Multi stimuli-responsive photoluminescent nanocomposite of silver nanoclusters with hyperbranched polyethylenimine derivatives†
Jia-Li Zhua,
Yi Liub,
Xun-Yong Liub,
Hua-Ji Liu*a and
Yu Chen*a
aDepartment of Chemistry, School of Sciences, Tianjin University, and Collaborative Innovation Center of Chemical Science and Engineering (Tianjin), Tianjin 300072, P. R. China. E-mail: liuhuaji@tju.edu.cn; chenyu@tju.edu.cn
bSchool of Chemistry and Materials Science, Ludong University, Yantai 264025, Shandong, P. R. China
First published on 24th December 2014
Abstract
Silver nanoclusters (AgNCs) functionalized with hyperbranched polyethylenimines with a certain number of trimethylacetamide groups (PEI–TMA) were prepared through three steps. The influence of the preparation conditions, including the pH value in the mixture of PEI–TMA and Ag+ and the Ag+/PEI–TMA feed ratio, on the photoluminescence properties of the obtained nanocomposite of AgNCs and PEI–TMA (AgNC–PEI–TMA) was studied. The obtained AgNC–PEI–TMA nanocomposite was characterized by transmission electron microscopy, dynamic light scattering and zeta potential measurements, verifying the formation of the nanocomposite. AgNC–PEI–TMA in water was not only thermoresponsive, but also responded to other stimuli, including pH, inorganic salts, and loaded organic guest. The cloud point temperature (Tcp) of aqueous solutions of AgNC–PEI–TMA could be modulated through changing the pH, and varying the type and concentration of the inorganic salts and the loaded organic guest. The obtained AgNC–PEI–TMA nanocomposite was photoluminescent, and its maximum emission wavelength was not influenced by outside stimuli. Its emission intensity was influenced negligibly by pH, traditional salting-out anions (Cl− and SO42−), and the relatively polar aspirin guest. However, the traditional salting-in I− anion could quench its fluorescence a little.
Introduction
In recent years ultrasmall fluorescent silver nanoclusters (AgNCs), composed of a few to roughly a hundred atoms, have attracted much interest.1–5 AgNCs possess many attractive features, such as high brightness,6 tunable emission wavelength,7,8 photostability,8,9 two-photon absorption,10 biocompatibility,11 and subnanometer size. These particular characters endow AgNCs enormous potential applications, such as in bioimaging,12–14 biodetection,15 and sensors.16–18During the past decade, polymers with stimulus-responsive properties, such as fast and reversible conformational or phase changes in response to variations in temperature and/or pH, have gained much interest in the fields of biology and medicine.19 One of the most appealing stimuli-responsive species are thermoresponsive hydrophilic polymers with lower critical solution temperatures (LCSTs) in aqueous solution, which means that their solubility in water dramatically decreases above a specific temperature.20 Such thermoresponsive polymers are frequently utilized to endow thermoresponsive properties to luminescent materials. Apart from the vast numbers of thermoresponsive photoluminescent materials, a few materials with dual stimuli-responsive properties have been developed.21–24 Nonetheless, multifunctional photoluminescent materials that respond to more than two stimuli have seldom been prepared.25–27 To date, many kinds of organic scaffolds have been used to stabilize luminescent AgNCs,2 however, the integration of AgNCs with thermoresponsive materials is scarce.28 Only microgel of poly(N-isopropylacrylamide-acrylic acid-2-hydroxyethyl acrylate) particles have been ever used to template the formation of fluorescent AgNCs, resulting in the temperature and pH dual stimuli-responsive composites.28 To the best of our knowledge, AgNCs have never been directly integrated with thermoresponsive polymers. Herein, we report multi stimuli-responsive AgNC nanocomposites integrated with thermoresponsive hyperbranched polymers. Furthermore, such AgNCs are not only photoluminescent, but are also able to interact with guest molecules as nanocarriers. Hence, it is possible for such AgNCs to be used as a drug-delivery system with a self-bioimaging function.
Experimental
Materials
Hyperbranched polyethylenimine (PEI, Aldrich, Mn = 104 g mol−1, Mw/Mn = 2.5) was dried under vacuum prior to use. Trimethylacetic anhydride (99%) was purchased from Alfa Aesar and used without further purification. Benzoylated cellulose tubing (MWCO 1200) was purchased from Sigma and used directly. Triethyl amine (A. R., TEA) was dried over CaH2 and distilled before use. De-ionized water was double-distilled before use. The preparation of PEI terminated with a certain number of trimethylacetamide (TMA) groups (PEI–TMA) is similar to that reported previously through the amidation reaction between PEI and trimethylacetic anhydride (see ESI†).29 Ascorbic acid (A. R.) and AgNO3 (A. R.) were purchased from Tianjin University Kewei Chemical Company and used directly. Aspirin (99%) was purchased from Tianjin Guangfu Fine Chemical Research Institute.Characterization
1H NMR spectra were recorded at 25 °C on a Varian INOVA 500 MHz spectrometer. 2D NOESY 1H NMR spectrum was recorded on a Bruker 500 MHz spectrometer at 278.6 K. Light transmittance of the polymer solution was measured on a temperature-controlled Purkinje General (China) T6 UV/Vis Spectrophotometer at 660 nm, and the heating rate was 0.2 °C/2 min. The cloud-point temperature (Tcp) was taken from the intersection of the maximal slope tangent and the initial horizontal tangent in the resulting transmittance versus temperature curve. The temperature error is ±0.2 °C. Fluorescence-emission spectra were recorded by using a Varian Cary Eclipse photoluminescent spectrometer with a scan rate of 600 nm min−1. The fluorescence quantum yield was determined by a comparative method using quinine sulfate in 0.1 M H2SO4 (quantum yield: 54%) as reference. Transmission electron microscopy (TEM) studies were performed with a JEOL JEM-2100F instrument at a voltage of 200 kV. Dynamic light scattering (DLS) and zeta potential measurements were performed using the Malvern Nano ZS instrument at 25 °C with 633 nm He–Ne laser light, and the light collection at 90°.Preparation of AgNC–PEI–TMA nanocomposite
In a typical assay, 12.5 mmol of PEI–TMA and 2.5 mmol of AgNO3 were dissolved in 10 mL of deionized water and the solution was stirred for 4 h. Subsequently, the pH was adjusted to 5 with acetic acid, and 30 μmol of ascorbic acid was added to the solution before it was stirred continuously for 2 days. All reactions were carried out at room temperature. The nanocomposite was purified by dialysis against water using a cellulose membrane (MWCO, 1200 g mol−1) for 3 days. Finally, the product was dried in a vacuum freezing drier for 24 h.Results and discussion
Multi stimuli-responsive luminescent silver nanocomposites were prepared through three steps (Scheme 1). First, PEI terminated with a certain number of TMA groups was prepared according to our previous work.29 Subsequently, PEI–TMA was mixed with silver ions to form the nano complex. Finally, silver ions were reduced by ascorbic acid to form the AgNCs stabilized by PEI–TMA.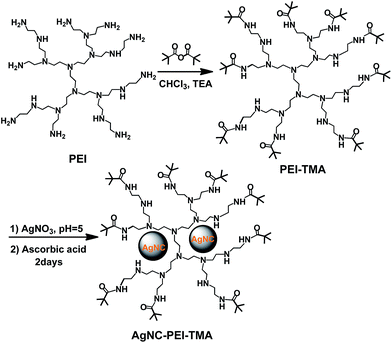 | ||
| Scheme 1 Preparation of AgNCs functionalized with hyperbranched polyethylenimine with a certain number of trimethylacetamide terminals. | ||
Efficient formation of AgNCs would lead to AgNC–PEI–TMA nanocomposites with stronger photoluminescence; therefore, with the aid of fluorescence spectra, we optimized the preparation conditions of AgNC–PEI–TMA. The fluorescence spectra of majority of the formed AgNC–PEI–TMAs revealed an excitation peak centered at around 360 nm and an emission peak centered at around 486 nm (ESI, Fig. S1†). It was found that the pH value of the mixture of PEI–TMA and Ag+ had marked influence on the emission intensity of the final AgNC–PEI–TMA nanocomposites (Fig. 1A). Maximum emission intensity is achieved when the pH value of the mixture is controlled to be 4. The emission intensity decreases gradually with the increase of the pH value. Lowering the pH value a little from 4 to 3.5 leads to an obvious reduction of emission intensity. It should be noted that the nanocomposites formed at pH = 4 or 3.5 were not stable and silver precipitates could be found a few days later. Whereas no silver precipitates could be observed after two months when the nanocomposites were prepared at or above pH = 5. The influence of the pH value of the mixture of PEI–TMA and Ag+ on the emission intensity of the final AgNC–PEI–TMA nanocomposites is tentatively explained as follows: PEI–TMA bears a lot of amine groups. The amine groups can form complexes with Ag+. After Ag+ ions are reduced into AgNCs, amine groups can interact with the surface Ag atoms, resulting that the formed AgNCs are covered and stabilized by PEI–TMA polymers. However, when PEI–TMA shells are in the neutral state or only bear a few of charges, the collision among different AgNC–PEI–TMA nanocomposite entities is severe, leading to the grown-up or agglomeration of the reduced AgNCs. This harms to the fluorescent emission. Increasing the acidity of the media increases the number of positive charges in the PEI–TMA shell through the protonation of the amine groups, which not only makes the nanocomposites soluble well in water, but also reduces the agglomeration chance of AgNCs due to the stronger electrostatic repelling force among different nanocomposites. This favors to the fluorescent emission. At highly acidic conditions most of amines of PEI–TMA polymers are protonated. Such polymers cannot interact well with Ag+ ions and the reduced ones, thus, only a large amount of silver precipitates are formed after the reducing reagents are added. Therefore, suitable pH value of the mixture of PEI–TMA and Ag+ is required for the preparation of stable AgNC nanocomposites with stronger emission intensity.
Fig. 1B shows the influence of molar ratio of Ag+/PEI–TMA on the emission intensity. In all the recipes the feed amount of Ag+ is intact. It is clear that in the Ag+/PEI–TMA range of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 the emission intensities have no big difference and the solution prepared under the condition of 1
5 the emission intensities have no big difference and the solution prepared under the condition of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 of Ag+/PEI–TMA molar ratio shows a little stronger emission. The increase of the Ag+/PEI–TMA ratio above 1
5 of Ag+/PEI–TMA molar ratio shows a little stronger emission. The increase of the Ag+/PEI–TMA ratio above 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 results in a big amount of silver precipitates since there are no enough PEI–TMAs to stabilize the formed AgNCs. The decrease of the Ag+/PEI–TMA ratio below 1
1 results in a big amount of silver precipitates since there are no enough PEI–TMAs to stabilize the formed AgNCs. The decrease of the Ag+/PEI–TMA ratio below 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 leads to the marked reduction of emission intensity. This can be explained as follows: PEI–TMA prepared in this work is thermoresponsive in water. Its phase transition temperature decreases with the increase of polymer concentration (ESI, Fig. S2†). When the Ag+/PEI–TMA ratio is below 1
5 leads to the marked reduction of emission intensity. This can be explained as follows: PEI–TMA prepared in this work is thermoresponsive in water. Its phase transition temperature decreases with the increase of polymer concentration (ESI, Fig. S2†). When the Ag+/PEI–TMA ratio is below 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, the phase transition temperature of AgNC–PEI–TMA is close or below the room temperature, thus some of them aggregate together, weakening the fluorescence emission.
5, the phase transition temperature of AgNC–PEI–TMA is close or below the room temperature, thus some of them aggregate together, weakening the fluorescence emission.
The nanocomposite prepared under the optimized conditions (the pH value of the mixture of PEI–TMA and Ag+ is 5, molar ratio of Ag+/PEI–TMA is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) was characterized by TEM, DLS and zeta potential. Fig. 2A shows the TEM image of the nanocomposite, and many small dark dots can be seen clearly. The average diameter of these small dots is 1.8 ± 0.3 nm, indicating the formation of AgNCs. The DLS curve of the aqueous solution of AgNC–PEI–TMA is mono-modal (Fig. 2B) and the average diameter of the nanocomposite is ca. 5.3 nm, similar to the size of PEI–TMA. In TEM, only the nano-sized AgNCs can be visualized clearly. The larger size observed by DLS implies that in the obtained nanocomposites AgNCs are encapsulated inside PEI–TMAs. Zeta-potential measurement shows that the as-prepared AgNC–PEI–TMA is positively charged at acidic condition (ζ = 8.1 mV at pH = 5) and the ζ value decreases with the pH increase (ζ = 1.7 mV at pH = 8), which further verifies the successful covering of PEI–TMA onto AgNC due to the polycationic nature of the PEI moiety.
5) was characterized by TEM, DLS and zeta potential. Fig. 2A shows the TEM image of the nanocomposite, and many small dark dots can be seen clearly. The average diameter of these small dots is 1.8 ± 0.3 nm, indicating the formation of AgNCs. The DLS curve of the aqueous solution of AgNC–PEI–TMA is mono-modal (Fig. 2B) and the average diameter of the nanocomposite is ca. 5.3 nm, similar to the size of PEI–TMA. In TEM, only the nano-sized AgNCs can be visualized clearly. The larger size observed by DLS implies that in the obtained nanocomposites AgNCs are encapsulated inside PEI–TMAs. Zeta-potential measurement shows that the as-prepared AgNC–PEI–TMA is positively charged at acidic condition (ζ = 8.1 mV at pH = 5) and the ζ value decreases with the pH increase (ζ = 1.7 mV at pH = 8), which further verifies the successful covering of PEI–TMA onto AgNC due to the polycationic nature of the PEI moiety.
Aqueous solution of AgNC–PEI–TMA was yellow and transparent, became turbid after being heated above certain temperature, and became transparent again when it was cooled down (ESI, Fig. S3†). This indicated that the obtained AgNC–PEI–TMA nanocomposite was thermoresponsive. Fig. 3 depicts the temperature dependence of the light transmittance of aqueous solution of AgNC–PEI–TMA at different pH, and an obvious phase transition can be seen when the solution pH is no less than 8. The phase transition temperature, here called the cloud point temperature (Tcp), can be obtained from Fig. 3. It is clear that the thermoresponsive property of the obtained AgNC–PEI–TMA is pH-sensitive. The pH of AgNC–PEI–TMA in water is close to 8.5. Increasing the acidity significantly increases Tcp. Conversely, adjusting the pH to approximately 9 lowers the Tcp. The decrease in Tcp becomes insignificant after further increasing the pH. The pH response of AgNC–PEI–TMA is interpreted as follows: as the acidity of an aqueous solution of AgNC–PEI–TMA increases, more amino groups of AgNC–PEI–TMA are protonated to form more polar quaternary ammonium groups. This significant increase in polarity increases Tcp. In contrast, increasing the basicity gradually turns the more polar quaternary ammonium groups into less polar amines, decreasing Tcp. It should be noted that the thermoresponsive properties of AgNC–PEI–TMA are the same as those of the PEI–TMA precursor.
The salt sensitivity of the thermoresponsive properties of the AgNC–PEI–TMA was measured. Three sodium salts with typical anions, including Cl−, SO42− and I−, were chosen. The Tcp value of AgNC–PEI–TMA can be modulated to different extents by the addition of different inorganic anions (Fig. 4). I− exhibits a salting-in effect that is embodied by an increase in Tcp in certain salt concentration regions, whereas Cl− and SO42− show an obvious salting-out effect that is embodied by a decrease in Tcp. Furthermore, SO42− is more efficient at lowering the Tcp than Cl−. The specific ranking of these three anions for reducing the Tcp in the high salt concentration regions is SO42− > Cl− > I−. This sequence is in accordance with the well-known Hofmeister series for biopolymers and synthetic water-soluble polymers.30–32
 | ||
| Fig. 4 The Tcp values of AgNC–PEI–TMA influenced by different salts: (■) Na2SO4, (▲) NaCl and (□) NaI (the concentration of AgNC–PEI–TMA is 8 mg mL−1). | ||
PEI moieties can accommodate organic molecules with carboxylic acid groups through acid–base neutralization.33,34 Thus, we studied whether AgNC–PEI–TMA could also accommodate organic acid guest, and how the organic guest affected the thermoresponsive properties of the AgNC–PEI–TMA. Aspirin was utilized as the guest model. First, 2D NOESY 1H NMR spectroscopy, which is a powerful technique for investigating the interactions between two different components in close proximity (<0.5 nm), was used to verify the loading of aspirin by AgNC–PEI–TMA (Fig. 5A). The 2D NOESY 1H NMR spectrum of a mixture of AgNC–PEI–TMA and aspirin shows positive cross-peaks between the aromatic ring of the aspirin guest and the PEI–TMA moiety (the methyl groups of the TMA unit and the inner methylene groups of the PEI skeleton) of AgNC–PEI–TMA (Fig. 5A), revealing the formation of a AgNC–PEI–TMA/aspirin complex through the interactions of the AgNC–PEI–TMA moiety with aspirin. Subsequently, the thermoresponsive properties of these complexes were studied. From Fig. 5B, it can be seen that the relatively polar aspirin guest can increase the Tcp, and that increasing the aspirin content in the complex results in a gradual increase in Tcp.
 | ||
| Fig. 5 (A) The typical 2D NOESY 1H NMR spectrum of supramolecular complex of AgNC–PEI–TMA with aspirin and (B) the Tcp values of AgNC–PEI–TMA influenced by aspirin. | ||
The obtained AgNC–PEI–TMA is photoluminescent. When the temperature is lower than Tcp, aqueous solution of the AgNC–PEI–TMA irradiated with 350 nm UV light are illuminated with greenish light, and the solution is still transparent. When the temperature is higher than Tcp, the solution becomes turbid and is still illuminated with greenish light (ESI, Fig. S4†). The detailed photoluminescent characterization shows that only one peak exists in the emission spectra and the emission bands shift to longer wavelengths at higher excitation wavelength (ESI, Fig. S5†). Moreover, the fluorescence quantum yield of AgNC–PEI–TMA in water is measured to be 3.2% (ESI, Fig. S6†).
The effects of outer stimuli, such as pH, salts and organic compound, on the luminescence properties were further studied. It can be seen that these outer stimuli have no influence on the maximum emission wavelength of AgNC–PEI–TMA (ESI, Fig. S7†). Only certain stimuli can influence the emission intensity of AgNC–PEI–TMA (Fig. 6). For instance, the pH value, NaCl, Na2SO4 and aspirin additives have negligible influence on the emission of AgNC–PEI–TMA. Whereas, NaI can quench the luminescence of AgNC–PEI–TMA a little (Fig. 6B), since I− is well-known fluorescence quencher when it is in close proximity to the emitting unit.
Conclusions
Multi stimuli-responsive photoluminescent AgNC–PEI–TMA nanocomposite that responded to temperature, salts, pH, and encapsulated organic guest were successfully prepared. Changing the pH, the type and concentration of salts added, and the concentration of organic guest is an effective way to modulate the Tcp of aqueous solutions of AgNC–PEI–TMA nanocomposites. Low pH, traditional salting-in anions, and polar organic guest increased Tcp significantly, whereas high pH and traditional salting-out anions favored a decrease in Tcp. The maximum emission wavelengths of AgNC–PEI–TMA sample were not influenced by such outer stimuli. Their emission intensities were influenced negligibly by pH in the range of 8–11, traditional salting-out anions, and the relatively polar aspirin guest. However, the traditional salting-in I− could quench the luminescence in a certain extent.The AgNC–PEI–TMA sample integrate multi stimuli-responsive properties, nanocarriers for organic guests, and luminescence properties, thus they have promising applications as “smart” materials in the fields of biomedicine and biotechnology.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (20804027, 21274106 and 21304043).Notes and references
- S. Choi, R. M. Dickson and J. Yu, Chem. Soc. Rev., 2012, 41, 1867–1891 RSC.
- I. Diez and R. H. Ras, Nanoscale, 2011, 3, 1963–1970 RSC.
- L. Shang, S. Dong and G. U. Nienhaus, Nano Today, 2011, 6, 401–418 CrossRef CAS PubMed.
- H. Xu and K. S. Suslick, Adv. Mater., 2010, 22, 1078–1082 CrossRef CAS PubMed.
- Y. Lu and W. Chen, Chem. Soc. Rev., 2012, 41, 3594–3623 RSC.
- J. Sharma, H. C. Yeh, H. Yoo, J. H. Werner and J. S. Martinez, Chem. Commun., 2010, 46, 3280–3282 RSC.
- X. Yuan, Z. T. Luo, Q. B. Zhang, X. H. Zhang, Y. G. Zheng, J. Y. Lee and J. P. Xie, ACS Nano, 2011, 5, 8800–8808 CrossRef CAS PubMed.
- C. I. Richards, S. Choi, J. C. Hsiang, Y. Antoku, T. Vosch, A. Bongiorno, Y. L. Tzeng and R. M. Dickson, J. Am. Chem. Soc., 2008, 130, 5038–5039 CrossRef CAS PubMed.
- S. Choi, J. Yu, S. A. Patel, Y. L. Tzeng and R. M. Dickson, Photochem. Photobiol. Sci., 2011, 10, 109–115 CAS.
- S. A. Patel, C. I. Richards, J. C. Hsiang and R. M. Dickson, J. Am. Chem. Soc., 2008, 130, 11602–11603 CrossRef CAS PubMed.
- L. Shang, R. M. Dörlich, V. Trouillet, M. Bruns and G. Ulrich Nienhaus, Nano Res., 2012, 5, 531–542 CrossRef CAS.
- T. Vosch, Y. Antoku, J.-C. Hsiang, C. I. Richards, J. I. Gonzalez and R. M. Dickson, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 12616–12621 CrossRef CAS PubMed.
- J. Yin, X. He, K. Wang, Z. Qing, X. Wu, H. Shi and X. Yang, Nanoscale, 2012, 4, 110–112 RSC.
- J. Yu, S. A. Patel and R. M. Dickson, Angew. Chem., Int. Ed., 2007, 46, 2028–2030 CrossRef CAS PubMed.
- W. W. Guo, J. P. Yuan, Q. Z. Dong and E. K. Wang, J. Am. Chem. Soc., 2010, 132, 932–934 CrossRef CAS PubMed.
- Z. Yuan, N. Cai, Y. Du, Y. He and E. S. Yeung, Anal. Chem., 2014, 86, 419–426 CrossRef CAS PubMed.
- F. Qu, N. B. Li and H. Q. Luo, Langmuir, 2013, 29, 1199–1205 CrossRef CAS PubMed.
- F. Qu, N. B. Li and H. Q. Luo, Anal. Chem., 2012, 84, 10373–10379 CrossRef CAS PubMed.
- I. Y. Galaev and B. Mattiasson, Trends Biotechnol., 1999, 17, 335–340 CrossRef CAS.
- C. Weber, R. Hoogenboom and U. S. Schubert, Prog. Polym. Sci., 2012, 37, 686–714 CrossRef CAS PubMed.
- Y. Chen and X. Li, Biomacromolecules, 2011, 12, 4367–4372 CrossRef CAS PubMed.
- S. Uchiyama, N. Kawai, A. P. d. Silva and K. Iwai, J. Am. Chem. Soc., 2004, 126, 3032–3033 CrossRef CAS PubMed.
- S. Marpu, Z. Hu and M. A. Omary, Langmuir, 2010, 26, 15523–15531 CrossRef CAS PubMed.
- F. Tang, N. Ma, L. Tong, F. He and L. Li, Langmuir, 2012, 28, 883–888 CrossRef CAS PubMed.
- J. B. Beck and S. J. Rowan, J. Am. Chem. Soc., 2003, 125, 13922–13923 CrossRef CAS PubMed.
- X. Liu, H. J. Liu, F. Cheng and Y. Chen, Nanoscale, 2014, 6, 7453–7460 RSC.
- J.-Y. Yin, H.-J. Liu, S. Jiang, Y. Chen and Y. Yao, ACS Macro Lett., 2013, 2, 1033–1037 CrossRef CAS.
- J. Zhang, S. Xu and E. Kumacheva, Adv. Mater., 2005, 17, 2336–2340 CrossRef CAS.
- H. W. Qin, H. J. Liu and Y. Chen, Chin. J. Polym. Sci., 2014, 32, 1338–1347 CrossRef CAS PubMed.
- W. Kunz, P. L. Nostro and B. W. Ninham, Curr. Opin. Colloid Interface Sci., 2004, 9, 1–18 CrossRef CAS PubMed.
- P. H. von Hippel and T. Schleich, Acc. Chem. Res., 1969, 2, 257–265 CrossRef CAS.
- Y. Zhang, S. Furyk, D. E. Bergbreiter and P. S. Cremer, J. Am. Chem. Soc., 2005, 127, 14505–14510 CrossRef CAS PubMed.
- X.-R. Mu, J.-G. Tong, Y. Liu, X.-Y. Liu, H.-J. Liu and Y. Chen, Polymer, 2013, 54, 2341–2346 CrossRef CAS PubMed.
- J. Zhang, H.-J. Liu, Y. Yuan, S. Jiang, Y. Yao and Y. Chen, ACS Macro Lett., 2013, 2, 67–71 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Typical excitation and emission spectra of AgNC–PEI–TMA; relationship of Tcp and concentration of PEI–TMA; typical photographs of the AgNC–PEI–TMA solution before and after phase transition; typical luminescence photographs, and typical photoluminescence spectra in the presence of additives. See DOI: 10.1039/c4ra14020f |
| This journal is © The Royal Society of Chemistry 2015 |




