Organocatalytic enantioselective synthesis of 1-vinyl tetrahydroisoquinolines through allenamide activation with chiral Brønsted acids†
E. Manoni‡
,
A. Gualandi‡,
L. Mengozzi,
M. Bandini and
P. G. Cozzi*
ALMA MATER STUDIORUM – Università di Bologna, Dipartimento di Chimica “G. Ciamician”, Via Selmi 2, 40126 Bologna, Italy. E-mail: piergiorgio.cozzi@unibo.it
First published on 6th January 2015
Abstract
In this paper we present a new approach for the realization of tetrahydroisoquinoline scaffolds via stereoselective proto-activation of suitable allenamide precursors. The elusive and rather unstable iminium ion derived from acrylaldehyde is generated in situ and this electrophilic intermediate can be engaged in stereoselective intramolecular Friedel–Crafts-type allylic alkylation with electron-rich aromatic rings. The highest enantioselectivity for tetrahydroisoquinoline intermediates, obtained by organocatalytic transformation, is reported.
Introduction
Natural and synthetic 1-substituted tetrahydroisoquinolines such as emetine, norcoclaurine, solifenacine, morphine, aporphinoid and erythrocarine alkaloids have received increased attention in medicinal and synthetic chemistry due to their wide portfolio of pharmacological applications (Fig. 1).1,2The interest in this class of pharmaceutically active alkaloids has prompted flourishing research towards the development of innovative synthetic methods for their preparation.3 In particular, due to the presence of stereogenic centres in stereochemically defined manner, enantioselective catalysis represents the forefront of chemical research, with particular emphasis on transition metal-based approaches.4
In this segment, the asymmetric functionalization of isoquinoline cores focuses on the introduction of a substituent on the C(1)-position (Reissert reaction),5 as this stereocenter is present in most of the naturally occurring alkaloids.
The first enantioselective catalytic version of the Reissert reaction4 was introduced by Shibasaki in 2000,6 and the time frame testimonies the difficulties related to these reactions. Afterwards, a number of enantioselective methodologies has been successively described,7 however moderate levels of stereoinduction were usually recorded. Quite recently, a highly stereoselective copper-catalysed alkynylation of tetrahydroisoquinoline derivatives was described by Ma,8 providing a new methodology in the synthesis of alkaloids.9
Additionally, metal catalysed stereoselective hydrogenation reactions of isoquinolines and 3,4-dihydroisoquinolines gave an affordable and frequently used methodology for useful intermediates in high ee.10
However, both the described approaches are not metal-free. Avoiding the use of expensive and toxic metals in late-stage synthesis may be highly desirable in view of pharmaceutical applications. A direct approach towards tetrahydroisoquinoline alkaloids would be the bio-mimetic Pictet–Spengler condensation11 starting from a suitably functionalized 2-phenylethylamine, in the presence of Brønsted acids12 or chiral thiourea catalysts.13 However, although a considerable number of enantioselective organocatalysed Pictet–Spengler reactions has been reported, the amine precursors were restricted to quite nucleophilic tryptamines and their derivatives.14 Three important factors have contributed to establish the success of these transformations: (a) the highly nucleophilic indole system, (b) the use of N(1)-free indoles enabling the establishment of hydrogen bond recognition with the Brønsted acid catalyst, (c) the peculiar electronic properties of the indolyl core, able to engage secondary interactions with aromatic substituents of the chiral organocatalyst.
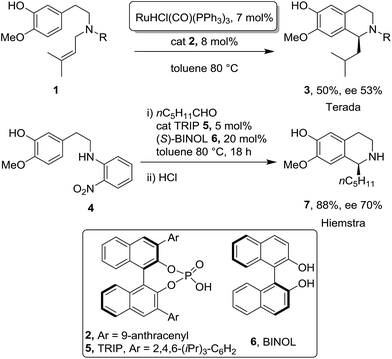
A phenylethylamine moiety was employed recently in a few remarkable examples of stereoselective Pictet–Spengler condensations under binary metal/metal-free catalytic regime. In details, Toda and Terada,15 reported a ruthenium-catalysed alkene isomerization combined with an enantioselective organocatalysed Pictet–Spengler type cyclization promoted by chiral phosphoric acid (Scheme 1). A fully organocatalytic enantioselective Pictet–Spengler cyclization was also described by Hiemstra,16 however extra steps were required for the preparation of suitable starting materials and for the preparation of the final alkaloids (Scheme 1). In both cases, the use of free OH groups is necessary in order to enhance both enantioselectivity and nucleophilic profile of the thematic moiety.
 | ||
| Scheme 1 Pictet–Spengler reactions with 2-phenylethylamine skeleton promoted by chiral phosphoric acids. | ||
In this paper, we describe a completely metal-free strategy for a stereoselective Friedel–Crafts-type cyclization to afford tetrahydroisoquinoline alkaloids, featuring synthetically flexible vinyl group at the α-position.17
Results and discussion
The access to α-vinyl-substituted tetrahydroisoquinolines using the Pictet–Spengler or Bischler–Napieralski approaches is problematic and unsuitable due to the difficulties to use acrylaldehyde as electrophile.18 Indeed, it has been demonstrated that only substituted vinyl moieties can be introduced through the Pictet–Spengler condensation.19The generation of an equivalent of acrylaldehyde, and its synthetically flexible route to α-vinyl-substituted tetrahydroisoquinoline building blocks, was described by Hiemstra and Rutjes in by means of [Sn(II)] or TFA-catalysed cyclization of allylic N,O-acetals (Fig. 2).20
 | ||
| Fig. 2 Different approaches to obtain the key synthetic intermediate for the synthesis of 1-vinyl tetrahydroisoquinolines. | ||
Allenamides appear as a suitable alternative synthetic route to this important intermediates. In particular, allenamides are highly susceptible towards metal and Brønsted acid promoted electrophilic activation21 and this was clearly demonstrated by Navarro-Vásquez and Domínguez in the TFA-catalysed synthesis of α-vinyl-substituted tetrahydroisoquinolines (Fig. 2).22 Moreover, allenamides were also used to access vinyl-isoquinolines by electrophilic activation with [gold(I)] complexes.23
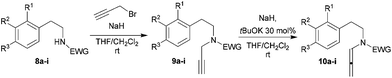
Based on these precedents in literature and recent findings by some of us,24 we envisioned the use of chiral Brønsted acids with allenamides, as a potent and effective way to prepare useful enantioenriched 1-vinyl tetrahydroisoquinoline precursors. We started our endeavour with the synthesis of the allenamides 10a–i carrying different electron-withdrawing groups (EWGs) at the nitrogen atom. The preparation of the amides 8a–i was straightforward and it is described in the ESI.† In order to improve the yield in the reaction with propargyl bromide, we carried out the propargylation of amides 8a–i in THF–CH2Cl2 due to the low solubility of the amides. During this step traces of corresponding allenamides 10a–i were isolated from the reaction mixture. The isolated propargylic amides 9a–i were then treated with tBuOK in THF following the described procedure22 to give the desired allenamides 10a–i that, however, were only obtained in low yields. After considerable efforts we found a more suitable and reproducible last-step procedure employing NaH as base (1 eq.) in the presence of a catalytic amount of tBuOK (30 mol%). The corresponding allenamides were obtained from low to excellent yields (Table 1).
| Entrya | Amide | R1/R2/R3 | EWG | Yieldb 9 (%) | Yieldb 10 (%) |
|---|---|---|---|---|---|
| a See ESI for detailed reaction conditions.b Isolated yields after chromatographic purification.c Ar = 3,5-(CF3)2C6H3. | |||||
| 1 | 8a | H/OMe/OMe | Ts | 89 | 91 |
| 2 | 8b | H/OMe/OMe | PhCO | 18 | 10 |
| 3 | 8c | H/OMe/OMe | ArCOc | 27 | 27 |
| 4 | 8d | H/OMe/OMe | Boc | 68 | 68 |
| 5 | 8e | H/OMe/OMe | CHO | 36 | 36 |
| 6 | 8f | H/OBn/OBn | CHO | 63 | 80 |
| 7 | 8g | H/–OCH2O– | CHO | 57 | 60 |
| 8 | 8h | OMe/OMe/H | CHO | 51 | 69 |
| 9 | 8i | H/OMe/H | CHO | 48 | 64 |
Therefore, allenamides 10a–e were efficiently cyclized to the desired racemic tetrahydroisoquinolines employing the reaction conditions described by Navarro-Vásquez and Domínguez (TFA 20 mol%, CH2Cl2, rt). The vinyl-substituted heterocyclic compounds 11a–e were isolated in moderate to good yields, through the Friedel–Crafts reaction of the iminium ion intermediates. Aromatic rings carrying EWGs were not investigated because it had already been reported that only electron-rich nucleophiles attach the iminium ion. In fact, substrates lacking of methoxy groups on the aromatic ring failed to give the desired product also after prolonged reaction time and harsh conditions (50% TFA, DMF, 70 °C).22 The formation of the six-membered ring was already shown to be favoured as homologous compounds failed to cyclize under the standard reaction conditions. It is worth to mention that adventitious water can result in hydrolysis of the secondary amide, as the iminium ion is quite unstable. Therefore, anhydrous conditions proved to be essential for the reaction.
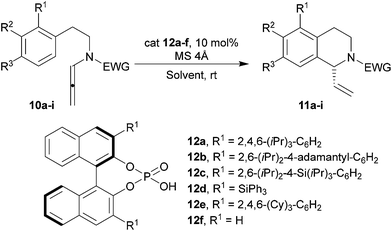
With the desired starting material in hand, we have investigated in details the stereoselective reaction by varying Brønsted acid, solvent, temperature, and using the differently substituted allenamides 10a–e (Table 2). Some of the Brønsted phosphoric acids screened were commercially available (12a, 12d), alternatively, they were prepared using standard protocols.25
| Entrya | Allenamide | Solvent | cat | Yieldb 11 (%) | eec 11 (%) |
|---|---|---|---|---|---|
| a All the reactions were carried out in dried conditions under nitrogen atmosphere by using 0.1 mmol of allenamides, 50 mg of 4 Å MS and 10 mol% of the catalyst, in 1 mL of solvent for 24 hours.b Isolated yields after chromatographic purification.c Determined by chiral HPLC analysis performed on the crude reaction mixture.d Determined by chiral HPLC analysis performed after reduction with LiAlH4 of the formyl protecting group to the corresponding methyl group (see ESI for details). | |||||
| 1 | 10a | DCM | 12a | 63 | 21 |
| 2 | 10b | DCM | 12a | 65 | 14 |
| 3 | 10c | DCM | 12a | 63 | 0 |
| 4 | 10d | DCM | 12a | 30 | 32 |
| 5 | 10e | DCM | 12a | 40 | 47 |
| 6 | 10e | Toluene | 12a | 18 | 72d |
| 7 | 10e | C6H5CF3 | 12a | 48 | 75d |
| 8 | 10e | C6H5F | 12a | 36 | 70d |
| 9 | 10e | C6H5CF3 | 12b | 27 | 79d |
| 10 | 10e | C6H5CF3 | 12c | 33 | 78d |
| 11 | 10e | C6H5CF3 | 12d | 40 | 81d |
| 12 | 10e | C6H5CF3 | 12e | 26 | 72d |
| 13 | 10f | C6H5CF3 | 12f | 0 | — |
| 14 | 10g | C6H5CF3 | 12f | 0 | — |
| 15 | 10h | C6H5CF3 | 12f | 0 | — |
| 16 | 10i | C6H5CF3 | 12f | 0 | — |
A selection of results is depicted in Table 2, clearly outlining the importance of the nitrogen-tethered EWGs (Table 2) in stabilizing the allenamide. In particular, although tosyl or other robust protecting groups (i.e. Boc, benzoyl, 3,5–(CF3)2–C6H3CO) proved competent under optimal conditions (cat = TFA or cat = phosphoric acid), low enantiomeric excesses were generally recorded with chiral promoters (Table 2, entries 1–4).
To improve the stereoselectivity of the transformation we hypothesised that the presence of a group able to interact by hydrogen-bond with the chiral phosphate could positively impact onto the stereochemical outcome of the protocol. As a result, we designed and realised the N-formyl allenamide via treatment of the corresponding amine with ethylformate under refluxing conditions for several hours (see ESI† for details).
Delightfully, our prediction was verified and the enantiomeric excess for the product 11e improved considerably (Table 2, entry 5). The fine-tuning of the solvent and the catalyst (Table 2, entries 6–12) allowed reaching an unprecedented ee of 81% for the synthesis of vinyl tetrahydroisoquinolines promoted by chiral Brønsted acid catalysis.
As we have already mentioned in the article, the presence of activated molecular sieves is quite crucial to obtain satisfying yields. Moreover, in the presence of molecular sieves the allenamides are not hydrolysed by adventitious water and can be recovered by chromatography. When very hindered chiral Brønsted acid, such as 12e, was used, 11e was isolated only in 26% yield (Table 2, entry 12). The enantiomeric excesses were evaluated by chiral HPLC analysis for all the derivatives. However, in the case of product 11e it was necessary to transform the formyl protecting group into the corresponding methyl by reduction (LiAlH4) in order to properly separate the enantiomers by chiral HPLC.
In order to evaluate the scope of this procedure, we have submitted allenamides 10f–i to the optimized reaction conditions. Unfortunately, in all cases we were unable to isolate any traces for the desired products using less hindered Brønsted acid 12f. In the case of the allenamides 10f, 10h and 10i the major hindrance of the substrates is probably retarding considerably the cyclization. In such conditions the hydrolysis of the iminium by adventitious water is competitive and we observed the presence of the corresponding amides. In the case of allenamides 10g we can suggest that the reduced nucleophilicity of the aromatic ring can be responsible for the failure of the reaction.
Finally, the absolute configuration of 11e, obtained with (R)-12d, was established to be R, by comparison of optical rotation with literature data.15 On the basis of this finding the model illustrated in Fig. 3 is suggested for the transition state of the reaction. In particular, given the importance of the formyl group, we assume that the recognition and the high enantiomeric excess obtained for the reaction is determined by the hydrogen bonding of the catalyst with the hydrogen atom of the formyl group.26
Experimental
General procedure for synthesis of compound 11e
To a solution of allenamide (0.1 mmol, 1 equiv.) in trifluorotoluene (1 mL) were added the Brønsted acid (0.01 mmol, 0.1 equiv.), molecular sieves 4 Å (50 mg) and then the reaction was stirred under nitrogen for 24 h. The solvent was removed under reduced pressure and the crude was directly purified by flash chromatography on silica gel (cyclohexane/ethyl acetate, 3/7) to give 11e: sticky white solid, 9.9 mg, 40% yield, 81% ee; the ee was determined via reduction of the product with LiAlH4 by HPLC analysis, Daicel Chiralpak® OD-H column: n-hexane/i-PrOH from 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, flow rate 0.70 mL min−1, 40 °C, λ = 285 nm: τmajor = 7.87 min, τminor = 14.93 min; 1H NMR (400 MHz, CDCl3) (two rotamers A
10, flow rate 0.70 mL min−1, 40 °C, λ = 285 nm: τmajor = 7.87 min, τminor = 14.93 min; 1H NMR (400 MHz, CDCl3) (two rotamers A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) B, ratio 1
B, ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1): δ 2.62–2.72 (2HB, m), 2.78–2.91 (2HA, m), 3.13 (1HB, ddd, J = 4.7 Hz, J = 11.0 Hz, J = 15.6 Hz), 3.45 (1HA, ddd, J = 4.2 Hz, J = 13.0 Hz, J = 13.0 Hz), 3.65 (1HA, dd, J = 5.8 Hz, J = 13.2 Hz), 3.80 (3HA, s), 3.81 (3HA + 3HB, s), 3.82 (3HB, s), 4.29 (1HB, m), 5.02 (1HB, d, J = 5.7 Hz), 5.09 (1HA, d, J = 17 Hz), 5.11 (1HB, d, J = 17.0 Hz), 5.19 (1HA, d, J = 10.1 Hz), 5.20 (1HB, d, J = 10.1 Hz), 5.78 (1HA, d, J = 5.6 Hz), 5.86–5.99 (1HA + 1HB, m), 6.55 (1HA, s), 6.56 (1HA + 1HB, s), 6.59 (1HB, s), 8.15 (1HA, s), 8.25 (1HB, s); 13C NMR (101 MHz, CDCl3): δ 27.6 (1CB), 29.2 (1CA), 35.1 (1CB), 40.2 (1CA), 52.7 (1CA), 55.8 (1CA), 55.8 (1CA), 55.9 (1CA), 56.0 (1CB), 58.7 (1CB), 110.1 (1CB), 110.6 (1CA), 111.3 (1CA), 111.5 (1CB), 117.0 (1CB), 117.3 (1CA), 125.2 (1CA + 1CB), 125.4 (1CA), 126.5 (1CB), 136.4 (1CA), 138.0 (1CB), 147.6 (1CB), 147.7 (1CA), 148.0 (1CA), 148.3 (1CB), 161.1 (1CA), 161.6 (1CB); ESI-MS: m/z = 248.2 [M + H]+, 270.0 [M + Na]+, 495.2 [2M + H]+; HMRS calcd for C18H19N: 247.12084; found 247.12075.
1): δ 2.62–2.72 (2HB, m), 2.78–2.91 (2HA, m), 3.13 (1HB, ddd, J = 4.7 Hz, J = 11.0 Hz, J = 15.6 Hz), 3.45 (1HA, ddd, J = 4.2 Hz, J = 13.0 Hz, J = 13.0 Hz), 3.65 (1HA, dd, J = 5.8 Hz, J = 13.2 Hz), 3.80 (3HA, s), 3.81 (3HA + 3HB, s), 3.82 (3HB, s), 4.29 (1HB, m), 5.02 (1HB, d, J = 5.7 Hz), 5.09 (1HA, d, J = 17 Hz), 5.11 (1HB, d, J = 17.0 Hz), 5.19 (1HA, d, J = 10.1 Hz), 5.20 (1HB, d, J = 10.1 Hz), 5.78 (1HA, d, J = 5.6 Hz), 5.86–5.99 (1HA + 1HB, m), 6.55 (1HA, s), 6.56 (1HA + 1HB, s), 6.59 (1HB, s), 8.15 (1HA, s), 8.25 (1HB, s); 13C NMR (101 MHz, CDCl3): δ 27.6 (1CB), 29.2 (1CA), 35.1 (1CB), 40.2 (1CA), 52.7 (1CA), 55.8 (1CA), 55.8 (1CA), 55.9 (1CA), 56.0 (1CB), 58.7 (1CB), 110.1 (1CB), 110.6 (1CA), 111.3 (1CA), 111.5 (1CB), 117.0 (1CB), 117.3 (1CA), 125.2 (1CA + 1CB), 125.4 (1CA), 126.5 (1CB), 136.4 (1CA), 138.0 (1CB), 147.6 (1CB), 147.7 (1CA), 148.0 (1CA), 148.3 (1CB), 161.1 (1CA), 161.6 (1CB); ESI-MS: m/z = 248.2 [M + H]+, 270.0 [M + Na]+, 495.2 [2M + H]+; HMRS calcd for C18H19N: 247.12084; found 247.12075.
Conclusions
In conclusion, we have reported the cyclization of allenamides to 1-vinyl tetrahydroisoquinolines in the presence of chiral phosphoric acids. The formyl protecting group turned out to be crucial for the catalysis by improving substrate/catalyst recognition via hydrogen bond interaction. The use of formyl group in other Brønsted acid catalysed reactions is suggested.27Acknowledgements
Bologna University, Fondazione Del Monte, and the European Commission through the project MOLARNET, FP7-ICT, 318516, are acknowledged for financial support.Notes and references
- (a) D. Jack and R. Williams, Chem. Rev., 2002, 102, 1669 CrossRef PubMed; (b) K. W. Bentley, Nat. Prod. Rep., 2006, 23, 444 RSC; (c) Q. Y. Zhang, G. Z. Tu, Y. Y. Zhao and T. M. Cheng, Tetrahedron, 2002, 58, 6795 CrossRef CAS; (d) A. J. Aladesanmi, C. J. Kelly and J. D. Leary, J. Nat. Prod., 1983, 46, 127 CrossRef CAS; (e) A. Zhang, J. L. Neumeyer and R. J. Baldessarini, Chem. Rev., 2007, 107, 274 CrossRef CAS PubMed; (f) K. Ye, Y. Ke, N. Keshava, J. Shanks, J. A. Kapp, R. R. Tekmal, J. Petros and H. C. Joshi, Proc. Natl. Acad. Sci. U. S. A., 1998, 95, 1601 CrossRef CAS; (g) A. Gualandi, L. Mengozzi, E. Manoni and P. G. Cozzi, Catal. Lett., 2015 DOI:10.1007/s10562-014-1396-0.
- K. Prasat, M. Chulabhorn and R. Somsak, Curr. Top. Med. Chem., 2014, 14, 239 Search PubMed.
- L. Mengozzi, A. Gualandi and P. G. Cozzi, Chem. Sci., 2014, 5, 3915 RSC.
- M. Chrzanowska and M. D. Rozwadowska, Chem. Rev., 2004, 104, 3341 CrossRef CAS PubMed.
- A. Reissert, Ber. Dtsch. Chem. Ges., 1905, 38, 1603 CrossRef CAS.
- K. Funabashi, H. Ratni, M. Kanai and M. Shibasaki, J. Am. Chem. Soc., 2001, 123, 10784 CrossRef CAS.
- (a) A. M. Taylor and S. L. Schreiber, Org. Lett., 2006, 8, 143 CrossRef CAS PubMed; (b) S. Wang and C. T. Seto, Org. Lett., 2006, 8, 3979 CrossRef CAS PubMed; (c) S. Wang, M. B. Onaran and C. T. Seto, Org. Lett., 2010, 12, 2090 Search PubMed; (d) T. Hashimoto, M. Omote and K. Maruoka, Angew. Chem., Int. Ed., 2011, 50, 8952 CrossRef CAS PubMed.
- W. Lin, T. Cao, W. Fan, Y. Han, J. Kuang, H. Luo, B. Miao, X. Tang, Q. Yu, W. Yuan, J. Zhang, C. Zhu and S. Ma, Angew. Chem., Int. Ed., 2014, 53, 277 CrossRef CAS PubMed.
- D. Ma, J. Dai, Y. Qiu, C. Fu and S. Ma, Org. Chem. Front., 2014, 1, 7892 Search PubMed.
- Y. M. Wilson, M. Dürrenberger, E. S. Nogueir and T. R. Ward, J. Am. Chem. Soc., 2014, 136, 8928 CrossRef CAS PubMed ; and references therein.
- E. D. Cox and J. M. Cook, Chem. Rev., 1995, 95, 1797 CrossRef CAS.
- (a) B. Herlé, M. J. Wanner, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem., 2011, 76, 8907 CrossRef PubMed; (b) I. Kerschgens, E. Claveau, M. J. Wanner, S. Ingemann, J. H. van Maarseveen and H. Hiemstra, Chem. Commun., 2012, 48, 12243 RSC; (c) N. Sewgobind, M. J. Wanner, S. Ingemann, R. de Gelder, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem., 2008, 73, 6405 CrossRef CAS PubMed.
- (a) R. S. Klausen and E. N. Jacobsen, Org. Lett., 2009, 11, 887 CrossRef CAS PubMed; (b) Y. Lee, R. S. Klausen and E. N. Jacobsen, Org. Lett., 2011, 13, 5564 CrossRef CAS PubMed.
- D. Huang, F. Xu, X. Lin and Y. Wang, Chem.–Eur. J., 2012, 18, 3148 CrossRef CAS PubMed.
- Y. Toda and M. Terada, Synlett, 2013, 752 CAS.
- E. Mons, M. J. Wanner, S. Ingemann, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem., 2014, 79, 7380 CrossRef CAS PubMed.
- For a review, see: M. Bandini, A. Melloni and A. Umani-Ronchi, Angew. Chem., Int. Ed., 2004, 43, 450 CrossRef PubMed.
- (a) H. Wang and A. Ganesan, Org. Lett., 1999, 1, 1647 CrossRef CAS; (b) A. van Loevezijn, J. D. Allen, A. H. Schinkel and G.-J. Koomen, Bioorg. Med. Chem. Lett., 2001, 11, 29 CrossRef CAS.
- (a) H. Wang and A. Ganesan, Org. Lett., 1999, 1, 1647 CrossRef CAS; (b) A. van Loevezijn, J. D. Allen, A. K. Schinkel and G.-J. Koomen, Bioorg. Med. Chem. Lett., 2001, 11, 29 CrossRef CAS.
- S. S. Kinderman, M. M. T. Wekking, J. H. van Maarseveen, H. E. Schoemaker, H. Hiemstra and F. P. J. T. Rutjes, J. Org. Chem., 2005, 70, 5519 CrossRef CAS PubMed.
- T. Lu, L. Zhenjie, Z.-X. Ma, Y. Zhang and R. P. Hsung, Chem. Rev., 2013, 113, 4862 CrossRef CAS PubMed.
- A. Navarro-Vázquez, D. Rodríguez, M. F. Martínez-Esperón, A. García, C. Saá and D. Domínguez, Tetrahedron Lett., 2007, 48, 2741 CrossRef PubMed.
- I. Singh, M. R. J. Elsegood and M. C. Kimber, Synlett, 2012, 565 Search PubMed.
- (a) M. Jia, G. Cera, D. Perrotta and M. Bandini, Chem.–Eur. J., 2014, 20, 9875 CrossRef CAS PubMed; (b) C. Romano, M. Jia, M. Monari, E. Manoni and M. Bandini, Angew. Chem., Int. Ed., 2014, 53, 13854 CrossRef CAS PubMed.
- For selected reviews of chiral phosphoric acid catalysis, see: (a) M. Terada, Chem. Commun., 2008, 4097 RSC; (b) M. Terada, Bull. Chem. Soc. Jpn., 2010, 83, 101 CrossRef CAS; (c) M. Terada, Synthesis, 2010, 1929 CrossRef CAS PubMed; (d) M. Terada, Curr. Org. Chem., 2011, 15, 2227 CrossRef CAS; (e) T. Akiyama, Chem. Rev., 2007, 107, 5744 CrossRef CAS PubMed; (f) F. Romanov-Michailidis, L. Guénée and A. Alexakis, Angew. Chem., Int. Ed., 2013, 52, 9266 CrossRef CAS PubMed.
- K. Kanomata, Y. Toda, Y. Shibata, M. Yamanaka, S. Tsuzuki, I. D. Gridneva and M. Terada, Chem. Sci., 2014, 5, 3515 RSC.
- M. Fañanás-Mastral, J. F. Teichert, J. A. Fernández-Salas, D. Heijnen and B. L. Feringa, Org. Biomol. Chem., 2013, 11, 4521 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Procedure for the reparation of the amides 8a–i and their fully characterization. Procedure and complete characterization of the compounds 10a–i and 11a–e. Copies of all 1H NMR and 13C spectra. Traces of HPLC analysis of racemic and enantioenriched compounds. See DOI: 10.1039/c4ra14942d |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2015 |