Facile method of preparation of PbS films for NO2 detection
Abstract
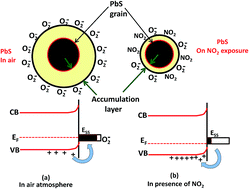
A simple and inexpensive chemical bath deposition method was employed for the preparation of lead sulfide (PbS) thin films. Thin films of PbS were characterized using XRD, SEM, TEM, SAED, contact angle and two probe techniques. The gas sensing properties of PbS thin films (t = 0.443 μm) have been investigated for H2S, NH3, C2H5OH, CH3OH, Cl2 and NO2. PbS thin films have been found to be more highly sensitive and selective towards oxidizing NO2 gas than the other test gases. The PbS sensor exhibits a maximum response of 74% towards 100 ppm of NO2 gas with a response time of 20 s. PbS thin films are able to detect up to 5 ppm NO2 concentration at room temperature with excellent reproducibility and stability. The sensing mechanism of PbS thin films towards NO2 gas is also discussed. The interaction of NO2 gas with PbS film was also investigated using impedance spectroscopy.

 Please wait while we load your content...
Please wait while we load your content...