A dopamine electrochemical sensor based on gold nanoparticles/over-oxidized polypyrrole nanotube composite arrays†
Meng Lin
Key Laboratory for Colloid and Interface Chemistry of State Education Ministry, School of Chemistry and Chemical Engineering, Shandong University, Jinan, 250100, China. E-mail: lixnm@hotmail.com; Fax: +86-0531-88564464; Tel: +86-0531-88364464
First published on 6th January 2015
Abstract
A novel sensor based on a gold nanoparticles (Au NPs)/over-oxidized polypyrrole (OPPy) nanotube (NT) composite arrays electrode proved to be highly effective for electrochemical determination of dopamine (DA). Oriented Au NPs/OPPy NT arrays were electrodeposited by using zinc oxide (ZnO) nanowire (NW) arrays as templates. The morphologies of ZnO NW arrays, OPPy NT arrays, and Au NPs/OPPy NT arrays were studied by scanning electron microscopy (SEM). Electrochemical behaviors of the modified electrodes were characterized by cyclic voltammetry (CV) and square wave voltammetry (SWV) techniques. The Au NPs/OPPy NT arrays electrode had strong catalytic activity toward the oxidation of DA. The peak currents obtained from SWV increased linearly with increasing DA concentrations in the range of 25 nM to 2.5 μM. The simultaneous analysis of ascorbic acid (AA) and DA was investigated, the results demonstrated that the Au NPs/OPPy NT arrays electrodes exhibited good selectivity and sensitivity for the determination of DA without interference by the oxidation of AA.
1. Introduction
Dopamine (DA) is one of the most significant neurotransmitters in biological systems. DA is widely distributed in the brain tissues for message transfer in mammalian central nervous systems.1 Low concentration of DA may lead to neurological disorders, such as Parkinson's disease, Huntington's disease and schizophrenia.2 Hence, the determination of the concentration of DA has received much attention. It is important to develop highly sensitive and selective methods for the analysis of DA.3 Various attempts have been applied to detect DA, such as High Performance Liquid Chromatography (HPLC) method,4 ion chromatography,5 fiuorometric,6 electrochemistry7 and so on. Due to the significant advantages of electrochemical techniques such as inexpensiveness, sensitivity and easy miniaturization, modified electrodes with polypyrrole (PPy),8 polyaniline (PANi),9 graphene,10 nano-porous gold11 etc. have been used in the determination of DA.12PPy nanomaterials have received considerable attention because of environmental stability, good electronic conductivity and good biocompatibility, which originating from their small dimensions and high surface area.13 Attribute to the additional generated electro-catalytic sites between the metal nanoparticles (NPs) dispersed into the polymer and the skeleton of PPy, PPy with entrapped metal NPs have received interest in the electrochemical sensors.14,15 An electrochemical sensor for the determination of DA using Au nanoparticles anchored into the matrix of PPy was reported by Li and Lin,16 but the arrangement of the polymer was out of control. PPy can be over-oxidized in NaOH solution during several sweeps of the cyclic potential. The irreversibly over-oxidized PPy (OPPy) has excellent cation exchanging and molecular sieving properties, thereby imparting superior selectivity and sensitivity toward the neurotransmitter analytes.13 Wen and co-workers developed a OPPy/multi-walled carbon nanotubes modified electrode for sensitively DA, and the electrode showed significantly enhancing oxidative current responses of DA.17
In this paper, we report the fabrication of the Au NPs/OPPy nanotube (NT) arrays composites electrode for the electrochemical determination of DA. Au NPs/OPPy NT arrays were electropolymerized with controlled dimensions and morphologies by using zinc oxide (ZnO) nanowire (NW) arrays as templates. Electrochemical behaviors of the Au NPs/OPPy NT arrays electrodes were investigated using cyclic voltammetry (CV) and square wave voltammetry (SWV) techniques. The electrochemical responses of DA at the Au NPs/OPPy NT arrays electrode enhanced greatly than that at the OPPy NT arrays electrode and ZnO NW arrays electrode. The Au NPs/OPPy NT arrays electrode for electrochemical sensor of DA in the presence of AA was studied. Based on the different electro-catalytic activities of the modified electrode towards DA and ascorbic acid (AA), a sensitive and selective modified electrode for the determination of DA was established.
2. Experimental
2.1 Materials and characterization
Pyrrole (98%), potassium chloride (KCl, 99.5%), zinc chloride (ZnCl2), and methylene chloride (CH2Cl2) were purchased from Aladdin Chemistry Co. Ltd (Shanghai, China). Gold chloride trihydrate (HAuCl4·3H2O) was obtained from Sinopharm Chemical Reagent (Shanghai, China). Tetrabutylammonium hexafluorophosphate, dopamine (DA), and ascorbic acid (AA) were obtained from Sigma-Aldrich. All chemicals were of reagent grade and used without further purification.All solutions were freshly prepared using freshly de-ionized water (18.2 MΩ cm, Pall Corp., USA). Phosphate buffered saline (PBS, 0.10 M) was freshly prepared from NaH2PO4, Na2HPO4, and the pH value was adjusted to 7.0 by using HCl and NaOH solutions. Stock solution of DA (5 mM) was prepared in 0.10 M PBS (pH 7.0) and diluted to give various concentrations of DA.
2.2 Preparation of ZnO NW arrays electrode
ZnO NW arrays were prepared by electrodeposition process according to the previous literature (Tena-Zaera et al., 2008). Prior to the electrodeposition, the indium-tin oxide (ITO) coated glass (1 × 5 cm2) was cleaned in an ultrasonic bath for 20 minutes each in acetone, ethanol, and distilled water, respectively. The electrolyte was a 1 × 10−3 M ZnCl2 and 2 M KCl ultrapure aqueous solution, saturated with bubbling oxygen for 30 minutes. The applied potential was operated at −1 V versus SCE for 90 minutes and the deposition was conducted at a temperature of 80 °C.2.3 Fabrication of Au NPs/OPPy NT arrays electrode
Electrochemical polymerization of PPy was carried out in a methylene chloride solution containing 0.01 M pyrrole and 0.05 M tetrabutylammonium hexafluorophosphate by chronoamperometry at +1.2 V for 90 s. The PPy was electropolymerized onto a ZnO NW arrays working electrode surface positioned parallel to a platinum plate counter electrode, and a saturated calomel electrode (SCE) in saturated KCl solution as a reference electrode. The PPy/ZnO hybrid nanoarrays electrode was rinsed with fresh ethanol and water, respectively, and then immersed in a 0.1 M NaOH solution for over-oxidation process. The over-oxidation of the PPy was performed in an aqueous solution of NaOH by cyclic voltammetry (CV) between 0 and +0.9 V at a scan rate of 50 mV s−1 for 15 cycles. The OPPy/ZnO hybrid nanoarrays electrode was ready for use after washed with de-ionized water.The Au NPs was electrochemically deposited on the OPPy/ZnO hybrid nanoarrays under CV scanning from +0.2 to −1.0 V in 0.2 mM HAuCl4 solution at a scan rate of 50 mV s−1 for 10 cycles. Then the obtained Au NPs/OPPy/ZnO hybrid nanoarrays electrode was taken out and rinsed with de-ionized water.16 Subsequently, the Au NPs/OPPy/ZnO hybrid nanoarrays electrode was immersed in 1 M HCl solution for a few seconds to remove ZnO from the hybrid nanoarrays. The obtained Au NPs/OPPy NT arrays electrode was rinsed with de-ionized water and stored in 0.10 M PBS (pH 7.0) prior to be used.
2.4 Characterization
Surface morphology of the electrodes was examined by scanning electron microscopy (SEM, Hitachi S-4800, Japan). CV of the electrode was conducted in PBS (0.10 M, pH 7.0) at a sweep rate of 100 mV s−1. SWV was carried out using a frequency of 15 Hz, an amplitude of 25 mA, and a step voltage of 4 mV. All electrochemical analysis was performed in a conventional three-electrode cell comprising the modified electrode as a working electrode, a platinum plate as a counter electrode, and a SCE in saturated KCl solution as a reference electrode using a CHI 660C electrochemical workstation (Chenhua Instruments, Shanghai, China).3. Results and discussion
3.1 Surface morphology
SEM images of the electrochemical deposition ZnO NW arrays, OPPy NT arrays and Au NPs/OPPy NT arrays on indium-tin oxide coated (ITO) glasses are shown in Fig. 1, Fig. 1a shows the image of electro-deposited ZnO NW arrays which have a random orientation consisted of the ZnO NWs about from 400 nm in diameter and around 1.5 μm in length.20 Fig. 1b illustrates the morphology of OPPy NT arrays after removing the ZnO arrays template. It can be observed that the free standing top-closed OPPy NT arrays are aligned nearly perpendicular to the substrate. In order to achieve Au NPs/OPPy NT composite arrays, PPy was firstly over-oxidized in NaOH solution by CV. Subsequently, Au NPs were electro-deposited by CV onto the surface of the OPPy nanoarrays. After melting the templates of ZnO NW arrays, the Au NPs/OPPy NT arrays were obtained (Fig. 1c). Obviously, the SEM images reveal that the electrodeposited Au NPs of about 30 nm in diameter were homogeneously distributed on the surface and attached along the OPPy NT arrays.19 The cauliflower surface structure of the Au NPs/OPPy NT arrays could allow a large surface area for electrochemical detection of DA.3.2 Characterization and electrochemical performance of the nanoarrays electrode
Fig. 2 shows the cyclic voltammograms recorded for the ZnO NW arrays (solid line), OPPy NT arrays (dash-solid line) and Au NPs/OPPy NT arrays (dash line) electrodes with 1 μM DA in 0.1 M pH 7.0 PBS, respectively. The CV curve for the OPPy NT arrays shows no obvious electrochemical response between −0.1 and +0.6 V, indicating that DA exhibits a poor electrochemical response on the OPPy NT arrays electrode. Under the identical conditions, a couple of sluggish CV peaks can be observed at the ZnO NW arrays electrode, a few apparent small reduction/oxidation peaks appeared at +0.05 and +0.25 V for DA, this demonstrates that the ZnO NW arrays could prove catalytic activity toward the oxidation of DA.21 The Au NPs/OPPy NT arrays electrode possesses enhanced electro-catalytic oxidation of DA with a shift of 100 mV. Compared to the ZnO NW arrays electrode, the Au NPs/OPPy NT arrays electrode exhibits a lower potential of DA oxidation process. Also, a much higher peak current for the oxidation of DA is noticed at the Au NPs/OPPy NT arrays than at ZnO NW arrays electrode.22 The oxidation peak of DA appeared at +0.15 V on the Au NPs/OPPy NT arrays electrode, and the peak current is four-fold to that of the ZnO NW arrays. These results indicate that the Au NPs/OPPy NT arrays electrode could have excellent electrochemical response to DA. This may be attributed to the larger effective area of Au NPs on the surface of OPPy NT arrays and the static interact between Au NPs and DA molecules.23 Due to the composite effect, the Au NPs/OPPy NT arrays electrode provides a higher catalytic efficiency and remarkable enhancement in the electrochemical detection of DA.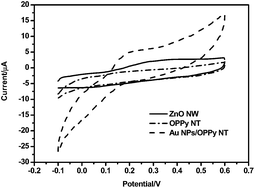 | ||
| Fig. 2 Cyclic voltammograms of 1 μM DA on ZnO NW arrays, OPPy NT arrays and Au NPs/OPPy NT arrays electrodes in 0.1 M PBS (pH = 7.0), the scan rate was 50 mV s−1. | ||
Square wave stripping voltammetry analysis has higher sensitivity than other electrochemical techniques in the determination of trace analytes.18,19 For analytical purpose, SWV was used to determine DA afforded by square wave techniques. We investigated the SWV responses of the Au NPs/OPPy NT arrays electrode between −0.2 and +0.4 V. The SWV peaks and calibration curve originating from the Au NPs/OPPy NT arrays electrode for DA at various concentrations in pH 7.0 PBS are displayed in Fig. 3. SWV for the Au NPs/OPPy NT arrays electrode reveals a distinctive peak at +0.15 V associated with oxidization of DA. The peak current from SWV at the Au NPs/OPPy NT arrays electrode is proportional to the amount of DA in solution. The calibration curve (Fig. 3b) shows a linear increase in current density with the DA concentration from 25 nM to 2.5 μM (R2 = 0.997) with a detection limit of 10 nM (s/n = 3), illustrating a high sensitivity of the Au NPs/OPPy NT arrays electrode to DA.
 | ||
| Fig. 3 (a) SWV curves of DA at Au NPs/OPPy NT arrays electrode in 0.1 M pH 7.0 PBS in the different concentrations of DA, (b) the relation between the peak current and the concentration of DA. | ||
3.3 Selectivity and reproducibility of the Au NPs/OPPy NT array electrodes
AA always influences the electrochemical detection of DA, it is necessary to simultaneously determine DA in the presence of AA.16 As shown in Fig. 4, the electrochemical behaviors of 1 μM DA in the presence of various concentrations of AA at the Au NPs/OPPy NT arrays electrode were investigated by SWV. The SWV responses illustrate that DA yielded a well-defined peak at +0.15 V at the Au NPs/OPPy NT arrays electrode. A slightly SWV peak is observed at −0.02 V while the concentration of AA increased to 10 μM. It can be seen that the peaks current of DA are unaltered with the increase of concentration of AA, suggesting the presence of AA did not interfere with the determination of DA.19 | ||
| Fig. 4 SWV curves of 1 μM DA on the Au NPs/OPPy NT arrays electrode in 0.1 M PBS (pH = 7.0) containing different concentrations of AA (2, 7, 10 μM). | ||
The reproducibility of the Au NPs/OPPy NT array electrode was considered to be excellent as evidenced by a relative standard deviation of 3.6% from five separate measurements. Compared to use the fresh prepared modified electrode, only 2–4% decreases in peak currents indicating that the electrode can be used for repeated electrochemical detection of DA in a reproducible manner. Each electrode was confirmed to be utilized for determination of DA without significant loss of sensitivity for more than 2 weeks.
4. Conclusions
In this present paper, the Au NPs/OPPy NT arrays electrode was synthesized by electrochemical deposition technique. The oriented ZnO NW arrays which were electrodeposited on ITO glass substrates were used as templates. SEM investigation showed that Au NPs had homogeneously attached on the surface the OPPy NT arrays. Due to increasing electronic conductivity and the effective surface area by the composition of Au NPs, the Au NPs/OPPy NT arrays electrode showed efficient electro-catalytic activity thorough CV and SWV towards DA. The electrochemical responses indicated that AA did not have any interference with determination of DA, and can be simultaneously detected. The availability of the OPPy NT arrays combined with metal NPs could be widely used in chemical and biological sensors.Notes and references
- G. J. Yang, J. J. Xu, K. Wang and H. Y. Chen, Electroanalysis, 2006, 18, 282 CrossRef CAS.
- T. Qian, S. Wu and J. Shen, Chem. Commun., 2013, 49, 4610 RSC.
- D. W. M. Arrigan, M. Ghita and V. Beni, Chem. Commun., 2004, 732 RSC.
- S. Sarre, Y. Michotte, P. Herregodts, D. Deleu, N. D. Klippel and G. Ebinger, J. Chromatogr., 1992, 75, 207 CrossRef.
- C. L. Guan, J. Ouyuang, Q. L. Li, B. H. Liu and W. R. G. Baeyens, Talanta, 2000, 50, 1197 CrossRef CAS.
- H. P. Wu, T. L. Cheng and W. L. Tseng, Langmuir, 2007, 23, 7880 CrossRef CAS PubMed.
- J. Breczko, M. E. Plonska-Brzezinska and L. Echegoyen, Electrochim. Acta, 2012, 72, 61 CrossRef CAS PubMed.
- A. J. Zhang, J. Chen, D. F. Niu, G. G. Wallace and J. X. Lu, Synth. Met., 2009, 159, 1542 CrossRef CAS PubMed.
- S. Prakash, C. R. K. Rao and M. Vijayan, Electrochim. Acta, 2009, 54, 5919 CrossRef CAS PubMed.
- Y. Wang, Y. M. Li, L. H. Tang, J. Lu and J. H. Li, Electrochem. Commun., 2009, 11, 889 CrossRef CAS PubMed.
- H. J. Qiu, G. P. Zhou, G. L. Ji, Y. Zhang, X. R. Huang and Y. Ding, Colloids Surf., B, 2009, 69, 105 CrossRef CAS PubMed.
- M. Zheng, Y. Zhou, Y. Chen, Y. Tang and T. Lu, Electrochim. Acta, 2010, 55, 4789 CrossRef CAS PubMed.
- M. Lin, M. S. Cho, W.-S. Choe, J. B. Yoo and Y. Lee, Biosens. Bioelectron., 2010, 26, 940 CrossRef CAS PubMed.
- F. Wang, L. Zhu and J. Zhang, Sens. Actuators, B, 2014, 192, 642 CrossRef CAS PubMed.
- K. Xue, S. Zhou, H. Shi, X. Feng, H. Xin and W. Song, Sens. Actuators, B, 2014, 203, 412 CrossRef CAS PubMed.
- J. Li and X. Lin, Sens. Actuators, B, 2007, 126, 527–535 CrossRef CAS PubMed.
- J. Wen, L. Zhou, L. Jin, X. Cao and B.-C. Ye, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2009, 877, 1793 CrossRef CAS PubMed.
- M. Lin, M. S. Cho, W.-S. Choe and Y. Lee, Biosens. Bioelectron., 2009, 25, 28 CrossRef CAS PubMed.
- M. Lin, M. S. Cho, W.-S. Choe, Y. Son and Y. Lee, Electrochim. Acta, 2009, 54, 7012 CrossRef CAS PubMed.
- R. Tena-Zaera, J. Elias, C. Lévy-Clément, I. Mora-Seró, Y. Luo and J. Bisquert, Phys. Status Solidi A, 2008, 205, 2345 CrossRef CAS.
- C. Xia, N. Wang, L. Wang and L. Guo, Sens. Actuators, B, 2010, 147, 629 CrossRef CAS PubMed.
- A. I. Gopalan, K.-P. Lee, K. M. Manesh, P. Santhosh, J. H. Kim and J. S. Kang, Talanta, 2007, 71, 1774 CrossRef CAS PubMed.
- G.-Z. Hu, D.-P. Zhang, W.-L. Wu and Z.-S. Yang, Colloids Surf., B, 2008, 62, 199 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra14360d |
| This journal is © The Royal Society of Chemistry 2015 |

