Electrical characteristics of gallium–indium–zinc oxide thin-film transistor non-volatile memory with Sm2O3 and SmTiO3 charge trapping layers
Abstract
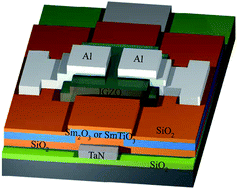
In this study, we report the structural and electrical characteristics of high-κ Sm2O3 and SmTiO3 charge trapping layers on an indium–gallium–zinc oxide (IGZO) thin-film transistor (TFT) for non-volatile memory device applications. The IGZO TFT non-volatile memory featuring a SmTiO3 charge trapping layer exhibited better characteristics, including a larger memory window (2.7 V), long charge retention time (105 s with charge loss <15%) and better endurance performance for program/erase cycles (104), compared with a Sm2O3 charge trapping layer. These results can be attributed to the SmTiO3 film possessing a high dielectric constant and deep trapping level. The high-κ SmTiO3 is an excellent candidate for use as the trapping layer in IGZO TFT non-volatile memories.

 Please wait while we load your content...
Please wait while we load your content...