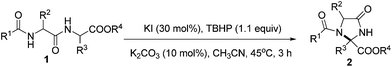
Dehydrogenative cyclization of N-acyl dipeptide esters for the synthesis of imidazolidin-4-ones†
Hui Yu* and
Jie Shen
Department of Chemistry, Tongji University, 1239 Siping Road, Shanghai, 200092, P. R. China. E-mail: yuhui@tongji.edu.cn; Fax: +86 21 65981097; Tel: +86 21 65981097
First published on 6th January 2015
Abstract
A dehydrogenative cyclization reaction for the synthesis of imidazolidin-4-ones was developed under mild conditions. Using tert-butyl hydroperoxide as oxidant and potassium iodide as catalyst, N-acyl dipeptide esters were converted to imidazolidin-4-ones in an atom-economical intramolecular C–N bond formation process in good yields.
In recent years, the direct α-functionalization of α-amino acid derivatives has gained increasing consideration as a powerful tool for the synthesis of non-natural α-amino acids required to assemble artificial peptides with novel properties.1 This method could also be used for the direct modification of existing peptides in the pursuit of enhanced stability and bioactivity.2 Among the developed methods for the α-functionalization of α-amino acids ester, the cross-dehydrogenative coupling (CDC) of C–H bonds has attracted much attention because of their atom-economical and environmentally friendly properties.3 N-Aryl and N-acyl glycine derivatives were popular substrates for the investigation of such reactions.4 Under transition metals catalyzed conditions, glycine derivatives were oxidized to generate aldimine intermediates, which were attacked subsequently by nucleophiles such as indoles, alkynes, and ketones to form new C–C bonds, and α-tertiary amino acid derivatives were obtained as the products. However, reports on the oxidative preparation of α-quaternary amino acid derivatives are rare for the difficult formation of ketimine intermediates. You and co-workers introduced a 2-pyridinecarbonyl group onto α-tertiary amino acid derivatives as an auxiliary group to promote the formation of ketimine intermediate and then an α-quaternary carbon centre was established by subsequent nucleophilic attack5 (Scheme 1a). Herein, we described a dehydrogenative cyclization process of dipeptide esters,6 which allows for simple access to N-heterocyclic compound containing α-quaternary carbon centre under transition metal-free conditions7 (Scheme 1b).
 | ||
| Scheme 1 Dehydrogenative coupling reaction for the formation of α-quaternary amino acid derivatives. | ||
Imidazolidin-4-ones are important N-heterocyclic compounds necessary in organic and pharmaceutical chemistry, and these compounds are used widely as herbicides and bactericides such as imazapyr, imazapic, and fenamidone.8 Traditional methods towards the synthesis of imidazolidin-4-one generally required multiple steps and harsh conditions.9 Steckhan and co-workers explored a convenient way to synthesis imidazolidin-4-ones by the cyclization of dipeptide esters, but the electrolysis condition using Pt as the anode must be determined.10 As a continuation of our efforts on the oxidative functionalization of C–H bonds adjacent to an amide N atom,11 we wish to present our work on the synthesis of imidazolidin-4-ones by an intramolecular functionalization of α-peptido C–H bond in an efficient manner.
Initially, N-acyl protected dipeptide ethyl ester N-Bz-Gly-α-PhGlyOEt 1a was chosen as the model substrate to optimize the reaction conditions of this dehydrogenative coupling cyclization. As shown in Table 1, the reaction of 1a with 1.1 equiv. TBHP (5 M solution in decane) was firstly examined in CH3CN with TBAI (30 mol%) as the catalyst at 45 °C. 3 hours later, all the start material disappeared monitored by TLC and the desired product imidazolidin-4-one 2a was isolated in 70% yield (Table 1, entry 1). When other iodide source such as I2, NIS and NH4I was used as the catalyst, the desired product 2a was obtained in low yield (Table 1, entries 2–4). CuI was also tested and no product could be found (Table 1, entry 5). When alkali metal iodides were used as the catalyst, 2a could be obtained in excellent yield and KI gave better result than NaI and CsI (Table 1, entries 6–8). Increasing the reaction temperature to 60 °C or decreasing the reaction temperature to room temperature led to lower yield of 2a (Table 1, entries 9–10). Other commercial oxidants such as TBHP (70% solution in water) and CHP were also tested to give lower yields of 2a, and in case of DTBP, no product was found (Table 1, entries 11–13). Different bases were screened and K2CO3 remained as the best one (Table 1, entries 14–16). Changing the solvent to EtOAc, toluene or CH2Cl2 resulted in the formation of 2a in only 0–25% yields (Table 1, entries 17–19). Finally, it was found that 92% yield of 2a could be isolated when the amount of K2CO3 was reduced to 10 mol%. On the basis of these results, entry 20 represents the best conditions.
| Entry | Catalyst (0.3 equiv.) | Oxidative (1.1 equiv.) | Solvent (2 mL) | Base (1.0 equiv.) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction condition: 0.5 mmol 1a, 0.55 mmol TBHP (5 M in decane), in 2.0 mL CH3CN at 45 °C for 3 h.b Isolated yield.c Reaction carried out at 60 °C.d Reaction carried out at room temperature.e 10 mol% K2CO3 was used. DTBP = di-tert-butyl peroxide. CHP = cumene hydroperoxide. | |||||
| 1 | TBAI | TBHP | CH3CN | K2CO3 | 70 |
| 2 | I2 | TBHP | CH3CN | K2CO3 | 20 |
| 3 | NIS | TBHP | CH3CN | K2CO3 | 40 |
| 4 | NH4I | TBHP | CH3CN | K2CO3 | NR |
| 5 | CuI | TBHP | CH3CN | K2CO3 | NR |
| 6 | NaI | TBHP | CH3CN | K2CO3 | 60 |
| 7 | KI | TBHP | CH3CN | K2CO3 | 90 |
| 8 | CsI | TBHP | CH3CN | K2CO3 | 62 |
| 9 | KI | TBHP | CH3CN | K2CO3 | 45c |
| 10 | KI | TBHP | CH3CN | K2CO3 | 61d |
| 11 | KI | TBHP aq. | CH3CN | K2CO3 | 83 |
| 12 | KI | CHP | CH3CN | K2CO3 | 75 |
| 13 | KI | DTBP | CH3CN | K2CO3 | NR |
| 14 | KI | TBHP | CH3CN | Cs2CO3 | 73 |
| 15 | KI | TBHP | CH3CN | DBU | 80 |
| 16 | KI | TBHP | CH3CN | Et3N | NR |
| 17 | KI | TBHP | EtOAc | K2CO3 | 25 |
| 18 | KI | TBHP | Toluene | K2CO3 | 15 |
| 19 | KI | TBHP | CH2Cl2 | K2CO3 | NR |
| 20 | KI | TBHP | CH3CN | K2CO3 | 92e |
Under the optimized reaction conditions, the scope of substrate was investigated with results summarized in Table 2. Firstly, the N-terminal protective group of the substrate was examined. Regardless of the electron-donating or electron-withdrawing group on the benzene ring, substituted benzoyl protected dipeptide ethyl esters 1b and 1c gave the corresponding imidazolidin-4-ones in good yields (Table 2, entries 1 and 2). 2-Naphthoyl protected dipeptide ethyl ester 1d also afforded the desired product in 86% yield under the optimized reaction (Table 2, entry 3). Substrates containing heterocyclic protective group such as 2-furoyl and 2-thenoyl reacted smoothly to furnish the product in moderate yields (Table 2, entries 4 and 5). Aliphatic acyl such as methylacryloyl protected dipeptide ethyl ester 1g could also be converted into the corresponding products in good yields, but acetyl protected dipeptide ethyl ester 1h remained untouched (Table 2, entries 6 and 7). N-Boc protected dipeptide ethyl ester 1i decomposed under the optimized reaction and no product could be found (Table 2, entry 8). Next, the effect of the α-substituent group adjacent to ester was studied. The reaction proceeded smoothly to afford the corresponding product in good yield when R3 was changed to 4-ClPh group, but failed with H and Me (Table 2, entries 9 and 10). Dipeptide methyl ester N-Bz-Gly-α-PhGlyOMe 1l also gave good yield under the reaction conditions (Table 2, entry 11). Moreover, the diastereoselectivity of this cyclization process was also investigated. N-Bz-L-Ala-α-PhGlyOMe 1m (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of two diastereomers) gave the corresponding product 2m in moderate yield and diastereoselectivity (73% yield, 3
1 mixture of two diastereomers) gave the corresponding product 2m in moderate yield and diastereoselectivity (73% yield, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 d.r.), and cis-2m was identified as the major stereoisomer in accordance with the literature.10 The same result was obtained if each of the pure diastereomer of 1m was employed to the reaction independently (Table 2, entry 12). Changing the N-terminal protective group to 4-methyl and 4-bromo-benzoyl group, the corresponding products 2n (79% yield, 4.5
1 d.r.), and cis-2m was identified as the major stereoisomer in accordance with the literature.10 The same result was obtained if each of the pure diastereomer of 1m was employed to the reaction independently (Table 2, entry 12). Changing the N-terminal protective group to 4-methyl and 4-bromo-benzoyl group, the corresponding products 2n (79% yield, 4.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 d.r.) and 2o (74% yield, 2.5
1 d.r.) and 2o (74% yield, 2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 d.r.) were obtained as anticipated (Table 2, entries 13 and 14). When dipeptide ethyl esters N-Bz-L-Ala-α-PhGlyOEt 1p and N-Bz-L-Val-α-PhGlyOEt 1q were utilized in this transformation, the desired products were isolated in 83% (2
1 d.r.) were obtained as anticipated (Table 2, entries 13 and 14). When dipeptide ethyl esters N-Bz-L-Ala-α-PhGlyOEt 1p and N-Bz-L-Val-α-PhGlyOEt 1q were utilized in this transformation, the desired products were isolated in 83% (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 d.r.) and 80% yield (5
1 d.r.) and 80% yield (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 d.r.) respectively (Table 2, entries 15 and 16).
1 d.r.) respectively (Table 2, entries 15 and 16).
| a Isolated yield. | |||
|---|---|---|---|
 |
|||
 |
Furthermore, substrates with extended carbon chain 3a–c were also employed aiming at medium member ring products. However, when 3a, 3b and 3c were subjected to the optimized reaction conditions, no desired cyclic products could be found and only peroxides 4a–c were isolated as the major products in 38%, 43% and 53% yield respectively (Scheme 2).
To gain insight into the reaction mechanism, 1.0 equiv. of TEMPO was added to the reaction, and the yield of 2a decreased remarkably to 35%, which suggests the possibility of a radical pathway. Based on the results in hand and the mechanism we reported before,11 a radical reaction process is proposed as shown in Scheme 3. The tert-butoxyl and tert-butylperoxy radicals were generated from the KI–TBHP system.12 Then the benzylic α-H atom on the C-terminal amino acid of dipeptide ester 1a was abstracted by the tert-butoxyl radical to afford radical A, which was trapped by the tert-butylperoxy radical to give the peroxide B. Finally, N-acylimino ester C was formed through the elimination of TBHP from B,13 which underwent cyclization reaction to give imidazolidin-4-one 2a in the presence of K2CO3.10 The mechanism detail requires further investigation.
Conclusions
In conclusion, we have demonstrated a simple, facile and transition-metal free approach to imidazolidin-4-ones through dehydrogenative cyclization of N-acyl dipeptide esters with moderate to good yields under mild conditions. Peroxide is believed to be the key intermediate for this cyclization process and further investigation into the mechanism is currently underway in our laboratory.Acknowledgements
This project was supported by the Fundamental Research Funds for the Central Universities (Tongji 20123231).Notes and references
- (a) Y. Yamada, T. Kubota, M. Nishio and K. Tanaka, J. Am. Chem. Soc., 2014, 136, 6505–6509 CrossRef CAS PubMed; (b) A. F. Taylor, S. K. Amundsen, M. Guttman, K. K. Lee, J. Luo, J. Ranish and G. R. Smith, J. Mol. Biol., 2014, 426, 3479–3499 CrossRef CAS PubMed; (c) Y. Sasaki and A. Ambo, Int. J. Med. Chem., 2012, 1–11 Search PubMed; (d) A. S. Saghyan, H. M. Simonyan, S. G. Petrosyan, A. V. Geolchanyan, G. N. Roviello, D. Musumeci and V. Roviello, Amino Acids, 2014, 46, 2325–2332 CrossRef CAS PubMed.
- (a) D. A. Butterfield, Free Radical Biol. Med., 2014, 74, 157–174 CrossRef CAS PubMed; (b) P. Liu, H. Zhang, H. Wang and Y. Xia, Proteomics, 2014, 14, 750–762 CrossRef CAS PubMed; (c) L. S. Witus, C. Netirojjanakul, K. S. Palla, E. M. Muehl, C. H. Weng, A. T. Iavarone and M. B. Francis, J. Am. Chem. Soc., 2013, 135, 17223–17229 CrossRef CAS PubMed.
- For reviews see: (a) S. A. Girard, T. Knauber and C. J. Li, Angew. Chem., Int. Ed., 2013, 52, 2–29 Search PubMed; (b) C. J. Scheuermann, Chem.–Asian J., 2010, 5, 436–451 CrossRef CAS PubMed.
- (a) C. Huo, C. Wang, M. Wu, X. Jia, H. Xie and Y. Yuan, Adv. Synth. Catal., 2014, 356, 411–415 CrossRef CAS; (b) W. T. Wei, R. J. Song and J. H. Li, Adv. Synth. Catal., 2014, 356, 1703–1707 CrossRef CAS; (c) J. Xie and Z. Z. Huang, Angew. Chem., Int. Ed., 2010, 49, 10181–10185 CrossRef CAS PubMed; (d) G. Zhang, Y. Zhang and R. Wang, Angew. Chem., Int. Ed., 2011, 50, 10429–10432 CrossRef CAS PubMed; (e) L. Zhao and C. J. Li, Angew. Chem., Int. Ed., 2008, 47, 7075–7078 CrossRef CAS PubMed.
- K. Li, G. Tan, J. Huang, F. Song and J. You, Angew. Chem., Int. Ed., 2013, 52, 12942–12945 CrossRef CAS PubMed.
- (a) A. Modak, U. Dutta, R. Kancherla, S. Maity, M. Bhadra, S. M. Mobin and D. Maiti, Org. Lett., 2014, 16, 2602–2605 CrossRef CAS PubMed; (b) G. Zhang, S. Wang, Y. Ma, W. Kong and R. Wang, Adv. Synth. Catal., 2013, 355, 874–879 CrossRef CAS.
- (a) K. Xu, Y. Hu, S. Zhang, Z. Zha and Z. Wang, Chem.–Eur. J., 2012, 18, 9793–9797 CrossRef CAS PubMed; (b) X. Zhang and L. Wang, Green Chem., 2012, 14, 2141–2145 RSC; (c) W. Wei, Y. Shao, H. Hu, F. Zhang, C. Zhang, Y. Xu and X. Wan, J. Org. Chem., 2012, 77, 7157–7165 CrossRef CAS PubMed; (d) E. Shi, Y. Shao, S. Chen, H. Hu, Z. Liu, J. Zhang and X. Wan, Org. Lett., 2010, 12, 3384–3387 CrossRef PubMed; (e) L. Ma, X. Wang, W. Yu and B. Han, Chem. Commun., 2011, 47, 11333–11335 RSC; (f) H. Yu, W. Huang and F. Zhang, Eur. J. Org. Chem., 2014, 3156–3162 CrossRef CAS; (g) H. Yu, F. Zhang and W. Huang, Synlett, 2014, 25, 843–846 CrossRef CAS PubMed.
- (a) D. Shaner, Bioact. Heterocycl. Compd. Cl., 2012, 83–89 CAS; (b) D. W. Ladner, Pestic. Sci., 1990, 29, 317–333 CrossRef CAS.
- (a) T. R. Blackmore and P. E. Thompson, Heterocycles, 2011, 83, 1953–1975 CrossRef CAS PubMed; (b) F. Ricardo, G. Jose, O. Eliandre, M. Rui and G. Paula, J. Org. Chem., 2007, 72, 4189–4197 CrossRef PubMed.
- A. Papadopoulos, B. Lewall, E. Steckhan, K. Ginzel, F. Knoch and M. Nieger, Tetrahedron Lett., 1991, 47, 563–572 CrossRef CAS.
- H. Yu and J. Shen, Org. Lett., 2014, 16, 3204–3207 CrossRef CAS PubMed.
- (a) R. A. Kumar, G. Saidulu, K. R. Prasad, G. S. Kumar, B. Sridhar and K. R. Reddy, Adv. Synth. Catal., 2012, 354, 2985–2991 CrossRef CAS; (b) Z. Q. Lao, W. H. Zhong, Q. H. Lou, Z. J. Li and X. B. Meng, Org. Biomol. Chem., 2012, 10, 7869–7871 RSC; (c) S. Tang, Y. Wu, W. Q. Liao, R. P. Bai, C. Liu and A. W. Lei, Chem. Commun., 2014, 50, 4496–4499 RSC; (d) X. Q. Li, X. S. Xu, P. Z. Hu, X. Q. Xiao and C. Zhou, J. Org. Chem., 2013, 78, 7343–7348 CrossRef CAS PubMed.
- (a) B. Schweizer-Chaput and M. Klussmann, Eur. J. Org. Chem., 2013, 666–671 CrossRef; (b) H. Blumenthal and J. Liebscher, ARKIVOC, 2009,(xi), 204–220 CrossRef CAS; (c) R. A. Kumar, G. Saidulu, B. Sridhar, S. T. Liu and K. R. Reddy, J. Org. Chem., 2013, 78, 10240–10250 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Detailed experimental procedures and characterization data for the products. See DOI: 10.1039/c4ra15019h |
| This journal is © The Royal Society of Chemistry 2015 |



