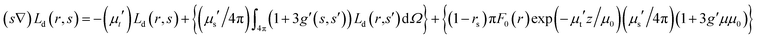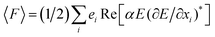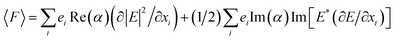DOI:
10.1039/C4RA15867A
(Paper)
RSC Adv., 2015,
5, 19002-19013
Design of a portable nanosensor for easy breast tomography
Received
5th December 2014
, Accepted 19th January 2015
First published on 28th January 2015
Abstract
In this study, a portable nanosensor for the early and easy detection of carcinoma tumors was designed and simulated. The nanosensor consists of deposited nanoparticles, which have a regular distance from each other. It has high sensitivity to the alteration of electromagnetic fields, which are scattered from different tissues (normal and tumor). Normal tissue permittivity differs from the permittivity of tumors; hence, their interaction with electromagnetic waves will lead to different results in the case of an electrical field and its gradient profile. It means that non-uniformity will occur and this is a meaningful signal for detection. Nevertheless, it is obvious that due to the tissue absorption coefficient, scattered photons will be very negligible and a small fraction of the photons will reach the detector. Hence, in this paper, a sensor based on nanoparticles is proposed, which has enough sensitivity to pick up scattered photons, amplify, and detect them. It should be noted that the roles of the nanosensor between the body surface and detector are signal amplification and sampling via the nanoparticle plasmonic effect. This means that the designed nanoparticles sample the scattered waves and amplify them in the near-field. Finally, our design and simulation results show that the digitized signals could be easily and clearly detected by force or temperature detectors. Thus, easy breast tomography will be carried out with no need for clinics and their equipment.
Introduction
An obvious regretful fact is that cancer is the second leading cause of death in the world; researchers have done their best to find purely safe treatments for cancer. The development of nanobiotechnology and nanomedicine is considered to be among the most promising advances in cancer treatment. It refers to the application of small materials and devices whose sizes are measured in the billionths of a meter in the detection, diagnosis, and treatment of cancer and other diseases by modern approaches.1–4 When compared with other available common therapies,5–7 such as chemotherapy, radiotherapy and surgery, nanomedicine has proven to be especially promising in fighting cancer. Drug delivery is the most important case with high accuracy compared to the common methods used in the treatment of cancer. Thus, it must be regulated with high precision targeting and imaging. It has been proven that the main disadvantages of common methods such as chemotherapy are related to the use of non-targeted drugs for killing cancerous tumors. However, it should be maintained that in the field of nanotechnology and nanomedicine, the accurate detection of tumors or cancer cells has been carried out by means of effective methods such as active targeting. One of the important features of active targeting is that several biological agents can be attached to the surface of nanoparticles (NPs) to construct multi-function sensors and simultaneously do several tasks1–3,8–11. It has been reported that nanomedicine and nanobiotechnology have enormous advantages but they are not regarded as user-friendly and easy methods. Moreover, they must be used in clinical trials. Thus, nowadays, researchers aspire to find user-friendly methods, which have the advantage of early detection. In this paper, a method has been proposed which can perform tumor tomography (especially for breast cancer) with and without active targeting agents. This method works based on the interaction of light (near- and far-infrared frequencies) with different tissues (normal tissue and tumor). In this method, scattered photons are detected by a sensor with high sensitivity. It is obvious that absorption in real tissue is too high and the photons, which reach the detector, are very negligible. Moreover, the wavelengths of 833, 1550, and 6199 nm were used in which the absorption rate in tissue component matters such as H2O, oxy- and deoxy hemoglobin is low; however, the scattered photons are small. Hence, for solving this problem, a sensor with high sensitivity, which has no extra noise, should be designed to detect photons. In this study, such a sensor was designed based on core–shell NPs, which was intended to detect and manipulate the small fraction of scattered photons. Simulation results show that the order of photons in critical conditions is about of 0.1 W cm−2. Therefore, to sort out this critical issue, photon amplification by NPs in the near-field state was used. That is to say, when light interacts with NPs, all the carriers on the NPs surface harmoniously resonate. This case which contributes to the NPs plasmonic resonance leads to a high intensity and non-uniform electric field in the closeness of the NPs.12–14 In other words, NPs resonance in the plasmonic state can amplify the signals, which arrive and detect them in the near-field state. Moreover, the plasmonic–plasmonic interaction between NPs can intensify the gradient of the electrical field and hence, the detected photons will be improved. In the following, the mathematics and theoretical section of our work are presented. Furthermore, the theoretical and simulation operations were conducted by MATLAB and COMSOL, respectively.
Mathematics and theory
Penetration of light in tissue
Light propagation in tissue is an important question in different biological applications, such as imaging, sensing, and targeting. Numerous methods, including analytical, numerical and statistical ones have been developed for predicting light propagation in tissue.17 Optical parameters are obtained by the use of measured parameters, such as reflection, transmission, and absorption and converting them into parameters which characterize light propagation in tissue. It has been reported that most of the advances regarding laser light transfer in tissues are based on the transport theory.17,18 It should be noticed that the superiority of this theory in tissue optics compared to many analytic methods is attributed to the non-homogeneity of biological tissues. Transport coefficients can be derived from the collision of a plane wave with a particle; thus, some of the waves are absorbed, some are scattered and some of them are unperturbed and transmitted. It seems that the transport equation is one of the complete methods for defining tissue optical parameters and light propagation in tissue. The radiative transport equation is given by| |
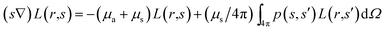 | (1) |
where L(r,s) refers to the radiance at position r in the direction of vector s and dΩ denotes differential solid angle. The parameters μa and μs indicate the medium absorption and scattering coefficients, respectively. The phase function p(s,s′) represents the fraction of scattered light from direction s′ into s. In this study, the delta-Eddington phase function is used as the phase function which is given by| |
Pdelta-Eddington(cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ) θ) ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) = = ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (1/4π){2fδ(1 − cos (1/4π){2fδ(1 − cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ) + (1 − f)(1 + 3g′ θ) + (1 − f)(1 + 3g′![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ)} θ)}
| (2) |
where cos(θ) = (s,s′) denotes the cosine angle between the incident and the scattered light. The first term indicates strong scattering in the forward direction and the second term contributes to diffuse scattering. Moreover, the parameters f and g′ are the fraction of light in the forward direction and the degree of asymmetry in the diffuse section, respectively.
Substituting eqn (2) into eqn (1) yields:
| |
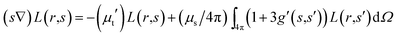 | (3) |
where
μt′ =
μa + (1 −
f)
μs is the transport coefficient and is less than
μt =
μa +
μs; this is due to the fact that the fraction of light is scattered to the
s direction from
s′. Moreover, the reduced scattering coefficient can be defined as
μs′ = (1 −
f)
μs.
The radiance is divided into collimated and diffuse components:
| |
L(r,s) ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) = =![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Lcol(r,s) Lcol(r,s) ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) + + ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ld(r,s) Ld(r,s)
| (4) |
The collimated radiance includes both the light scattered into the direction parallel to the incident beam and any un-scattered light which is represented as:
| |
Lcol(r,s)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) = = ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (1 − rs)πF0(r)exp(−μt′z/μ0)δ(μ − μ0) (1 − rs)πF0(r)exp(−μt′z/μ0)δ(μ − μ0)
| (5) |
In this equation, rs is the specular reflection coefficient given by the Fresnel reflection formula. F0 is the initial irradiance and μ is the angle between s and z and μ0 = sz. By substituting eqn (5) into eqn (4) and simplifying it, we have the following:
| |
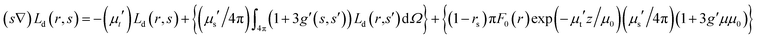 | (6) |
For further simplification, the diffuse radiance can be written as
| | |
Ld(r,s) = (1/4π)φd(r) + (3/4π)Fd(r)s
| (7) |
where
φd and
Fd represent the isotropic and anisotropic contribution to the diffuse radiance, respectively. Finally, by substituting
eqn (7) into
eqn (6) and further simplifying, the diffuse radiance is represented as the following:
| | |
(s∇)φd(r) + 3(s∇)(Fd(r)s) = −(μa)φd(r) − 3(μt′ − gμs′)(Fd(r)s) + {μs′(1 − rs)πF0(r)exp(−μt′z/μ0)(1 + 3g′μμ0)}
| (8) |
Eqn (8) contains isotropic and anisotropic diffuse forms. For solving this equation, one of them can be written based on the other, which leads to the following Helmholtz equation:
| | |
∇2φd(r) − 3μtr′μaφd(r) = −3{μs′(1 − rs)πF0(r)exp(−μt′z/μ0)(μtr′ + g′μt′)}
| (9) |
This equation is solved based on diverse boundary conditions at the interface between two media. Then, by solving this equation, the radiances at different points of the simulation area as well as the nanosensor surface position are determined. Indeed, the goal of this section is the propagation of the light with different wavelengths into tissue, which is calculated by considering of the medium absorption and scattering coefficients. Finally, in this study, the optical properties of real tissue were used from experimental results such as in vitro and in vivo data.17
Electric field gradient and optical force
When an electromagnetic wave interacts with NPs, the Δpphoton is transferred; then, based on momentum conservation, the particle undergoes an equal and opposite momentum change by −Δpphoton. This momentum change exerts a force on the particle, which is given by the change rate of the particle momentum. Two types of forces arise at the interface due to the light–matter interaction. First one is the scattering force (Fsca), which acts along the direction of light propagation and the second one is the gradient force (Fgrad), which pulls the particle towards the higher intensity region. However, it should be noted that the optical force is raised in a non-uniform electric field and it will disappear in a uniform field. Because tissue and tumor have different interactions with light, a non-uniform electric field is caused in the medium by the inhomogeneity in the medium. Nevertheless, it should be pointed out that nanosensor output non-uniformity is produced due to the plasmonic resonance of NPs, i.e., at the closeness of any deposited NPs, non-uniformity is created which is related to the interaction of the scattered light with NPs. Hence, due to the non-uniformity of NPs which is distributed in the nanosensor output, its measurable parameter such as optical force could be analyzed. For better investigation, the optical force is theoretically discussed in this section. It has been reported that the time-average force on a dipole in a harmonic electromagnetic field can be defined as15,16| | |
〈F〉 = (1/4)Re(α)∇|E|2 + (κ/2)Im(α)Re(E* × B) + (1/2)Im(α)Im[(E*∇)E]
| (10) |
where F, α, E, B, and κ denote optical force, polarizability, electrical field, magnetic field, and wave vector, respectively. In this equation, the first term on the right is the gradient force and the next terms are cooperatively titled as the scattering force. To prove eqn (10), researchers began with an expression for the negative gradient of the classical dipole–field interaction Hamiltonian:| |
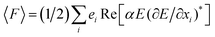 | (11) |
where i, ei refer to the Cartesian component and the related normal vector, respectively. By simplifying eqn (11), the following is obtained:| |
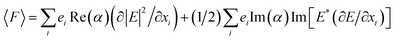 | (12) |
In this equation, the first term is related to the gradient force and the second one is related to the scattering force. Previous studies and investigations have indicated that as a result of manipulating core–shell NPs, the magnitude of the gradient force is far greater than that of the scattering force and the aforementioned case can be negligible.13 Hence, in this study, the gradient force is calculated and referred to as the optical force.
Nanoparticles local temperature
When an electromagnetic wave interacts with any NP, the optically stimulated NPs generate heat. In other words, NPs can effectively produce heat under light irradiance. The mechanism of heat generation by NPs is very simple; the electromagnetic field strongly derives mobile carriers into the NPs and the energy gained by the carriers turns into heat. Then, the heat diffuses away from the NPs and this leads to an increase in the local temperature around them. It should be noted that the heat released by NPs becomes strong in the case of plasmonic resonance in metal NPs. The occurrence of plasmonic resonance depends on NPs size, materials, shape, electromagnetic wavelength, and medium dielectric constant. In this paper, multi-shell NPs with different materials were used. Hence, by changing the core and shell sizes, the plasmonic resonance (magnitude and peak wavelength) will be altered. Moreover, in the core–shell structure, plasmonic–plasmonic interactions could arise. In other words, the plasmonic in the core and outer shell will interact with each other; this interaction can manipulate the electric and current density in the structure. Thus, the magnitude of energy gained by the mobile carriers is altered. Under this condition, the temperature distribution around the NPs is given by the heat transfer equation20,21| | |
ρ(r)c(r)∂T(r,t)/∂t = ∇κ(r)∇T(r,t) + Q(r,t)
| (13) |
where r, t, T(r,t), p(r), c(r), and κ(r) denote the coordinate, time, local temperature, mass density, specific heat, and thermal conductivity, respectively. Q(r,t) represents an energy source from wave dissipation in NPs and is obtained through the following equation| | |
Q(r,t) = 〈j(r,t)E(r,t)〉t
| (14) |
where j(r,t) and E(r,t) denote the current density and electric field in the system, respectively. For solving eqn (13), several numerical and analytical methods have been commonly used. It is notable that in this work, the time variant section of temperature is not important and can be neglected; hence, we solved the temperature equation in the steady-state condition. By substituting eqn (14) into eqn (13) and solving it in the steady-state condition, the local temperature around any NP is described by the equation20| | |
ΔT(r) = VNPsQ(r)/4πκ0r
| (15) |
where r, κ0, and VNPs refer to the NPs radius, the thermal conductivity of the surrounding medium, and NPs volume, respectively. Moreover, Q(r) represents an energy source from wave dissipation in NPs. It is severely raised due to the interaction of light with NPs at which the NPs resonate in the plasmonic state. In other words, by controlling the plasmonic key parameters, the heat generation by NPs will be changed. Based on the above-mentioned equations, it can be pointed out that the heat generation rate and temperature enhancement depend on the physical properties of a material. It has been indicated that the heat generation efficiency by Au NPs is about 10−3 under plasmonic resonance conditions.20 Nevertheless, it should be mentioned that this efficiency can be optimized by plasmonic hybridization in core–shell NPs. Moreover, the heat generation by NPs can be dramatically enhanced in the presence of several NPs. This contributes to the interaction mechanism between NPs, namely the accumulative effect and Coulomb interaction. The accumulative effect refers to the addition of heat fluxes produced by the NPs. However, in the case of the Coulomb interaction, the generated heat is affected by the interaction of neighboring NPs and the total amount of generated heat by the two interacting NPs will be different from the amount of heat produced by two single NPs; this is attributed to the partial screening of the electric field inside the NPs. Moreover, this effect may come from the plasmon resonance shift, which results from the NPs Columbic interaction. With respect to the recent use of the NPs Columbic interactions for enhancing heat generation, the plasmonic–plasmonic interactions between adjacent NPs were used in the present study to enhance the temperature signal. In the following, the nanosensor structure design and simulation results on light propagation in a real tissue and output signal sampling by NPs are presented.
Nanosensor structure
In this study, the designed sensor is based on the deposition of core–shell NPs in a defined distance from each other. The 3-D and 2-D illustrations of this sensor are depicted in the left and right sides of Fig. 1, respectively. This schematic shows that the nanosensor covers the whole breast and the electromagnetic or the laser source is placed on top of it. The size of the smallest element of the nanosensor (pixel) is about 10.2 um and it includes an N × M NPs matrix in which N and M are matrix incidences. It should be mentioned that in this work, NPs with a core and two shells such as Au/SiO2/Au were used. The step-by-step multi-shell NP formation process and moreover their optical characterization has been reported in.12 In this reference, it is seen that the experimental and theoretical characterizations of Au/SiO2/Au NPs are interestingly fitted to each other and easily manipulated by changing the NPs geometry. The gap between the core and outer shell is made of dielectric materials because NPs plasmonic hybridization, as an important parameter, is manipulated. Another important parameter in the design of the nanosensor was the medium materials in which the NPs were inserted; this is due to the fact that the medium material's thermal conductivity and its dielectric constant have an impact on nanosensor outputs. Aerogel silica or silica was used as the medium materials in this study. In most simulations, aerogel silica was used instead of silica due to its controllable thermal conductivity and dielectric constant. Moreover, the NPs outer shell size radius was approximately considered to be 150 nm. The inner shell and core radius were altered to improve the nanosensor output, which is related to the plasmonic hybridization in the core and outer shell. Furthermore, for the incidence of laser light on matter, the three wavelengths of 833 nm, 1550 nm, and 6199 nm were used. Finally, two distinct phenomena including optical force and thermal effect could be sensed by piezoelectric detectors.25,26 For practical fabrication of the portable nanosensor, which is schematically illustrated in Fig. 1, the nanoreplica molding process can be used. This process has been demonstrated as a low cost and suitable method for manufacturing periodic NPs based structures. It is notable that this method is performed using low force at room temperature to create nanometer-scale structures with high uniformity, which is very important in our work, over a large area using a patterned silicon wafer as a reusable molding template.27
 |
| | Fig. 1 3-D and 2-D illustration of the nanosensor, NPs arrangement in each pixels, Laser incidence in the z-coordinate, and schematic of breast coverage by the nanosensor. | |
Discussion
The high efficiency nanotechnology-based method has been recently introduced to detect carcinoma tumors. This method is based on active targeting in which functionalized NPs are injected to the body. Because of their attached elements such as proteins, enzymes, and antibodies, the functionalized NPs are considered to be smart agents. They can precisely find a target and accumulate near to its location. After targeting is accomplished by the smart NPs, the effective therapy will be fulfilled by the drug delivery. However, in general, it should be maintained that this approach is not effective because it cannot be easily used and it calls for clinical equipment. Nevertheless, in the present study, a new approach has been proposed, which is able to portably detect breast cancer tumor independently of any injected agents. In addition, the test can be accomplished without the need for anyone or clinics. This method is based on the interaction of light or an electromagnetic wave with breast tissue in which the detection of scattered photons is carried out by a very sensitive sensor. It has been proven that the dielectric constant and permittivity of the normal and abnormal cells are different from each other,22–24 i.e., the optical properties of carcinoma and normal cells are different and the amount of photon scattering and absorption are different for two distinct tissues (normal tissue and tumor). In other word, when a tumor initially starts to be constructed, the dense matter of the tissue with an abnormal state is replaced by normal tissue. In its construction mode, the electrical properties of the tissue, such as permittivity, conductivity, and so on is altered which is attributed to the new blood vascularization and proliferation rate in the tumor medium. Thus, the interaction of light flux with the normal and abnormal tissue results in different electrical field profiles and its gradient in the output, which is related to enormous non-uniformity. Thus, it is easy to detect the field non-uniformities by different practical methods. Nevertheless, an important drawback of this method is too much absorption of light in tissue, which limits the penetration of light into tissue. For solving this problem, two approaches were used in this study: I. Wavelength selection in which the minimum light is absorbed into tissue (for minimizing tissue loss, three different wavelengths 833, 1550, and 6199 nm were used at which the absorption of H2O and oxy- and deoxy-hemoglobin are small); II. Improving the nanosensor specification. The latter case was carried out based on NPs plasmonic resonance and plasmonic hybridization, which produces a high intensity field and field gradient near the NPs. In the following sections, a detailed and step-by-step description of the proposed method is given. Fig. 2 illustrates the proposed method. In this, the arrangement of the nanosensor, detector, core–shell NPs, pixels of the nanosensor and a pixel of the detector are schematically depicted. As shown in this schematic, when an electromagnetic wave with a definite wavelength (833, 1550, or 6199 nm) is radiated on the breast area, most incident photons will be absorbed in the tissues with respect to their absorption coefficient and a few photons will be scattered, which can be detected by the nanosensor. Moreover, piezoelectric and nanosensor typical data for the design are reported in this schematic. It should be noted that the simulated piezoelectric detector, similar to the reported case in,25 could sense a force about 1 μN. In the following of simulation, at first, the propagation of light into a tissue was simulated by solving the Helmholtz equation. The simulation results indicate that the photons detected by the nanosensor were very small and were not easily recorded where the detected ones were disturbed by background noise.
 |
| | Fig. 2 Illustrations of the framework; irradiance of light (w cm−2 sr) in the x-coordinate and measuring the optical force or temperature by detectors in the y- and z-coordinates and zoom out of nanosensor's and detector's pixel. | |
However, it should be considered that the photons scattered from distinct tissues were different, which is related to their optical properties. It means that when an electromagnetic wave interacts with different matter, which have distinct optical properties (scattering, absorption, and anisotropy), the scattered photons in the sensors from different tissues construct a tangible profile after photon amplification by the NPs. Thus, in this study, a nanosensor was designed to detect a small fraction of scattered photons, amplify them in the near-field and finally above all, sample the continuous output signals. The latter case is the most important result in the clear detection of the output, which contributes to the NPs plasmonic resonance. It should be noted that continuous and attenuated signal detection is very hard due to distortion by several noises. Indeed, the sampling rate is controlled by manipulating the NPs distance from each other and their plasmonic–plasmonic interactions, which surely influence signal detection. Finally, the digitized signals are scanned by detectors to construct a field and its gradient profile based on a variety of tissues. In other words, the nanosensor output signal is sampled by the NPs plasmonic resonance, which is easily controllable. Indeed, output signal sampling refers to the use of NPs plasmonic resonance in the near-field state. For this, the core and shell plasmonics interact with each other with different strengths, which depend on the distance between the core and outer shell. The plasmonic interaction inside the NPs has a strong effect on the optical force and local temperature around the NPs, which are used to detect the scattered photons. The optical force is related to the electrical field gradient and the NPs local temperature depends on the electrical field and current density. Moreover, in the design of the nanosensor, the Coulomb interaction of the NPs was used to improve the heat generated by the NPs. It should be pointed out that the Coulomb interaction between NPs might have an impact on the partial screening of the electric field inside the NPs. Thus, this case can have a dramatic impact on the NPs plasmonic resonance. The high sensitivity of a nanosensor severely depends on the NPs interactions, which are determined by the distance of the NPs from each other in the nanosensor. With regard to this issue, the designed nanosensor can detect the small variation of tissues as the optical properties change. In the following section, the simulation results in the case of detecting a tumor inside the tissue are illustrated and examined.
Results
In this section, the simulation results including light propagation in a typical tissue at different wavelengths and nanosensor outputs are discussed. Initially due to the significance of light propagation into a tissue, the light penetration into Murine albino tissue is investigated. Table 1 shows the tissue optical properties at different wavelengths. By the incidence of light with angle = 0 rad, minimum photon lost will occur, which is due to tissue surface light reflection with regard to Fresnel's law. Fig. 3a illustrates that as the wavelength increases, the penetration depth of light into tissue dramatically increases. The alteration of the diffusion flux into tissue regarding non-zero incidence angle is illustrated in Fig. 3b. This figure illustrates that the penetration of light is severely limited, as the incidence angle increases. Thus, by selecting a suitable wavelength and incident angle, the magnitude of photons, which reach the sensor's surface, will be controllable. Nevertheless, the absorbed photons in real tissue are very high. Therefore, the designed sensor should be able to detect a small fraction of light.
Table 1 Tissue: murine (albino) optical properties vs. wavelengths17,18
| |
488 nm |
800 nm |
1064 nm |
1320 nm |
| Absorption coefficient (μa) (cm−1) |
12.2 |
5.7 |
5.9 |
6.1 |
| Scattering coefficient (μs) (cm−1) |
173.5 |
97 |
60.9 |
44.2 |
| Anisotropy factor (g) |
0.93 |
0.94 |
0.92 |
0.91 |
 |
| | Fig. 3 Anisotropic diffusion flux (w m−2) vs. tissue depth (cm); (a) irradiance angle = 0 rad, (b) irradiance angle = pi/4 rad. | |
In the following section, the important output signals, which are finally related to the optical force and temperature (electrical gradient and total power dissipation), will be discussed. It should be maintained that for investigating the interaction of an electromagnetic wave with matter, the matter dielectric constant will be needed. At first, the ideal state is simulated in which the dielectric constant data are used from Table 2. In the ideal case, it was assumed that the imaginary part of the tissue dielectric constant, which is related to the photons absorptions rate, is small. Due to its ideal state, light deep penetration occurred in the tissue. The interaction results are illustrated in Fig. 4. The purpose of these simulations were to compare signal detections before (Fig. 4a and d) and after (Fig. 4b and e) the use of the nanosensor. It is clearly observed that before the nanosensor was used, the signal was continuous and its resolutions were very low. Therefore, the detection of the original signal was very hard, considering the auto-fluorescence photon radiations. In the case of the electric field gradient, which is illustrated in Fig. 4d, non-uniformity is produced, which is due to the distinct interaction of matter with light. However, it is limited to continuity and sharp alteration. In contrast, the signal clarity in the nanosensor output increases, which is attributed to the NPs plasmonic resonance effect on the signals. In other word, the output signals are sampled by the NPs, by which their distance and amplitudes are easily manipulated. Indeed, the output signal digitization was the main purpose of this study in order help to detect the output signal with high resolution. The 2-D illustration of the dissipation power energy and electric gradient field are depicted in Fig. 4c and f, respectively. In these figures, the signal digitization by the NPs plasmonic effect is easily observable. Due to the different interactions of light with distinct materials, the non-uniform profile is illustrated in all figures. The non-uniformity at the output of the nanosensor dramatically increases, which will be detected by detectors. It should be noted that the plasmonic resonance was enhanced in the portable nanosensor (for instance, we can consider the results of Fig. 4c and f), which contains arrays of regulated and patterned multi-shell NPs, is comparable with surface-enhanced Raman nanodomes,27 plasmonic nanogalaxies,28 and surface enhancing by Au NPs29 results, which contain some theoretical-experimental works in this field but with different applications. In the referred works, the experimental data for enhancement by NPs plasmonic resonance have been reported to be an order of 106 to 107 folds. In the following, we will explore the original results of our work, which are related to the detection of the non-uniform field created by different tissues. In the real medium, the penetration of light into tissue is limited and a very small fraction of light can reach the detector. For this, the data from Table 3 are considered. In this case, the imaginary part of the tissue is supposed to be larger than the ideal one. Thus, the majority of photon flux density is absorbed into tissues and only a little density, i.e., about 1000 w m−2 reach the detector. Therefore, we are trying to design a nanosensor, which is able to detect a small fraction of scattered light. The nanosensor was designed by manipulating some parameters, such as NP materials (core, shell_I, and shell_II), NPs radius, core–shell plasmonic hybridization, medium material and irradiance wavelength. Nanosensor output signals, which are detected as temperatures or optical forces are illustrated in Fig. 5 and 6, respectively. All the subfigures in Fig. 5 indicate that the digitized non-uniformity in the temperature profiles is related to the interaction of light with different matter and the NPs plasmonic effect. In other words, when the continuum non-uniform field interacts with NPs, the photons are amplified and regularly sampled, which are due to NPs plasmonic resonance. Thus, the amplified and digitized signal can be easily measured. Fig. 5a depicts the temperature profile for the tissue dielectric constants from Table 3. It illustrates that the fraction of light absorbed into the tissue is too high in the non-ideal state and consequently the temperature will be low. Thus, in the following, the alteration of the nanosensor parameter effects on total dissipation power and temperature, are investigated. It should be noted that the other parts of Fig. 5 are related to the manipulation of parameters, i.e., either nanosensor parameters or tissues optical properties. As shown in Fig. 5b, the absorption rate of a tumor increases, which is related to the tumor construction area and its physiological condition (neoplasia in tumor4,25). This figure shows that the temperature difference between normal and abnormal tissue increases compared to Fig. 5a. In other words, the majority of photons in the tumor area are absorbed, which leads to severe non-uniformity. Moreover, the increase in the tumor dielectric imaginary part might be related to the active targeting by functionalized NPs. In active targeting, a large amount of functionalized NPs find the tumor and accumulate near to it, which results in an increased absorption rate in this region. Furthermore, as illustrated in Fig. 3a, the NPs plasmonic resonance acts as a photon amplifier and sampler of the received signals. The incidence wavelength effect on light propagation in the tissue and output signal is illustrated in Fig. 5c. It can be easily observed that the temperature magnitude increases, which is not observed in the earlier figures; this is attributed to the increase in the received photon flux on the nanosensor surface at a higher wavelength. The effect of NPs distance with each other on the output is illustrated in Fig. 5d. When the NPs distances from each other increase, the NPs plasmonic–plasmonic interaction decrease. Hence, the sole effect of the NPs plasmonic detector screen can be considered. Based on its results, it is considered as an appropriate case for designing a nanosensor. In another case, the simultaneous effect of increasing the tumor absorption rate and incidence wavelength was investigated and its results are illustrated in Fig. 5e. In this case, the large amount of light propagation at 1550 nm and severe non-uniformity due to the tumor absorption coefficient are realized and their influence on total power dissipation is easily observed. In Fig. 5f, the concurrent effect of the incidence wavelength at 6199 nm and NPs distance increasing from each other is perused. In this figure, the digitization is not fully done by the NPs, which is contributed to the excitation wavelength at 6199 nm. It is correct that the penetration of light is severely increased by increasing the wavelength, but this wavelength is far-away from the NPs plasmonic resonance and thus, the NP carriers do not effectively resonate in this wavelength. This is contributed to the photoelectric effect in the case of the interaction of an electromagnetic wave with the NPs. In the following, the effect of the medium materials and NPs core and second shell materials were simulated, which is depicted in Fig. 5g and h, respectively. It can be obviously observed that the use of SiO2 rather than the silica aerogel results in a decrease in the temperature because the silica thermal conductivity is bigger than the silica aerogel. The following simulation is related to the nanosensor changing the parameters effect on the optical force as another measurable signal of the detector. Similar to Fig. 5, all the subfigures in Fig. 6 indicate that the sampled non-uniformity in the optical force profile is related to the interaction of light with different matter and the NPs plasmonic effect. Fig. 6a depicts the optical force in the non-ideal state. It should be noted that the optical force is directly related to the electric field gradient in the medium. Hence, the non-uniformity of the electric field gradient directly affects the optical force. The alteration of the tumor absorption coefficient, incidence wavelength, NPs distance from each other, medium and core–shell materials have severe impacts on the electrical field non-uniformity and its magnitude. Therefore, it should be maintained that changing the electrical field non-uniformity and its magnitude will dramatically affect the optical force profile, which are illustrated in Fig. 6b–h, respectively. These figures reveal that the magnitudes of the optical force due to the NPs plasmonic effect, are in the order of μN, which can be easily measured by piezoelectric detectors.25,26 Indeed, the detector senses the distinct forces due to the difference in tissues, which is related to the non-uniformity
in the detected signals. It can be easily noted that the signals are digitized by the NPs, which lightly cause them to be measured. The maximum optical force, shown in Fig. 6h, is due to the alteration of medium materials. Fig. 5 and 6 reveal that the NPs plasmonic resonance remarkably affects temperature and optical force. That is to say, NPs plasmonic resonance manipulates the total dissipation energy and electric field non-uniformity near the field state and leads to photon amplification. Furthermore, the tumor active targeting, which was influenced by functionalized NPs was investigated in the present study, which is shown in Fig. 6e. Finally, the simulation results claim that the output sampled signals could be easily and clearly detected by force and temperature detectors. Thus, easy breast tomography will be carried out with no need for clinics and their equipment.
Table 2 Initial properties of tissue dielectric constant and nanosensor specification (ideal state); ntumor is approximated based on normal tissue
| ntissue19 |
1.37 + i0.016 |
| ntumor |
1.37 + i0.16 |
| Wavelength |
833 nm |
| NPs_material (core and shell_II) |
Au |
| Medium_material |
Silica aerogel |
| NPs_material (shell_I) |
SiO2 |
 |
| | Fig. 4 Total power dissipation density (w m−3) and electrical field gradient vs. y-coordinates; (a) total power dissipation density before nanosensor, (b) total power dissipation density after nanosensor, (c) 2-D illustration of total power dissipation around the NPs (d) electric field gradient before nanosensor, (e) electric field gradient after nanosensor, (f) 2-D illustration of electric field gradient around the NPs. | |
Table 3 General properties of tissue dielectric constant and nanosensor specification (non-ideal state)
| ntissue |
1.37 + i0.16 |
| ntumor |
1.37 + i0.5 |
| Wavelength |
833 nm |
| NPs_material (core and shell_II) |
Au |
| Medium_material |
Silica aerogel |
| NPs_material (shell_I) |
SiO2 |
 |
| | Fig. 5 Temperature (K) vs. y-coordinate (nm); (a) non-Ideal state simulation (based on Table 3); (b) increasing of imaginary part of tumor indices (ntumor = 1.37 + i0.9); (c) changing of wavelength to 1550 nm; (d) increasing of NPs distance from each other (from 110 nm to 400 nm); (e) increasing of imaginary part of ntumor = 1.37 + i0.9 and alteration of wavelength to 1550 nm; (f) increasing of NPs distance from each other (from 110 nm to 400 nm) and alteration of wavelength to 6199 nm; (g) alteration of medium materials: SiO2 rather than silica aerogel; (h) changing of core and second shell materials: Ag rather than Au. | |
 |
| | Fig. 6 Optical force (N) vs. y-coordinate (nm); (a) non-Ideal state simulation (based on Table 3); (b) increasing of imaginary part of tumor indices (ntumor = 1.37 + i0.9); (c) changing of wavelength to 1550 nm; (d) increasing of NPs distance from each other (from 110 nm to 400 nm); (e) increasing of imaginary part of ntumor = 1.37 + i0.9 and alteration of wavelength to 1550 nm; (f) increasing of NPs distance from each other (from 110 nm to 400 nm) and alteration of wavelength to 6199 nm; (g) alteration of medium materials: SiO2 rather than silica aerogel; (h) changing of core and second shell materials: Ag rather than Au. | |
Conclusions
In this study, a portable nanosensor for the early and easy detection of carcinoma breast cancer was designed and simulated. Based on theoretical simulations, it was indicated that the backscattered photons, which reached the sensor surface were very small and were significantly affected by background noises. Hence, it is very hard to detect them without any photon intensification. Moreover, the interaction of the incidence light with matter with different optical properties caused electrical field non-uniformity, which should be detected. For this, the NPs based sensor in which the NPs plasmonic resonance causes to the signal amplification and digitization, is employed. It is notable that the original drawback of earlier works, which was related to the unclear and attenuated signals on the detector, is improved by the NPs plasmonic influence. In other words, the photons received on the nanosensor surface are sampled and amplified by NPs, which leads to the easy detection of a large amount attenuated signals. Thus, the digitized signal is easily filtered and controlled for other signal processing. Finally, easy breast tomography will be carried out with no need for clinics and their equipment.
Acknowledgements
This study is partially supported by photonic group of school of engineering-emerging technologies, University of Tabriz and moreover is partially supported by EU-FP7-IAPP NanobacterphageSERS and Biyomedtek/Nanobiyomedtek. A. Salmanogli was supported by this project as a recruited fellow. E. Piskin was supported as member of Turkish Academy of Sciences (TUBA).
Notes and references
- N. Paras, Multimodal Imaging, NCI Reports, 2010 Search PubMed.
- G. Lanza, New Contrast Agents with Improved Spatial and Temporal Resolution, NCI Reports, 2010 Search PubMed.
- S. Nie, Nanotechnology for Image Guided Interventions, NCI Reports, 2010 Search PubMed.
- R. A. Gatenby and R. J. Gillies, Nat. Rev. Cancer, 2004, 4, 891 CrossRef CAS PubMed.
- M. Kara Bucci, A. Bevan and M. Roach, Ca-Cancer J. Clin., 2005, 55, 117 CrossRef.
- K. L. Kimberly, K. Christine and F. R. William, Obstet. Gynecol. Clin. N. Am., 2005, 32, 627 CrossRef PubMed.
- A. E. Omoti and C. E. Omoti, Pharm. Pract., 2006, 4, 55 Search PubMed.
- P. F. Jiao, H. Y. Zhou, L. X. Chen and B. Yan, Curr. Med. Chem., 2011, 18, 2086 CrossRef CAS.
- A. SalmanOgli, Cancer Nanotechnol., 2011, 2, 1 CrossRef CAS PubMed.
- G. von Maltzahn, J.-H. Park, K. Y. Lin, N. Singh, C. Schwöppe, R. Mesters, W. E. Berdel, E. Ruoslahti, M. J. Sailor and S. N. Bhatia, Nat. Mater., 2011, 10, 545 CrossRef CAS PubMed.
- B. Van de Broek, N. Devoogdt, A. D'Hollander, H. L. Gijs, K. Jans, L. Lagae, S. Muyldermans, G. Maes and G. Borghs, ACS Nano, 2011, 5, 4319 CrossRef CAS PubMed.
- R. Bardhan, Nanostructures for Plasmon Enhanced Fluorescence Sensing: From Photophysics to Biomedicine, PhD thesis, Texas University, Houston, 2010.
- A. SalmanOgli and A. Rostami, IEEE Trans. Nanotechnol., 2013, 12(12), 831 CrossRef CAS.
- A. SalmanOgli and A. Rostami, IEEE Trans. Nanotechnol., 2013, 12, 558 Search PubMed.
- V. Wong and M. A. Ratner, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 73, 075416 CrossRef.
- A. SalmanOgli, A. Rostami, M. Faranoush, M. dolatyari and G. Rostami, RSC Adv., 2014, 4, 30984 RSC.
- W. Cheong, S. A. Prahl and A. J. Welch, IEEE J. Quantum Electron., 1990, 26, 2166 CrossRef.
- J. H. Joseph and W. J. Winsombe, J. Atmos. Sci., 1976, 33, 2452 CrossRef.
- H. Ding, J. Q. Lu, W. A. Wooden, P. J. Kragel and X.-H. Hu, Phys. Med. Biol., 2006, 51, 1479 CrossRef PubMed.
- A. O. Govorov and H. H. Richardson, Nano Today, 2007, 2(2), 30 CrossRef.
- A. SalmanOgli and A. Rostami, IEEE Trans. Nanotechnol., 2012, 11, 1183 CrossRef.
- S. Haltiwanger, The Electrical Properties of Cancer Cells, http://www.royalrife.com/haltiwanger1.pdf.
- B. Ro, Med. Times, 1976, 95(95), 657 Search PubMed.
- Y. Jiang, J. P. Grbovic, C. Y. Cantrell and J. P. Freyer, Biophys. J., 2005, 89, 3884 CrossRef CAS PubMed.
- J. Xu, M. J. Dapino, D. Gallego-Perez and D. Hansford, Sens. Actuators, A, 2009, 153, 24 CrossRef CAS PubMed.
- N. Ferrell, J. Woodard and D. J. Hansford, Sens. Actuators, A, 2011, 170, 84 CrossRef CAS PubMed.
- C. J. Choi, Z. Xu, H. Y. Wu, G. L. Liu and B. T. Cunningham, Nanotechnology, 2010, 21, 415301 CrossRef PubMed.
- Z. Zhang, P. Yang, H. Xu and H. Zheng, J. Appl. Phys., 2013, 113, 033102 CrossRef PubMed.
- A. Gopinath, S. V. Boriskina, W. R. Premasiri, L. Ziegler, B. M. Reinhard and L. Dal Negro, Nano Lett., 2009, 9, 3922 CrossRef CAS PubMed.
|
| This journal is © The Royal Society of Chemistry 2015 |
Click here to see how this site uses Cookies. View our privacy policy here. 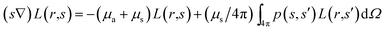
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ)
θ) ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (1/4π){2fδ(1 − cos
(1/4π){2fδ(1 − cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ) + (1 − f)(1 + 3g′
θ) + (1 − f)(1 + 3g′![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ)}
θ)}
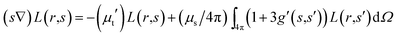
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
=![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Lcol(r,s)
Lcol(r,s) ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) +
+ ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ld(r,s)
Ld(r,s)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (1 − rs)πF0(r)exp(−μt′z/μ0)δ(μ − μ0)
(1 − rs)πF0(r)exp(−μt′z/μ0)δ(μ − μ0)