A highly sensitive and selective “turn-on” fluorescent probe for hypochlorous acid monitoring†
Ye-Xin Liao,
Mao-Die Wang,
Kun Li*,
Zhao-Xuan Yang,
Ji-Ting Hou,
Ming-Yu Wu,
Yan-Hong Liu and
Xiao-Qi Yu*
Key Laboratory of Green Chemistry and Technology (Ministry of Education), College of Chemistry, Sichuan University, Chengdu, 610064, P. R. China. E-mail: kli@scu.edu.cn; xqyu@scu.edu.cn
First published on 28th January 2015
Abstract
A novel “turn-on” fluorescent probe 3 for monitoring hypochlorous acid was investigated, which showed high selectivity and sensitivity to hypochlorous acid over other ROS and was loaded into cell media to image HClO.
Reactive oxygen species (ROS, such as HClO and H2O2), play important roles in many biological processes. Excessive ROS conduce oxidative stress (OS), which will cause irreversible damage to living cells or tissues.1 Hypochlorite acid (HClO), or its anionic form the hypochlorite anion (ClO−), generated by the myeloperoxidase (MPO)-mediated reaction between H2O2 and Cl− in vivo,2 is a highly reactive species. HClO/ClO− is an essential microbicidal agent in the immune defence system, such as in leukocytes.3 On the other hand, HClO/ClO− readily reacts with proteins, lipids and nucleic acids by oxidation or chlorination,4 resulting in some related diseases such as inflammatory diseases, cardiovascular disease, neurodegenerative diseases, and cancer.5 However, the exact role that HClO/ClO− plays in biological processes is still not clearly known yet.6 In addition, HClO/ClO− is also used as a bleaching and microbicidal agent in industry or in the healthcare services,7 such as for purifying drinking water and for preparing the famous “84” disinfectant. Excess HClO/ClO− residue in drinking water is harmful to plants, animals and human beings.
With some excellent advantages such as high temporal–spatial resolution, high selectivity and sensitivity,8 fluorescent probes have become outstanding tools in modern analytical technology. Many fluorescent probes for monitoring HClO/ClO− have been designed and developed based on some specific selective reactions, which include oxime oxidation,9 sulfur or selenium oxidation,10 dehydrogenation,11 lactam oxidation,12 electron-rich group (such as anilino group, pyrrole ring and phenyl group) oxidation,13 double bond oxidation14 and others.15 With the unclear mechanism of HClO/ClO− in biological processes and the significance of monitoring HClO/ClO− in industry, a further investigation into more selective and sensitive HClO/ClO− probes is desperately needed.
Electron-rich groups can induce an “enhanced photoinduced electron transfer (PET)” process to a fluorophore and quench its fluorescence, which is a more effective way to quench the fluorescence rather than through the use of single atom electron donors like N, S or Se, resulting in a weaker background, as Peng and co-workers reported.13f As an electron-rich group, the anilino group was demonstrated to be an excellent quencher in “off–on” type fluorescent probes for ROS.16 Thus, the anilino group was introduced to our target probe named 3 (Scheme 1) as a modulator to quench the fluorescence of the fluorophore 1-(benzo[d]thiazol-2-yl)naphthalen-2-ol (compound 1) through an enhanced PET process, giving a non-fluorescent compound. We hoped that HClO/ClO− could react with probe 3 by oxidizing the anilino group to release compound 1, and to manifest a fluorescence “turn-on” phenomenon as a signal conversion. As expected, after reacting with HClO/ClO−, probe 3 was oxidized and compound 1 (Scheme 1) was generated, along with a dramatic fluorescence enhancement.
 | ||
| Scheme 1 The synthetic processes of probe 3 and the reaction between 3 and HClO. (a) Na2S2O5, DMF, 120 °C; (b) 4-chloronitrobenzene, 60% NaH, DMF, 90 °C; (c) H2, Pd/C, MeOH, r.t. overnight. | ||
The synthetic processes of the target probe 3 were shown in Scheme 1. Starting with 2-aminothiophenol and 2-hydroxyl-1-naphthaldhyde, compound 1 was obtained by using Na2S2O5 as a catalyst. After compound 1 reacted with 1-fluoro-4-nitrobenzene, compound 2 was generated, followed by hydrogen reduction catalyzed by Pd/C. Finally, the target probe 3 was synthesized, and was characterized using 1H NMR, 13C NMR and high-resolution mass spectroscopy (HRMS).
In order to determine the optimal detecting condition, we first studied the fluorescence intensity in different solvents. As shown in Fig. S1,† in solvents like H2O, DMSO, DMF, THF, MeCN, MeOH and EtOH, no obvious fluorescence changes were observed upon addition of 5 μM of 3 with 50 μM of HClO/ClO−. On the other hand, considering that cetyltrimethyl ammonium bromide (CTAB, critical micelle concentration 0.9 mM in water17), has been successfully applied in some sensing applications to disperse insoluble compounds in water and to promote the reaction,18 CTAB was introduced in order to disperse probe 3 to form a stable sensing solution. As expected, when 1 mM of CTAB was added, the fluorescence intensity was enhanced 100 fold after 5 μM of 3 reacted with 50 μM of HClO/ClO− in water. Hence, water with CTAB (1 mM) was chosen as the detecting media in the following experiments.
Subsequently, the absorbance and fluorescence properties of 3 before and after dealing with HClO/ClO− were studied. In the absorbance spectrum of 3, there are two side peaks at 295 nm and 335 nm. Besides, 3 exhibits extremely weak fluorescence in DMSO/HEPES buffer (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99, v/v, 1 mM CTAB, pH 7.40, 20 mM HEPES) with a quantum yield of 0.005. After 3 reacted with HClO/ClO−, a wide absorbance peak around 410 nm appeared (see Fig. S2a†). We also found a similar peak in the absorbance spectrum of compound 1. A dramatic enhancement was observed in the fluorescence spectra centred at 465 nm after 3 was mixed with HClO/ClO−, which fitted well with the emission spectrum of 1 when the spectra were normalized (Fig. S2b†), finally yielding a blue emission with a quantum yield of 0.212. In addition, the HPLC results for the reaction between 3 and HClO/ClO− showed that a new peak appeared at 3.83 min, as shown in Fig. 1, while there was a similar peak at the same retention time in the HPLC spectrum of compound 1. Meanwhile, an [M − H]− peak for compound 1 (calculated as 276.0489) was also found at m/z 276.0483 in the high resolution mass spectrum (HRMS) for the reaction between 3 and HClO/ClO− (Fig. S3†). These results indicated that compound 1 was generated during the interaction between 3 and HClO/ClO−.
99, v/v, 1 mM CTAB, pH 7.40, 20 mM HEPES) with a quantum yield of 0.005. After 3 reacted with HClO/ClO−, a wide absorbance peak around 410 nm appeared (see Fig. S2a†). We also found a similar peak in the absorbance spectrum of compound 1. A dramatic enhancement was observed in the fluorescence spectra centred at 465 nm after 3 was mixed with HClO/ClO−, which fitted well with the emission spectrum of 1 when the spectra were normalized (Fig. S2b†), finally yielding a blue emission with a quantum yield of 0.212. In addition, the HPLC results for the reaction between 3 and HClO/ClO− showed that a new peak appeared at 3.83 min, as shown in Fig. 1, while there was a similar peak at the same retention time in the HPLC spectrum of compound 1. Meanwhile, an [M − H]− peak for compound 1 (calculated as 276.0489) was also found at m/z 276.0483 in the high resolution mass spectrum (HRMS) for the reaction between 3 and HClO/ClO− (Fig. S3†). These results indicated that compound 1 was generated during the interaction between 3 and HClO/ClO−.
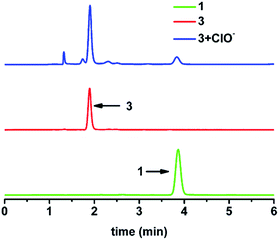 | ||
| Fig. 1 The HPLC results for 1 (green line), 3 (red line) and the reaction between 3 and HClO/ClO− (blue line). | ||
Next, the response of 3 towards HClO/ClO− was investigated at different pH values (Fig. S4†). As can be seen, in the solutions containing probe 3 and HClO/ClO−, almost no fluorescence at 465 nm was found when the pH value was below 5.26. The proposed mechanism suggests that the amino group of probe 3 might be protonated at a low pH value, which would block the oxidation of the amino group by HClO/ClO−. When the pH value increased from 5.26 to 6.99, the fluorescence intensity at 465 nm enhanced gradually, until finally reaching a maximum. Moreover, the effect of the pH value on compound 1 showed that 1 also exhibited relatively weak fluorescence when it was in acidic conditions (pH < 5, Fig. S4†), and we attributed this phenomenon to the pH effect of 1, in some degree. When the pH value was above 7.40, the fluorescence intensity at 465 nm decreased with an increase in the pH value. ClO− should be the dominating species at a high pH value.12g As HClO is a more powerful oxidizing agent than ClO−, we considered that ClO− was not efficient enough to oxidize probe 3. So we suspected that probe 3 reacted with HClO rather than ClO−. Meanwhile, 3 exhibited no fluorescence across the whole pH test range (pH 3–12), as shown in Fig. S4.†
With the determination of these reaction conditions, the spectral responses of 3 in the presence of HClO were then recorded. Absorption spectra of 3 upon the addition of different concentrations of HClO showed that the absorption peak centred at 410 nm arises gradually (Fig. 2a). The fluorescence responses of probe 3 (5 μM) upon titration with HClO in Fig. 2b exhibited that the fluorescence intensity increased to a maximum with the addition of 35 μM of HClO, and was then quenched up until the addition of 50 μM of HClO, when an equilibrium was finally reached. We subsequently set 50 μM as the test concentration of HClO in the following experiments. Additionally, it was shown that the fluorescence intensity of the reaction solution at 465 nm was linearly proportional (R2 = 0.9994, Fig. S5†) to the concentrations of HClO from 0 to 20 μM, and the detection limit was as low as 26.2 nM. Time-dependent fluorescence responses of the reaction of probe 3 and HClO showed that the reaction completed within 15 min (Fig. S6†). Thus, we set 30 min as the reaction time in the following investigations to ensure a completed reaction.
Furthermore, we did an investigation on the selectivity of probe 3 for different ROS (Fig. 3). Among the ROS, 3 only responded to HClO and a slight enhancement with ONOO−. Some evidence shows that a normal concentration of steady-state ONOO− is at a nanomolar level, when HClO is at a micromolar level in physiological conditions. The concentration level of ONOO− wouldn’t be high enough in comparison with the level of HClO.12d Therefore, we consider 3 to be a highly selective probe for HClO over other ROS. Addtionally, some reactive sulfur species (RSS), including GSH, Cys, Hcy and S2−, which exist in an abundant amount in vivo, were incubated with probe 3 at high concentration levels. However, no remarkable fluorescence responses for these test reaction systems were recorded.
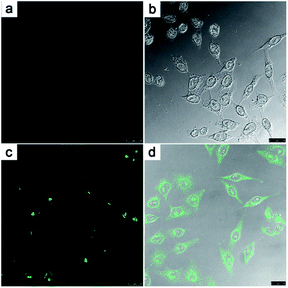
Finally, probe 3 was loaded into HeLa cells to image HClO. As seen in Fig. 4, when HeLa cells were incubated with only probe 3 (with 1 mM CTAB) for 30 min in PBS at 37 °C, no notable fluorescence responses were observed upon excitation at 405 nm (Fig. 4a). After dealing with HClO for another 20 min, the cells pretreated with 3 showed remarkable fluorescence enhancement (Fig. 4c). Herein, we successfully applied probe 3 for HClO imaging in cell media.
In conclusion, we report the novel, easily available, “turn-on” fluorescent probe 3 based on the mechanism of enhanced PET, which exhibited excellent selectivity and a high sensitivity toward HClO with a detection limit of 26.2 nM. Moreover, 3 was successfully applied for the detection of HClO in HeLa cells.
Acknowledgements
This work was financially supported by the National Program on Key Basic Research Project of China (973 Program, 2012CB720603 and 2013CB328900), the National Science Foundation of China (nos 21232005, 21472131, J1310008 and J1103315), and the Specialized Research Fund for the Doctoral Program of Higher Education in China (20120181130006).Notes and references
- C. C. Winterbourn, Nat. Chem. Biol., 2008, 4, 278 CrossRef CAS PubMed.
- (a) S. J. Weiss, R. Klein, A. Slivka and M. Wei, J. Clin. Invest., 1982, 70, 598–607 CrossRef CAS; (b) J. P. Eiserich, M. Hristova, C. E. Cross, A. D. Jones, B. A. Freeman, B. Halliwell and A. van der Vliet, Nature, 1998, 391, 393–397 CrossRef CAS PubMed.
- J. M. Zgliczynski, T. Stelmaszynska, W. Ostrowiski, J. Naskalski and J. Sznajd, Eur. J. Biochem., 1968, 4, 540 CrossRef CAS PubMed.
- (a) P. Nagy and M. T. Ashby, J. Am. Chem. Soc., 2007, 129, 14082–14091 CrossRef CAS PubMed; (b) D. Pattison and M. Davies, Biochemistry, 2006, 45, 8152 CrossRef CAS PubMed; (c) M. P. Curtis, A. J. Hicks and J. W. Neidigh, Chem. Res. Toxicol., 2011, 24, 418–428 CrossRef CAS PubMed.
- (a) H. Ohshima, M. Tatemichi and T. Sawa, Arch. Biochem. Biophys., 2003, 417, 3–11 CrossRef CAS; (b) E. A. Podrez, H. M. Abu-Soud and S. L. Hazen, Free Radical Biol. Med., 2000, 28, 1717–1725 CrossRef CAS; (c) Y. W. Yap, M. Whiteman and N. S. Cheung, Cell. Signalling, 2007, 19, 219–228 CrossRef CAS PubMed; (d) B. S. van der Veen, M. P. J. de Winther and P. Heeringa, Antioxid. Redox Signaling, 2009, 11, 2899 CrossRef CAS PubMed.
- S. A. Weitzman and L. I. Gordon, Blood, 1990, 76, 655 CAS.
- W. A. Rutala and D. J. Weber, Clin. Microbiol. Rev., 1997, 10, 597–610 CAS.
- (a) M. Fernández-Suárez and A. Y. Ting, Nat. Rev. Mol. Cell Biol., 2008, 9, 929–943 CrossRef PubMed; (b) W. Zhang, P. Li, F. Yang, X. F. Hu, C. Z. Sun, W. Zhang, D. Z. Chen and B. Tang, J. Am. Chem. Soc., 2013, 135, 14956–14959 CrossRef CAS PubMed.
- (a) Y. K. Yang, H. J. Cho, J. H. Lee, I. J. Shin and J. S. Tae, Org. Lett., 2009, 11, 859–861 CrossRef CAS PubMed; (b) X. H. Cheng, H. Z. Jia, T. Long, J. Feng, J. G. Qin and Z. Li, Chem. Commun., 2011, 47, 11978–11980 RSC; (c) G. H. Cheng, J. L. Fan, W. Sun, K. Sui, X. Jin, J. Y. Wang and X. J. Peng, Analyst, 2013, 138, 6091–6096 RSC; (d) M. Emrullahoğlu, M. Üçüncü and E. Karakuş, Chem. Commun., 2013, 49, 7836–7838 RSC; (e) S. Y. Yu, C. Y. Hsu, W. C. Chen, L. F. Wei and S. P. Wu, Sens. Actuators, B, 2014, 196, 203–207 CrossRef CAS PubMed.
- (a) X. Q. Chen, K. A. Lee, E. M. Ha, K. M. Lee, Y. Y. Seo, H. K. Choi, H. N. Kim, M. J. Kim, C. S. Cho, S. Y. Lee, W. J. Lee and J. Y. Yoon, Chem. Commun., 2011, 47, 4373–4375 RSC; (b) Y. Koide, Y. Urano, K. Hanaoka, T. Terai and T. Nagano, J. Am. Chem. Soc., 2011, 133, 5680–5682 CrossRef CAS PubMed; (c) Q. A. Best, N. Sattenapally, D. J. Dyer, C. N. Scott and M. E. McCarroll, J. Am. Chem. Soc., 2013, 135, 13365–13370 CrossRef CAS PubMed; (d) S. R. Liu and S. P. Wu, Org. Lett., 2013, 15, 878–881 CrossRef CAS PubMed; (e) Z. R. Lou, P. Li, Q. Pan and K. L. Han, Chem. Commun., 2013, 49, 2445–2447 RSC; (f) A. P. Singh, O. G. Tsay, D. P. Murale, T. H. Jun, H. J. Liew, Y. H. Suhb and D. G. Churchill, Analyst, 2013, 138, 2829–2832 RSC; (g) B. S. Wang, P. Li, F. B. Yu, P. Song, X. F. Sun, S. Q. Yang, Z. R. Lou and K. L. Han, Chem. Commun., 2013, 49, 1014–1016 RSC; (h) X. J. Wu, Z. Li, L. Yang, J. H. Han and S. F. Han, Chem. Sci., 2013, 4, 460–467 RSC; (i) Q. L. Xu, K. A. Lee, S. Y. Lee, K. M. Lee, W. J. Lee and J. Y. Yoon, J. Am. Chem. Soc., 2013, 135, 9944–9949 CrossRef CAS PubMed; (j) G. H. Cheng, J. L. Fan, W. Sun, J. F. Cao, C. Hu and X. J. Peng, Chem. Commun., 2014, 50, 1018–1020 RSC; (k) F. Y. Liu, Y. L. Gao, J. T. Wang and S. G. Sun, Analyst, 2014, 139, 3324–3329 RSC.
- (a) S. Goswami, A. Manna, S. Paul, C. K. Quah and H. K. Fun, Chem. Commun., 2013, 49, 11656–11658 RSC; (b) Z. S. Wu, X. J. Wu, Z. Li, Y. H. Yang, J. H. Han and S. F. Han, Bioorg. Med. Chem. Lett., 2013, 23, 4354–4357 CrossRef CAS PubMed.
- (a) Z. Zhang, Y. Zheng, W. Hang, X. M. Yan and Y. F. Zhao, Talanta, 2011, 85, 779–786 CrossRef CAS PubMed; (b) F. F. Wei, Y. Lu, S. He, L. C. Zhao and X. S. Zeng, Anal. Methods, 2012, 4, 616–618 RSC; (c) L. Yuan, W. Y. Lin, Y. N. Xie, B. Chen and J. Z. Song, Chem.–Eur. J., 2012, 18, 2700–2706 CrossRef CAS PubMed; (d) S. Goswami, S. Das, K. Aich, P. K. Nandi, K. Ghoshal, C. K. Quah, M. Bhattacharyya, H. K. Funcd and H. A. Abdel-Aziz, RSC Adv., 2014, 4, 24881–24886 RSC; (e) J. T. Hou, M. Y. Wu, K. Li, J. Yang, K. K. Yu, Y. M. Xie and X. Q. Yu, Chem. Commun., 2014, 50, 8640–8643 RSC; (f) W. Z. Yin, H. J. Zhua and R. Y. Wang, Dyes Pigm., 2014, 107, 127–132 CrossRef CAS PubMed; (g) Y. R. Zhang, X. P. Chen, J. Shao, J. Y. Zhang, Q. Yuan, J. Y. Miao and B. X. Zhao, Chem. Commun., 2014, 50, 14241–14244 CAS.
- (a) J. Shepherd, S. A. Hilderbrand, P. Waterman, J. W. Heinecke, R. Weissleder and P. Libby, Chem. Biol., 2007, 14, 1221–1231 CrossRef CAS PubMed; (b) Z. N. Sun, F. Q. Liu, Y. Chen, P. K. H. Tam and D. Yang, Org. Lett., 2008, 10, 2171–2174 CrossRef CAS PubMed; (c) J. Y. Kim and Y. M. Kim, Analyst, 2014, 139, 2986–2989 RSC; (d) J. J. Hu, N. K. Wong, Q. S. Gu, X. Y. Bai, S. Ye and D. Yang, Org. Lett., 2014, 16, 3544–3547 CrossRef CAS PubMed; (e) Y. K. Yue, C. X. Yin, F. J. Huo, J. B. Chao and Y. B. Zhang, Sens. Actuators, B, 2014, 202, 551–556 CrossRef CAS PubMed; (f) H. Zhu, J. L. Fan, J. Y. Wang, H. Y. Mu and X. J. Peng, J. Am. Chem. Soc., 2014, 136, 12820–12823 CrossRef CAS PubMed.
- (a) J. S. Park, H. J. Kim, Y. D. Choi and Y. M. Kim, Analyst, 2013, 138, 3368–3371 RSC; (b) Z. R. Lou, P. Li, P. Song and K. L. Han, Analyst, 2013, 138, 6291–6295 RSC; (c) M. T. Sun, H. Yu, H. J. Zhu, F. Ma, S. Zhang, D. J. Huang and S. H. Wang, Anal. Chem., 2014, 86, 671–677 CrossRef CAS PubMed.
- (a) S. M. Chen, J. X. Lu, C. D. Sun and H. M. Ma, Analyst, 2010, 135, 577–582 RSC; (b) X. D. Lou, Y. Zhang, Q. Q. Li, J. G. Qin and Z. Li, Chem. Commun., 2011, 47, 3189–3191 RSC; (c) L. Yuan, W. Y. Lin, J. Z. Song and Y. T. Yang, Chem. Commun., 2011, 47, 12691–12693 RSC; (d) Y. Zhou, J. Y. Li, K. H. Chu, K. Liu, C. Yao and J. Y. Lib, Chem. Commun., 2012, 48, 4677–4679 RSC; (e) J. Zhao, H. J. Li, K. Yang, S. G. Sun, A. P. Lu and Y. Q. Xu, New J. Chem., 2014, 38, 3371–3374 RSC.
- (a) Y. Koide, Y. Urano, S. Kenmoku, H. Kojima and T. Nagano, J. Am. Chem. Soc., 2007, 129, 10324–10325 CrossRef CAS PubMed; (b) Y. Xiao, Z. Ye, G. Wang and J. Yuan, Inorg. Chem., 2012, 51, 2940–2946 CrossRef CAS PubMed; (c) K. Setsukinai, Y. Urano, K. Kakinuma, H. J. Majima and T. Nagano, J. Biol. Chem., 2003, 278, 3170–3175 CrossRef CAS PubMed.
- T. Imae, R. Kamiya and S. Ikeda, J. Colloid Interface Sci., 1985, 108, 215–225 CrossRef CAS.
- (a) G. P. Li, D. J. Zhu, Q. Liu, L. Xue and H. Jiang, Org. Lett., 2013, 15, 924–927 CrossRef CAS PubMed; (b) R. Hu, J. Feng, D. H. Hu, S. Q. Wang, S. Y. Li, Y. Li and G. Q. Yang, Angew. Chem., Int. Ed., 2010, 49, 4915–4918 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: 1H NMR, 13C NMR and HRMS for all of the compounds. See DOI: 10.1039/c4ra14579h |
| This journal is © The Royal Society of Chemistry 2015 |