PubMed.
- L. Zhang, L. Chai, H. Wang and Z. Yang, Mater. Lett., 2010, 64, 1193–1196 CrossRef CAS PubMed.
- Y. Wang, X. Jing and J. Kong, Synth. Met., 2007, 157, 269–275 CrossRef CAS PubMed.
- N. Bicak and B. Karagoz, J. Polym. Sci., 2006, 44, 6025–6031 CrossRef CAS.
- Z. Sun, Y. Geng, J. Li, X. Jing and F. Wang, Synth. Met., 1997, 84, 99–100 CrossRef CAS.
- W. Yu, L. Zhang, Y. Meng, S. Dai, Z. Su, L. Chai and H. Wang, Synth. Met., 2013, 176, 78–85 CrossRef CAS PubMed.
- S. Bourdo, B. Berry and T. Viswanathan, J. Appl. Polym. Sci., 2005, 98, 29–33 CrossRef CAS.
- X. G. Li, W. Duan, M. R. Huang and Y. Yang, J. Polym. Sci., 2001, 39, 3989–4000 CrossRef CAS.
- D. J. Pezolt, S. J. Collick, H. A. Johnson and L. A. Robbins, Environ. Prog., 1997, 16, 16–19 CrossRef CAS.
- M. L. Chen and Z. H. Zhou, Polyhedron, 2014, 81, 142–146 CrossRef CAS PubMed.
- R. Procaccini, W. Schreiner, M. Vázquez and S. Ceré, Appl. Surf. Sci., 2013, 268, 171–178 CrossRef CAS PubMed.
- L. Chai, T. Wang, L. Zhang, H. Wang, W. Yang, S. Dai, Y. Meng and X. Li, Carbon, 2014, 81, 748–757 CrossRef PubMed.
- H. Wang, L. Chai, A. Hu, C. Lü and B. Li, Polymer, 2009, 50, 2976–2980 CrossRef CAS PubMed.
- J. K. Harris, G. D. Rose and M. L. Bruening, Langmuir, 2002, 18, 5337–5342 CrossRef CAS.
- H. Li, Y. Lai, J. Huang, Y. Tang, L. Yang, Z. Chen, K. Zhang, X. Wang and L. P. Tan, J. Mater. Chem. B, 2015, 3, 342–347 RSC.
- J. Dey, S. Kumar, S. Nath, R. Ganguly, V. Aswal and K. Ismail, J. Colloid Interface Sci., 2014, 415, 95–102 CrossRef CAS PubMed.
- P. Sang, Y. Wang, L. Zhang, L. Chai and H. Wang, Trans. Nonferrous Met. Soc. China, 2013, 23, 243–252 CrossRef CAS.
- X. G. Li, L. X. Wang, Y. Jin, Z. L. Zhu and Y. L. Yang, J. Appl. Polym. Sci., 2001, 82, 510–518 CrossRef CAS.
- L. Chai, L. Zhang, H. Wang, W. Yu and P. Sang, Mater. Lett., 2010, 64, 2302–2305 CrossRef CAS PubMed.
- X. G. Li, M. R. Huang, W. Duan and Y. L. Yang, Chem. Rev., 2002, 102, 2925–3030 CrossRef CAS PubMed.
- M. Liu, M. Ye, Q. Yang, Y. Zhang, Q. Xie and S. Yao, Electrochim. Acta, 2006, 52, 342–352 CrossRef CAS PubMed.
- G. Ćirić-Marjanović, B. Marjanović, P. Bober, Z. Rozlívková, J. Stejskal, M. Trchová and J. Prokeš, J. Polym. Sci., Part A: Polym. Chem., 2011, 49, 3387–3403 CrossRef.
- M. Huang, X. Li and Y. Yang, Polym. Degrad. Stab., 2000, 71, 31–38 CrossRef.
- G. Ćirić-Marjanovicć, M. Trchová and J. Stejskal, J. Raman Spectrosc., 2008, 39, 1375–1387 CrossRef.
- W. T. Yu, L. Y. Zhang, H. Y. Wang and L. Y. Chai, J. Hazard. Mater., 2013, 260, 789–795 CrossRef CAS PubMed.
- L. Zhang, L. Chai, J. Liu, H. Wang, W. Yu and P. Sang, Langmuir, 2011, 27, 13729–13738 CrossRef CAS PubMed.
- I. Losito, C. Malitesta, I. De Bari and C.-D. Calvano, Thin Solid Films, 2005, 473, 104–113 CrossRef CAS PubMed.
- J. M. Lázaro Martínez, E. Rodríguez-Castellón, R. M. T. Sánchez, L. R. Denaday, G. Y. Buldain and V. Campo Dall'Orto, J. Mol. Catal. A: Chem., 2011, 339, 43–51 CrossRef PubMed.
- R. L. Frost, Y. Xi and B. J. Wood, Thermochim. Acta, 2012, 545, 157–162 CrossRef CAS PubMed.
- B. Pelissier, A. Beaurain, H. Fontaine, A. Danel and O. Joubert, Microelectron. Eng., 2009, 86, 1013–1016 CrossRef CAS PubMed.
- R. S. Vieira, M. L. M. Oliveira, E. Guibal, E. Rodríguez Castellón and M. M. Beppu, Colloids Surf., A, 2011, 374, 108–114 CrossRef CAS PubMed.
- M. R. Huang, Q. Y. Peng and X. G. Li, Chem.–Eur. J., 2006, 12, 4341–4350 CrossRef CAS PubMed.
- L. Zhirong, A. Uddin and S. Zhanxue, Spectrochim. Acta, Part A, 2011, 79, 1013–1016 CrossRef PubMed.
- M. H. Habibi and B. Karimi, J. Ind. Eng. Chem., 2014, 20, 1566–1570 CrossRef CAS PubMed.
- C. Sheindorf, M. Rebhun and M. Sheintuch, J. Colloid Interface Sci., 1981, 79, 136–142 CrossRef CAS.
- T. Wang, L. Zhang, H. Wang, W. Yang, Y. Fu, W. Zhou, W. Yu, K. Xiang, Z. Su, S. Dai and L. Chai, ACS Appl. Mater. Interfaces, 2013, 5, 12449–12459 CAS.
- S. Saha and P. Sarkar, J. Hazard. Mater., 2012, 227–228, 68–78 CrossRef CAS PubMed.
- T. D. Çiftçi and E. Henden, Powder Technol., 2015, 269, 470–480 CrossRef PubMed.
- L. Yan, H. Yin, S. Zhang, F. Leng, W. Nan and H. Li, J. Hazard. Mater., 2010, 178, 209–217 CrossRef CAS PubMed.
- M. Ayad, W. Amer and J. Stejskal, Thin Solid Films, 2009, 517, 5969–5973 CrossRef CAS PubMed.
- J. Han, J. Dai and R. Guo, J. Colloid Interface Sci., 2011, 356, 749–756 CrossRef CAS PubMed.
- J. Stejskal, M. Trchová, L. Brožová and J. Prokeš, Chem. Pap., 2009, 63, 77–83 CAS.
- C. Izumi, H. F. Brito, A. M. D. Ferreira, V. R. Constantino and M. L. Temperini, Synth. Met., 2009, 159, 377–384 CrossRef CAS PubMed.
- M. R. Huang, S. J. Huang and X. G. Li, J. Phys. Chem. C, 2011, 115, 5301–5315 CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: ESI-1 equipment; ESI-2 synthesis sketch map; ESI-3 conversion rate of monomer, total TOC and Cu in filtrate of PmPD particles; ESI-4 SEM and TEM images of PmPD-Cu1:1 in different time; ESI-5 Raman spectra of PmPD particles; ESI-6 parameters of Langmuir and Freundlich models simulated by no-liner fit for the adsorption of arsenic on PmPD particles; ESI-7 kinetic parameters of pseudo-first and models for the adsorption of arsenic on PmPD-Cu1:0.25; ESI-8 effect of coexisting ions on AsO43− adsorption with PmPD-Cu1:0.25. See DOI: 10.1039/c4ra16499g |
| ‡ Shuo Dai, Bing Peng and Liyuan Zhang contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2015 |