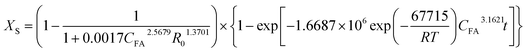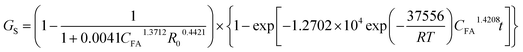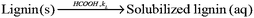DOI:
10.1039/C4RA14634D
(Paper)
RSC Adv., 2015,
5, 20992-21000
Kinetic modeling of atmospheric formic acid pretreatment of wheat straw with “potential degree of reaction” models†
Received
16th November 2014
, Accepted 16th February 2015
First published on 16th February 2015
Abstract
Pretreatment of lignocellulosic biomass by aqueous formic acid pertains to a biomass fractionation process to obtain multi-products for biorefining. In the present work, wheat straw was pretreated by aqueous formic acid under atmospheric pressure. The kinetics of delignification and polysaccharide solubilization were investigated using novel “potential degree of reaction” models, which were developed based on the multilayered structure of plant cell wall and reaction severity. Parameters termed as “potential degree of delignification (dD)” and “potential degree of solubilization (dS)” were introduced into the kinetic models for delignification and polysaccharide solubilization, respectively. The models fitted the experimental results very well. These models were also applicable as general models for describing the kinetics of various chemical pretreatments of different biomass feedstocks.
Introduction
Production of fuel ethanol from lignocellulosic biomass has been considered as a promising solution to the energy crisis, since lignocellulose is the most abundant organic material and offers a sustainable fashion for producing fuels.1,2 However, due to the negative effect of chemical compositions and physical structures, known as biomass recalcitrance, the enzymatic digestibility of lignocellulosic cellulose is greatly constricted.3,4 Therefore, various methods of pretreatment have been developed to overcome biomass recalcitrance. Among these methods, organosolv pretreatment is an effective way for both enhancing cellulose digestibility and fractionating biomass to obtain cellulose-rich solid, hemicellulosic sugars and high-purity lignin.5 Short-chain organic acids, such as formic acid, have been considered as a good lignin solvent for biomass delignification. Most of hemicelluloses and lignin are removed during formic acid pretreatment, which greatly exposes cellulose fibers for enzymatic hydrolysis.6,7
Some papers have been published on the kinetic modeling of delignification and polysaccharide solubilization in organosolv pulping or pretreatment of lignocellulosic biomass. The processes are usually considered as first-order reactions in a pseudo-homogenous system.8,9 There are generally two kinetic models for delignification and polysaccharide hydrolysis. One is to consider that biomass contains only separated type of polymer (lignin, cellulose or hemicellulose) and different phases of degradation are attributed to different rate-controlling mechanisms of overall process. In delignification process, the lignin is directly degraded to soluble fragments, and in the hydrolysis of polysaccharide, the polysaccharide is directly degraded to monosaccharides.10–13 The other is to view the polymer composed of several (normally two or three) fractions dissolving at different rates, and these fractions may be degraded consecutively or simultaneously. The consecutive model assumes that the lignin contains three types, namely initial, bulk and residual lignins, and they react successively. The simultaneous model assumes that different types of lignin begin to dissolve at the same time and react concurrently with pretreatment progress.14 For polysaccharide hydrolysis, one of the widely used models is two-fraction model, which considers that the polysaccharide consists of two fractions: fast (easy) and slow (difficult) hydrolyzing fractions which react simultaneously. A parameter, α, defined as the mass ratio of easy-to-hydrolyze fraction to the total polysaccharides has been introduced in the two-fraction model.15 It has been proved that considering the biomass contains several types of polymer could well describe the kinetic behavior of delignification or lignocellulosic polysaccharide hydrolysis.16,17 However, the link between the polymer fraction and the physical or chemical characteristics of the raw material, and the relationship between the kinetic parameter and the reaction severity (temperature, catalyst and solvent concentration) have not been established in these works. Therefore, it is necessary to establish a kinetic model that could accurately predict the behavior of delignification and polysaccharide hydrolysis with regard to biomass structure and reaction severity.
In our previous works,18,19 novel pseudo-homogeneous kinetic models for acetic acid (AcH) pretreatment of sugarcane bagasse were proposed by introducing the parameters of “potential degree of reaction”, namely “potential degree of delignification (dD)” for delignification and “potential degree of hydrolysis (hd)” for polysaccharide solubilization based on the multilayered structure of plant cell wall. The relationships between the parameters and the reaction severity were established mathematically. In the present work, wheat straw was pretreated by aqueous formic acid under atmospheric pressure, and the kinetics of delignification and polysaccharide solubilization were studied using these novel models. One of the objectives of this work is to further verify and improve the novel kinetic models by using another type of organic acid (formic acid) and lignocellulosic biomass (wheat straw), and obtain the corresponding kinetic parameters. This result can provide as a tool for further optimizing and controlling the process for efficiently utilizing this widely-distributed biomass feedstock. The second objective of this work is to further investigate the fundamental and significance of “potential degree of reaction” for kinetic modeling of biomass pretreatment.
Experimental
Materials
Wheat straw used in the experiment work was obtained from Shandong province in East China. It was air-dried and cut into 1–2 cm pieces before using. The main components of the wheat straw were determined to be 35.64 ± 2.12% glucan, 23.88 ± 1.08% xylan, 3.12 ± 0.16% arabinan, 2.03 ± 0.14% acetyl group, 24.72 ± 1.96% klason lignin, and 2.70 ± 0.31% acid-soluble lignin. The standard compounds used for HPLC calibration, including glucose, xylose and arabinose, were purchased from Sigma-Aldrich (Shanghai agent).
Pretreatment process
Formic acid pretreatment (FAP) of wheat straw was carried out in a 500 ml three-neck glass flask heated by constant temperature water bath (<100 °C) or electric jacket (≥100 °C) under atmospheric pressure. 10 g of cut wheat straw was packed into the flask followed by addition of 100 ml 60–88 wt% formic acid solution. Electrical stirring with a Teflon paddle was used at 300 rpm for keeping the system as homogeneous as possible. The reaction temperature of pretreatment was controlled at 70 °C, 80 °C, 90 °C, 100 °C and the atmospheric boiling point of the aqueous formic acid solution (∼107 °C). After delignification, the mixture was filtered. The obtained solid was first washed with 100 ml 60–88 wt% formic acid solution and then filtered to remove as much liquid as possible. The solid was then washed by running water till neutrality and dried for composition analysis.
Analytical methods
The main chemical compositions of the wheat straw and pretreated solid were determined according to NREL's Laboratory Analytical Procedure.20 The lignin, xylan and glucan solubilization ratios at time t [LS(t), XS(t) and GS(t), respectively] are defined as follows:| |
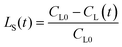 | (1) |
| |
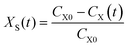 | (2) |
| |
 | (3) |
where CL0, CX0 and CG0 are initial concentrations (g l−1) of solid lignin, xylan and glucan in the pseudo-homogenous system; CL(t), CX(t) and CG(t) are concentrations (g l−1) of solid lignin, xylan and glucan in the pseudo-homogenous system at time t. The monosaccharides concentrations in the hydrolyzate were determined by a Shimadzu 10AVP HPLC system (Shimadzu Corp., Japan) equipped with a RID-10A refractive index detector and an Aminex HPX-87H column (300 × 7.8 mm, Bio-Rad, USA) at 65 °C. 5 mM H2SO4 was used as the eluent at a flow rate of 0.8 ml min−1 as described in our previous work.21
Data processing
The kinetic constants and parameters for “potential degree of reaction” were fitted according to experimental data by the least square method with Matlab 2013 software to minimize the objective function (fobjective), which was the quadratic sum of the difference between calculated data f(xi) and experimental data (yi) shown as the following expression:| |
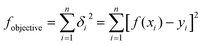 | (4) |
Kinetics
Kinetic models for delignification
In the present work, the liquid-to-solid ratio was 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (L kg−1). The delignification process was assumed to take place in a pseudo-homogeneous system. Since the pretreatment was performed under a mild condition (<110 °C without addition of mineral acid as a catalyst), the condensation of lignin can be negligible in comparison with the solubilization of lignin.10,13 The delignification process thus can be interpreted as the following simple solubilization of lignin from solid phase to liquid phase (Scheme 1):
1 (L kg−1). The delignification process was assumed to take place in a pseudo-homogeneous system. Since the pretreatment was performed under a mild condition (<110 °C without addition of mineral acid as a catalyst), the condensation of lignin can be negligible in comparison with the solubilization of lignin.10,13 The delignification process thus can be interpreted as the following simple solubilization of lignin from solid phase to liquid phase (Scheme 1):
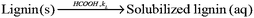 |
| | Scheme 1 Reaction of lignin solubilization in aqueous formic acid pretreatment of wheat straw. | |
Similarly to previously reported works of organosolv delignification kinetics, the delignification process is assumed to be a first-order reaction. Therefore, the rate of lignin solubilization into liquid phase is:
| |
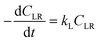 | (5) |
where
kL is the delignification rate constant,
CLR is the residual solid lignin concentration (g l
−1) in the pseudo-homogeneous system. According to the definition of lignin solubilization ratio (
LS) (
eqn (1)),
LS also can be expressed as:
| |
 | (6) |
where
CLR0 is the initial solid lignin concentration (g L
−1). Therefore, the rate of lignin solubilization can be described as:
| |
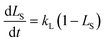 | (7) |
By solving eqn (7) with its corresponding initial conditions (LS = 0 at t = 0), the time dependent expression of LS can be described as follows:
This is the simplest kinetic model for delignification. Therefore, if the solubilization of lignin follows eqn (8), a line should be obtained by plotting ln(1 − LS) versus reaction time t. However, according to our experimental results, most the plots of ln(1 − LS) versus reaction time t showed an apparent deviation from linear relationship (ESI, Fig. 1†). The discrepancy indicates that the simplest model (eqn (7)) cannot be employed to accurately describe the kinetics of delignification during formic acid pretreatment. It also demonstrates that lignocellulosic lignin cannot be simply considered as a homogenous polymer having the same reactivity. Its solubilization rate and degree actually depend on reaction severity such as solvent concentration and reaction temperature.
As described in many reported works, it is commonly found that lignin cannot be completely removed during organosolv pretreatment. There is always a certain percentage of lignin remaining in the solid residue. In our previous work,18,19 we introduced a parameter termed “potential degree of delignification (dD)” to stand for the maximum solubilization ratio of lignin under a certain pretreatment severity. The possible reasons for the recalcitrance of a fraction of lignin to delignification are described as follows.
The plant cell wall has a multilayered structure generally consisting of three types of layers, i.e. middle lamella (M), primary wall (P) and secondary wall (S). The secondary wall also consists of three layers, called the S1, S2 and S3 lamellae, respectively.3 Cellulose, hemicellulose, and lignin have different distribution in these layers22 as shown in Fig. 1. Lignin is found to be the dominant composition in the outer portion of the compound middle lamellae. The proportion of lignin in the lignocellulosic matrix decreases with increasing distance into the middle lamella. The concentrations of lignin in the primary wall and in the S1 layer of the secondary wall are much higher than those in the S2 and S3 sections. Cell corner (CC) is found to have the highest lignin concentration on average. When eucalyptus wood was delignified by an organosolv process, delignification proceeded from the lumen wall towards the middle lamella before reaching the cell corners.23 It has been found that the lignin removal followed the order of S → M → CC by organosolv pretreatment of wheat straw, Taiwania (Taiwania cryptomerioides) and eucalyptus (Eucalyptus grandis).24 However, it also has been reported that the organosolv pulping of softwood was limited to preferential removal of lignin from the cell corner and middle lamella regions.25 Although no definite conclusion has been made on the mechanism and redistribution of lignin in the cell wall structure during organosolv delignification, it is obvious that lignin distributed in different part of cell wall have different reactivity in delignification process due to the complex cell wall structure. A possible reaction scheme thus has been proposed in the present work for the delignification and polysaccharide solubilization during organosolv pretreatment as shown in Fig. 1. At the beginning of the reaction, solvent and catalyst fill in the lumen as the liquid bulk phase. The solvent molecules and catalysts (H+) diffuses gradually by permeation into cell wall in the order of Lu → S3 → S2 → S1 → P → M, and chemical reactions start to take place in the wall layer where the catalyst and solvent reach. The reaction products (lignin and sugars) are dissolved in the solvents and diffuse from cell wall layers to the bulk phase. Since the molecular size of the lignin fragments and sugars including oligosaccharide and monosaccharide are much larger than those of catalysts, water and solvent molecules, the diffuse resistance of the reaction products is greater. Thereby, the reaction products may accumulate as the reaction proceeds in the cell wall thus gradually limiting the further diffusion of catalyst, solvent, water as well as newly formed products. It thus can be reasonably proposed that the mass diffusion limitation increases with the increase of distance from lumen to middle lamella, and corresponding reachability and reactivity of lignin and polysaccharide towards catalyst and solvent decrease. Thereby, there is a phenomenally maximum degree of delignification under a certain reaction severity. The reaction condition, namely temperature, concentration of solvent and reaction time determine the maximum degree of delignification and polysaccharide solubilization. Therefore, for lignin solubilisation, a parameter, i.e. “potential degree of delignification (dD)” defined as the observed maximum degree of delignification under a certain reaction severity is incorporated into the kinetic model. The observed rate of delignification is thus can be expressed with respect to the concentration of “reactive” lignin at time t (CLA) as:
| |
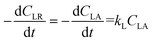 | (9) |
 |
| | Fig. 1 Graphical explanation of cell wall multilayered structure and its effect on the reaction of lignin and polysaccharides. The distributions of cellulose, hemicellulose and lignin were adapted from (ref. 22). | |
It illustrates that the rate of delignification is a pseudo-homogenous first-order reaction only with respect to the “reactive” fraction of lignin, but not to the whole lignin fraction in cell wall. However, the reactive lignin is difficult to determine, and in the experiments we usually determine the total residual lignin concentration (CLR). Based on the definition of dD, CLA can be expressed as:
| | |
CLA = CLR − CLR0(1 − dD)
| (10) |
where 0 ≤
dD ≤ 1. According to the definition of
LS (
eqn (6)), the kinetic model of delignification also can be expressed as:
| |
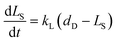 | (11) |
The integral forms of eqn (11) is
| | |
LS = dD[1 − exp(−kLt)]
| (12) |
Kinetic models for polysaccharide solubilization
For polysaccharide solubilisation, we also found that the simplest model (Saeman's model) cannot be applicable to our system. Similarly to delignification process, the process of polysaccharide solubilization can be described by the following reaction (Scheme 2):where k is the rate of polysaccharide solubilization. Similar kinetic model with introduction of a parameter “potential degree of solubilization (dS)” was developed. The kinetic model for xylan and glucan (cellulose) hydrolysis during formic acid pretreatment thus can be expressed as:| |
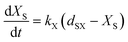 | (13) |
| |
 | (14) |
where kX and kG are the rate constants of xylan and glucan solubilization, respectively. Similarly, dSX and dSG are the “potential degrees of solubilization” for solid xylan and glucan, respectively (0 ≤ dS ≤ 1). The integral forms of eqn (13) and (14) are:| | |
XS = dSX[1 − exp(−kXt)]
| (15) |
| | |
GS = dSG[1 − exp(−kGt)]
| (16) |
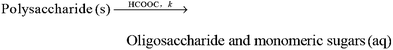 |
| | Scheme 2 Reaction for polysaccharide solubilization during aqueous formic acid pretreatment of wheat straw. | |
Results
Determination of kinetic constants
Wheat straw was pretreated with different concentrations of formic acid for different time at different temperatures. The solubilization ratios of lignin, xylan and cellulose versus pretreatment time are shown in Fig. 2–4. Corresponding regressed kinetic parameters are listed in Table 1. Both the rates and degrees of lignin and polysaccharides solubilizations were greatly dependent on pretreatment severity (formic acid concentration and reaction temperature). For example, when wheat straw was pretreated at 70 °C with 70% formic acid for 1 h, LS was only 0.19; however, it increased to 0.60 when temperature was increased to boiling temperature (∼107 °C). Similarly, LS increased to 0.30 when the formic acid concentration was increased to 88% at 70 °C for a 1 h reaction time, while the corresponding LS was 0.82 at 107 °C. Similar phenomenon was also observed for the rate of xylan and cellulose solubilizations.
 |
| | Fig. 2 Experimental and model-predicted data for lignin solubilization at different temperatures and formic acid concentrations during formic acid pretreatment of wheat straw. (A): 60% formic acid; (B): 70% formic acid; (C): 80% formic acid; (D): 88% formic acid. | |
 |
| | Fig. 3 Experimental and model predicted data for xylan solubilization at different temperatures and formic acid concentrations during formic acid pretreatment of wheat straw. (A): 60% formic acid; (B): 70% formic acid; (C): 80% formic acid; (D): 88% formic acid. | |
 |
| | Fig. 4 Experimental and model predicted data for glucan solubilization at different temperatures and formic acid concentrations during formic acid pretreatment of wheat straw. (A): 60% formic acid; (B): 70% formic acid; (C): 80% formic acid; (D): 88% formic acid. | |
Table 1 Kinetic constants determined by regression of experimental data
| Temperature (°C) |
Kinetic constants (h−1 or null) |
Formic acid concentration (%) |
| 60 |
70 |
80 |
88 |
| Lignin solubilization |
| 70 |
dD |
|
0.3064 |
0.3207 |
0.3473 |
| kL |
|
1.1712 |
1.3201 |
2.8030 |
| 80 |
dD |
|
0.3263 |
0.3584 |
0.4561 |
| kL |
|
2.1264 |
2.8865 |
3.0822 |
| 90 |
dD |
|
0.4823 |
0.6000 |
0.6424 |
| kL |
|
3.2777 |
3.6353 |
4.2339 |
| 100 |
dD |
0.4963 |
0.5799 |
0.6966 |
0.7851 |
| kL |
3.4366 |
4.5324 |
6.3157 |
7.3967 |
| 107 |
dD |
0.5504 |
0.6169 |
0.728 |
0.8222 |
| kL |
4.3787 |
4.7266 |
8.0353 |
9.0889 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
| Xylan solubilization |
| 70 |
dSX |
|
0.1237 |
0.1525 |
0.1727 |
| kX |
|
0.8205 |
1.0935 |
2.6760 |
| 80 |
dSX |
|
0.3681 |
0.4058 |
0.6000 |
| kX |
|
1.0323 |
1.2516 |
4.0036 |
| 90 |
dSX |
|
0.5016 |
0.5958 |
0.7523 |
| kX |
|
1.4723 |
4.4917 |
7.1628 |
| 100 |
dSX |
0.6932 |
0.7195 |
0.8205 |
0.8331 |
| kX |
2.93 |
3.2384 |
8.0024 |
8.4503 |
| 107 |
dSX |
0.7447 |
0.8011 |
0.8708 |
0.8935 |
| kX |
6.6481 |
8.2834 |
13.2358 |
14.7217 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
| Glucan solubilization |
| 70 |
dSG |
|
0.0845 |
0.0989 |
0.1126 |
| kG |
|
1.5057 |
1.7636 |
2.2027 |
| 80 |
dSG |
|
0.1112 |
0.1216 |
0.1227 |
| kG |
|
2.2559 |
2.2793 |
3.1698 |
| 90 |
dSG |
|
0.127 |
0.1419 |
0.1702 |
| kG |
|
3.0062 |
3.1338 |
3.3835 |
| 100 |
dSG |
0.1447 |
0.1747 |
0.2173 |
0.2466 |
| kG |
3.5592 |
3.8556 |
4.1628 |
7.2043 |
| 107 |
dSG |
0.1629 |
0.1897 |
0.2428 |
0.2979 |
| kG |
3.7577 |
4.8262 |
6.7804 |
9.0370 |
An expanded Arrhenius equation can be used to describe the relationship between rate constants and reaction temperature and formic acid concentration as follows:
| |
 | (17) |
where
k0,
Ea and
CFA are pre-exponential factor, activation energy and formic acid concentration (mol l
−1), respectively; and
α is reaction order with respect to formic acid concentration. By taking logarithm,
eqn (17) can be expressed as:
| |
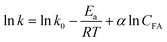 | (18) |
Therefore, by plotting ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k versus 1/T and CFA with multivariate linear regression, corresponding parameters can be fitted. The results are shown in Table 2. dD and dS are the function of reaction severity, which is affected by formic acid concentration and reaction temperature. Therefore, a temperature-dependent severity factor (R0) is introduced to describe the effect of temperature, which is defined as follows:
k versus 1/T and CFA with multivariate linear regression, corresponding parameters can be fitted. The results are shown in Table 2. dD and dS are the function of reaction severity, which is affected by formic acid concentration and reaction temperature. Therefore, a temperature-dependent severity factor (R0) is introduced to describe the effect of temperature, which is defined as follows:
| |
 | (19) |
where
T′ is reaction temperature, in unit of °C. Since 0 ≤
dD ≤ 1, and
dD trends to 1 with the increase of
T′ and
CFA, the relation of
dD with
CFA and
R0 can be correlated by a modified logistic model:
| |
 | (20) |
where
A is an adjustable parameter,
m and
n are corresponding order parameters for
CFA and
R0. Similarly, by taking logarithm on both sides,
eqn (20) can be expressed as:
| |
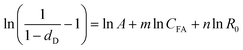 | (21) |
Table 2 Determination of kinetic constants by multivariate linear regression
| k |
k0 |
Ea (J mol−1) |
α |
R2 |
F |
P |
| kL |
3.5518 × 104 |
43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 828 828 |
1.7973 |
0.9480 |
127.6418 |
0.0000 |
| kX |
1.6687 × 106 |
67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 715 715 |
3.1621 |
0.8808 |
51.7126 |
0.0000 |
| kG |
1.2702 × 104 |
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 556 556 |
1.4208 |
0.9329 |
97.3014 |
0.0000 |
| dD and dS |
A |
m |
n |
R2 |
F |
P |
| dD |
9.7566 × 10−4 |
2.5510 |
0.7511 |
0.9416 |
112.8625 |
0.0000 |
| dSX |
0.0017 |
2.5679 |
1.3701 |
0.9545 |
146.8681 |
0.0000 |
| dSG |
0.0041 |
1.3712 |
0.4421 |
0.9579 |
159.0758 |
0.0000 |
Corresponding parameters can be determined by plotting ln[1/(1 − dD)−1] versus CFA and R0 using multivariate linear regression. Similar expression can be used to correlate dSX or dSG with CFA and R0. The results are shown in Table 2, and corresponding comparison of experiment-determined data with model-predicted data are shown in ESI data Fig. S2.† The low P values indicates that the eqn (15) and (18) are significant and reliable to correlate k and dD or dS with reaction temperature and CFA. Therefore, the lignin solubilization ratios of wheat straw at time t can be estimated by the following equation:
| |
 | (22) |
The parameters regressed by similar procedure for xylan and glucan solubilization ratios are shown in Table 2. The xylan and glucan hydrolysis ratios thus can be estimated by the following equations respectively:
| |
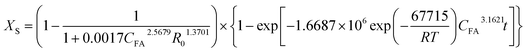 | (23) |
| |
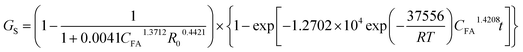 | (24) |
Mechanism analysis for delignification and polysaccharides solubilization
The experimental results demonstrated that the “potential degree of reaction” models could very well describe the kinetics of lignin and polysaccharide solubilization during formic acid pretreatment of wheat straw. It indicated that only a part of the lignin or polysaccharide was reactive under a certain reaction severity, and phenomenologically the reaction rate was first-order with respect to the residual “reactive” fraction. Therefore, when using Saemen's model (the simplest model), which considers that the total lignin or polysaccharides were reactive no matter what reaction severity is used, to describe the kinetics of lignin and polysaccharide solubilization, significant deviation were observed (ESI data Fig. S1†). The heterogeneity in the solubilization of lignocellulose compositions (lignin, hemicelluloses and cellulose) mainly lies on the multilayered structure of cell wall as aforementioned. Such structure results in the different reachability and reactivity of the compositions and diffusion limitation of the reactants and products at different cell wall layer. Therefore, by introducing the parameters termed as “potential degree of reaction”, the solubilization of lignin and polysaccharides can be simply but well described by pseudo-homogenous first-order reaction models.
The activation energy of delignification determined in the present work is 43.8 kJ mol−1, which is in the range of reported data on organic acid delignification.9,13,18 It has been known that ether linkages of various types are over two-thirds (70–75%) of all the intermonomeric linkages of lignin monomers. β-aryl ether linkages are the most frequent ones (48/100 C9), while α-aryl ether linkages account for about 15–20/100 C9. Therefore, cleavages of ether bonds mainly contribute to lignin degradation during organosolv delignification.26 Kinetics on the hydrolysis of lignin model compounds showed that α-aryl ether bonds were hydrolyzed generally 100 times faster than β-aryl ether bonds27 The relatively low activation energy determined in the present work indicated that the breakage of α-aryl ethers bonds probably were mainly responsible for the fragmentation of lignin during formic acid treatment. However, β-aryl ether linkages accounts for a large proportion of ether linkages, and whether the cleavage of β-aryl takes place during formic acid pretreatment should be further investigated.
The kinetic results showed that formic acid had very significant influence on the delignification and polysaccharides solubilization, which were reflected by the high reaction order for formic acid concentration. It also had been found that the reaction order and activation energy of xylan hydrolysis were higher than delignification and cellulose hydrolysis. Thus xylan hydrolysis was more sensitive to the increase of formic acid concentration and temperature. To study the relations of XS and GS versus LS, experimental data obtained under different pretreatmental conditions (temperature, pretreatment time and FA concentration) were plotted in Fig. 5. The plots illustrates that both XS and GS showed non-linear relation with LS. Generally XS slowly increased when LS < 0.3, but quickly increased when LS is between 0.3 and 0.6. When LS > 0.6, increase of XS became slow. GS increased gradually as LS increased. This phenomenon can be explained by the structure of the cell wall. Lignin binds the cells, fibers and vessels, and formic acid pretreatment cut off the internal bonds in lignins as well as lignin–carbohydrate complex. When delignification reaches a certain degree, the hemicellulose hydrolysis was thus accelerated. However, as aforementioned, not all of the hemicellulose has the same reactivity. There is a certain part of hemicelluloses in the deep cell wall layer that are more recalcitrant to solubilize, so XS increased slowly after it reached 0.9. For glucan solubilization, with the removal of the packaging by lignin and hemicelluloses, glucan becomes more accessible for formic acid, H+ and water. Therefore, with the increase of LS, the rate of glucan solubilization increased.
 |
| | Fig. 5 Effect of delignification on xylan and glucan solubilization. | |
Application of the kinetic models
The kinetic models can provide much information for process control. As shown in Fig. 2–4, the solubilizations of lignin, xylan and cellulose increased with formic acid concentration and temperature. Most of lignin and polysaccharide were solubilized in the first hour, and thereafter LS, XS and GS were only slightly increased as the pretreatment proceeded. The general objective of pretreatment is to remove lignin and hemicelluloses but keep remaining cellulose as a solid. To further investigate the effect of formic acid concentration and temperature on lignin, xylan and cellulose solubilization, delignification selectivity (LS/GS) and xylan removal selectivity (XS/GS) were calculated according to eqn (20)–(22). Corresponding 3D surface plots are shown in Fig. 6. LS/GS and XS/GS generally increased with formic acid concentration and temperature. The highest LS/GS can be obtained at high CFA but relatively low temperature. This is mainly because that high CFA accelerates the dissolution of lignin fragments and relatively low temperature reduces the solubilization of cellulose. However, to increase XS/GS the pretreatment should be operated at a low CFA but high temperature. This is mainly because that more formic acid can dissociate to form H+ at low CFA (high water concentration), which can accelerate xylan hydrolysis. The xylan has higher activation energy than cellulose for solubilization, thus increasing temperature is beneficial for removing xylan. Therefore, the pretreatment condition should be selected based on the use of the pretreated solid and the required solubilization ratios of lignin, hemicelluloses and cellulose. For enzymatic hydrolysis, delignification is important to eliminate the physical blocking and non-productive adsorption of enzymes caused by lignin. Thus the pretreatment should be conducted with high formic acid concentration (80–90%) at relatively low temperature such as 90–100 °C to reduce cellulose solubilization. However, more details should be discussed according to the enzymatic digestibility of pretreated solid.
 |
| | Fig. 6 Model-predicted effects of formic acid concentration and temperature on delignification selectivity (LS/GS) (A) and xylan removal selectivity (XS/GS) (B) for 1 hour pretreatment. | |
Since the parameters for “potential degree of reaction” are introduced based on the features of biomass cell wall structure and pretreatment severity, the novel models (eqn (10), (13) and (14)) must be also applicable to other pretreatments and biomass feedstocks. However, the dD, dS and rate constants (k) are varied depending on the pretreatment conditions and the type of biomass used. Our previous work demonstrated that these models were well applicable to dilute acid hydrolysis of sugarcane bagasse,28 acetic acid pretreatment of sugarcane bagasse,18,19 and preparation of cellulose nanocrystal from bleached kraft eucalyptus pulp.29 To further investigate the applicability of the novel models, various reported experimental data from literatures for delignification and polysaccharide solubilization of different biomass feedstocks (including non-woody biomass such as sugarcane bagasse and giant reed, and woody biomass such as olive and eucalyptus) by different pretreatments (including dilute acid pretreatment and organosolv pretreatments) were fitted by eqn (12)–(14) as shown in Fig. 7. The results indicated that the model-calculated data fitted the experimental ones very well. The determination coefficients (R2) were in the range of 0.94–0.999, demonstrating the excellent predictability of the models. These results further prove that “potential degree of reaction” concept can be applicable for both woody and non-woody biomass as well as different pretreatment process to describe the kinetics of delignification and polysaccharide solubilization during chemical pretreatments of lignocellulosic biomass.
 |
| | Fig. 7 Fits of literature reported experimental data by novel “potential degree of reaction model” for delignification and polysaccharide solubilizations of different biomass by different pretreatment methods. (A): dilute acid hydrolysis of olive tree biomass, data from (ref. 30); (B): alkaline ethanol pulping of giant reed, data from (ref. 31); (C): formic acid pulping of sugarcane bagasse, data from (ref. 11); (D): acetic acid pretreatment of Eucalyptus globules, data from (ref. 32). | |
Conclusions
Novel pseudo-homogenous kinetic models with incorporation of parameters termed as “potential degree of reaction” were developed to accurately describe the kinetics of delignification and polysaccharide solubilization during atmospheric formic acid pretreatment of wheat straw. These models were developed based on the multilayered structure of plant cell wall and reaction severity of pretreatment. Due to the complicated structure of cell wall, the mass diffusion limitation of reactants and products increases with the increase of distance from lumen to middle lamella, and corresponding reachability and reactivity of lignin and polysaccharide towards catalyst and solvent decrease. Thus phenomenological maximal degrees of delignification and polysaccharides solubilization are observed under a certain pretreatment severity. The novel kinetic model can excellently fit the experimental data. The relationship between kinetic constants and formic acid concentration and temperature could be well correlated by expanded Arrhenius equations. The parameters for “potential degree of reaction” could be well correlated by modified logistic model. It was also found that the novel models can be used as general models to describe the kinetics of delignification and polysaccharide solubilization for various chemical pretreatments of different biomass feedstocks.
Acknowledgements
Authors are grateful for the supports of this work by National Natural Science Foundation of China (NSFC) (no. 21106081), National Basic Research Program of China (973 Program) (no. 2011CB707406) and National Energy Administration Project (no. NY20130402).
Notes and references
- R. Huang, R. Su, W. Qi and Z. He, BioEnergy Res., 2011, 4, 225 CrossRef.
- M. Lau, B. Bals, S. Chundawat, M. Jin, C. Gunawan, V. Balan, A. Jones and B. Dale, Energy Environ. Sci., 2012, 5, 7100 CAS.
- X. Zhao, L. Zhang and D. Liu, Biofuels, Bioprod. Biorefin., 2012, 6, 465 CrossRef CAS.
- X. Zhao, L. Zhang and D. Liu, Biofuels, Bioprod. Biorefin., 2012, 6, 561 CrossRef CAS.
- X. Zhao, K. Cheng and D. Liu, Appl. Microbiol. Biotechnol., 2009, 82, 815 CrossRef CAS PubMed.
- X. Cui, X. Zhao, J. Zeng, S. Loh, Y. Choo and D. Liu, Bioresour. Technol., 2014, 166, 584 CrossRef CAS PubMed.
- X. Zhao and D. Liu, Bioresour. Technol., 2012, 117, 25 CrossRef CAS PubMed.
- J. F. Saeman, Ind. Eng. Chem., 1945, 37, 43 CrossRef CAS.
- G. Vazquez, G. Antorrena and J. Gonzalez, Wood Sci. Technol., 1994, 28, 403 CAS.
- J. Parajo, J. Alonso and V. Santos, Bioresour. Technol., 1995, 51, 153 CrossRef CAS.
- Q. Tu, S. Fu, H. Zhan, X. Chai and L. Lucia, J. Agric. Food Chem., 2008, 56, 3097 CrossRef CAS PubMed.
- G. Vazquez, G. Antorrena and J. Gonzalez, Wood Sci. Technol., 1995, 30, 31–38 CrossRef CAS.
- G. Vazquez, G. Antorrena, J. Gonzalez, S. Freire and S. Lopez, Bioresour. Technol., 1997, 59, 121 CrossRef CAS.
- A. Shatalov and H. Pereira, Ind. Crops Prod., 2005, 21, 203 CrossRef CAS PubMed.
- W. Zhu, C. J. Houtman, J. Y. Zhu, R. Gleisner and K. F. Chen, Process Biochem., 2012, 47, 785 CrossRef CAS PubMed.
- M. Gilarranz, F. Rodriguez, A. Santos, M. Oliet, F. Garcia-Ochoa and J. Tijero, Ind. Eng. Chem. Res., 1999, 38, 3324 CrossRef CAS.
- M. Oliet, F. Rodriguez, A. Santos, M. Gilarranz, F. Garcia-Ochoa and J. Tijero, Ind. Eng. Chem. Res., 2000, 39, 34 CrossRef CAS.
- X. Zhao and D. Liu, BioEnergy Res., 2013, 6, 436 CrossRef.
- X. Zhao, Y. Morikawa, F. Qi, J. Zeng and D. Liu, Bioresour. Technol., 2014, 151, 128 CrossRef CAS PubMed.
- A. Sluiter, B. Hames, R. Ruiz, C. Scarlata, J. Sluiter, D. Templeton and D. Crocker, Technical Report NREL/TP-510-42618, Laboratory Analytical Procedure: National Renewable Energy Laboratory, 2008, http://www.nrel.gov/biomass/pdfs/42618.pdf Search PubMed.
- Y. Morikawa, X. Zhao and D. Liu, RSC Adv., 2014, 4, 37878 RSC.
- A. J. Panshin and C. de Zeeuw, Textbook of Wood Technology, McGraw-Hill Book Co, New York, 4th edn, 1980, p. 107 Search PubMed.
- E. Muurinen, Ph.D thesis, University of Oulu, Oulu, Finland, 2000.
- I. Wang, M. Kuo and Y. Ku, Bull.–Taiwan For. Res. Inst., 1993, 8, 177 Search PubMed.
- L. Paszner and N. Behera, Holzforschung, 1989, 43, 159 CrossRef CAS.
- T. McDonough, Tappi J., 1993, 76, 186 CAS.
- K. Sama, Ph.D thesis, State University of New York, 1998.
- X. Zhao, Y. Zhou and D. Liu, Bioresour. Technol., 2012, 105, 160 CrossRef CAS PubMed.
- Q. Wang, X. Zhao and J. Y. Zhu, Ind. Eng. Chem. Res., 2014, 53, 11007 CrossRef CAS.
- I. Romero, E. Ruiz, E. Castro and M. Moya, Chem. Eng. Res. Des., 2010, 88, 633 CrossRef CAS PubMed.
- A. Shatalov and H. Pereira, Carbohydr. Polym., 2005, 59, 435 CrossRef CAS PubMed.
- G. Vazquez, G. Antorrena and J. Gonzalez, Wood Sci. Technol., 1995, 29, 267 CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Kinetic modeling of dilute acid hydrolysis for formation of xylose, and simultaneous saccharification and fermentation of dilute acid pretreated solid. See DOI: 10.1039/c4ra14634d |
|
| This journal is © The Royal Society of Chemistry 2015 |
Click here to see how this site uses Cookies. View our privacy policy here. 
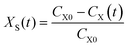

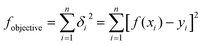
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (L kg−1). The delignification process was assumed to take place in a pseudo-homogeneous system. Since the pretreatment was performed under a mild condition (<110 °C without addition of mineral acid as a catalyst), the condensation of lignin can be negligible in comparison with the solubilization of lignin.10,13 The delignification process thus can be interpreted as the following simple solubilization of lignin from solid phase to liquid phase (Scheme 1):
1 (L kg−1). The delignification process was assumed to take place in a pseudo-homogeneous system. Since the pretreatment was performed under a mild condition (<110 °C without addition of mineral acid as a catalyst), the condensation of lignin can be negligible in comparison with the solubilization of lignin.10,13 The delignification process thus can be interpreted as the following simple solubilization of lignin from solid phase to liquid phase (Scheme 1):
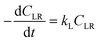

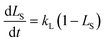
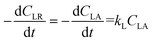

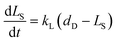
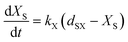

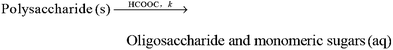
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)

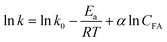
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k versus 1/T and CFA with multivariate linear regression, corresponding parameters can be fitted. The results are shown in Table 2. dD and dS are the function of reaction severity, which is affected by formic acid concentration and reaction temperature. Therefore, a temperature-dependent severity factor (R0) is introduced to describe the effect of temperature, which is defined as follows:
k versus 1/T and CFA with multivariate linear regression, corresponding parameters can be fitted. The results are shown in Table 2. dD and dS are the function of reaction severity, which is affected by formic acid concentration and reaction temperature. Therefore, a temperature-dependent severity factor (R0) is introduced to describe the effect of temperature, which is defined as follows:

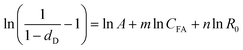
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 828
828![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 715
715![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 556
556