Functional polysaccharides from medicinal mushroom Cordyceps sinensis as a potent food supplement: extraction, characterization and therapeutic potentials – a systematic review
Abstract
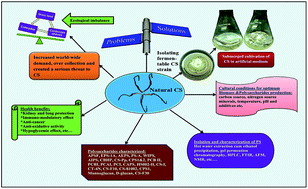
As a rich source of novel polysaccharides, Cordyceps sinensis (CS), one of the valued traditional Chinese medicinal fungi, is a major focus of many natural products research efforts. More than 33 polysaccharides have been characterized till the date. Polysaccharides from CS possess a wide spectrum of biological activities like antitumor, antioxidant, immunomodulatory activity, kidney and lung protection, etc. This review covers the recent literature and updates the information on polysaccharides from CS, covering about 130 research articles emphasizing the isolation, characterization of polysaccharides and their biological functions.

 Please wait while we load your content...
Please wait while we load your content...