Role of ionic liquid [BMIMPF6] in modifying the crystallization kinetics behavior of the polymer electrolyte PEO-LiClO4
S. K. Chaurasiaab,
Shalua,
A. K. Guptaa,
Y. L. Vermaa,
V. K. Singha,
A. K. Tripathia,
A. L. Saroja and
R. K. Singh*a
aDepartment of Physics, Banaras Hindu University, Varanasi-221005, India. E-mail: rksingh_17@rediffmail.com; Fax: +91 542 2368390; Tel: +91 542 2307308
bDepartment of Physics and Astrophysics, University of Delhi, Delhi-110007, India
First published on 15th December 2014
Abstract
We report on the modification in crystallization kinetics behavior of PEO + 10 wt% LiClO4 polymer electrolyte by the addition of an ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate (BMIMPF6). Three techniques have been used for studying crystallization kinetics, viz., (i) isothermal crystallization technique using DSC, (ii) non-isothermal crystallization technique using DSC, and (iii) by monitoring the growth of spherulites with time in the polymer electrolyte films using a polarizing optical microscope (POM). Results from all the three techniques show that the presence of ionic liquid BMIMPF6 suppresses the crystallization rate due to its plasticization effect. Isothermal crystallization data was well described by the Avrami equation, and Avrami exponent n lies in the range of 1 to 2, which signifies 2D crystal growth geometry occurring in these polymer electrolytes under the investigated temperature range. However, the Avrami crystallization rate constant ‘K’ increases exponentially with crystallization temperature and ionic liquid content as well. However, the non-isothermal crystallization kinetics of these polymer electrolytes is discussed in terms of three different models (Jeziorny's, Ozawa's and Mo's method), and it is found that Mo's method better explains the non-isothermal crystallization data. In addition, crystalline morphology and spherulite growth were studied by POM, which shows the suppression in crystallization in the presence of ionic liquid, as confirmed by spherulite growth rate (Gs) analysis.
Introduction
The development of ion-conducting materials (such as polymers, gels, and glasses) has recently attracted global interest in developing a variety of electrochemical devices including rechargeable batteries, fuel cells, supercapacitors, solar cells, and actuators.1–5 A straightforward strategy adopted for this purpose is to integrate mobile ionic species into different solid/polymeric soft matrices. Polymers have advantages over other ion conducting materials because they exhibit various favorable properties such as ease of fabrication in thin film form, and they are mechanically, thermally and electrochemically more stable.6 One of the necessary conditions, in addition to thermal/mechanical/chemical stability, is to make a polymer backbone conducive for providing high ionic mobility for ionic movement. Earlier, many ion conducting polymer matrices or polymer electrolytes were formed by complexing ionic salts (such as Li+, Na+, Mg2+, and Zn2+) with polar polymers (such as PEO, PMMA, PVA, and PVdF) and were much studied, but their room temperature ionic conductivity is very low (∼10−6 to 10−7 S cm−1) because of its high degree of crystallinity, which hinders the motion of the ions in the polymer network.7–11 Therefore, various approaches have been adopted to improve their ionic conductivity, which includes (a) addition of low molecular weight plasticizers/organic carbonates,12,13 such as ethylene carbonate (EC), propylene carbonate (PC), diethyl carbonate (DEC), and (b) use of inorganic fillers14–16 such as SiO2, Al2O3, CNT, TiO2. The use of conventional plasticizers in polymer electrolytes can enhance the ionic conductivity by lowering their glass transition temperature (Tg), which further increases the amorphous phase of the polymer matrix but also cause poor mechanical and thermal stability and a narrower electrochemical potential window due to their volatile nature, which limits their application in devices.17 Recently, a new approach has been adopted to enhance the ionic conductivity of the polymer electrolyte membranes by incorporating ionic liquids (ILs) into the polymer matrix.18–20 ILs have attracted considerable attention as an excellent alternative to the conventional plasticizers due to their distinct properties such as wide liquidus range, non-volatility, non-flammability, negligible vapour pressure, wide electrochemical stability window, high ionic conductivity and excellent thermal and chemical stability.21–23 ILs are entirely composed of bulky and asymmetric organic cations and inorganic anions. Due to the unique properties of ILs, polymer electrolytes based on ionic liquid offer high conductivity along with improved thermal and mechanical properties. In polymer electrolytes, ionic liquid plays the role of a plasticizer as well as a supplier of free charge carriers for ion conduction.24In most of the polymer electrolytes, the amorphous phase is found to be more conducting than the crystalline phase. Accordingly, it is very important to learn about the crystallization kinetics behavior of polymer as well as polymer electrolytes.25–28 Crystallization is a process of phase transformation that involves the transformation of a disordered amorphous phase into a single or multi ordered phase. Polymer crystallization is a complex process that affects the final properties of the materials.29 It has also been observed that some polymers can crystallize, while some cannot. Among the polymers that crystallize, the degree of crystallization, the structure of crystal and crystal size depend upon a number of parameters, such as temperature, time, concentration of solution, and stress present during the crystallization.30 Some studies are available on the modification in crystallization behaviour of polymers on the addition of complexing salts31 or by changing its molecular weight,32 using inorganic fillers,33 carbon nanotubes,34 and also in confined geometries.35 The polymer PEO is known to be a “semicrystalline” polymer that consists of both crystalline and amorphous phases, and its high capability in forming complexes with many salts, as well as its high chemical stability, led to its emergence as a promising host matrix for the preparation of polymer electrolytes. However, PEO tends to crystallize due to its highly ordered chain structure, which impedes the ion transport in the polymeric matrix. This is a general observation for semicrystalline polymer electrolytes and is well documented in literature.36,37 Therefore, it would be interesting to study the crystallization kinetics behavior of such a semicrystalline polymer PEO including its crystal structure and crystalline morphology. The degree of crystallinity is of particular interest for the better understanding of the structure–property relationship to significantly improve the performances of the solid state devices containing PEO-based polymer electrolytes.
This paper reports the crystallization kinetics behavior of the polymer electrolyte (PEO + 10 wt% LiClO4) with different amounts of added ionic liquid (BMIMPF6). Crystallization kinetics was studied by isothermal and non-isothermal crystallization methods using DSC, and the affirmative confirmation of the crystallization behavior was obtained by examining the expansion of spherulites by polarizing optical microscope (POM). It has been found that the addition of BMIMPF6 into PEO + 10 wt% LiClO4 slows the crystallization rate due to the plasticization effect of the ionic liquid BMIMPF6.
Experimental section
Materials and method
The starting materials used for the preparation of polymer electrolyte films are poly(ethylene oxide), PEO of average mol. wt 6 × 105 g mol−1, lithium salt LiClO4 and ionic liquid, 1-butyl-3-methylimidazolium hexafluorophosphate BMIMPF6, obtained from Sigma Aldrich. The ionic liquid BMIMPF6 was vacuum dried at 10−6 torr for 24 h before use.Polymer electrolyte films of PEO + 10 wt% LiClO4 + x wt% BMIMPF6 (for x = 0, 5, 10, 15 and 20) were prepared by a solution casting method. The particular weight ratio of 10 wt% LiClO4 in PEO was chosen for incorporating varying amounts of ionic liquid BMIMPF6 to prepare PEO + 10 wt% LiClO4 +x wt% BMIMPF6 polymer electrolyte films because of its excellent mechanical stability as well as reasonable ionic conductivity compared to other high lithium salt-containing samples. Though the high conducting films containing higher loading of lithium salt LiClO4 could be prepared without BMIMPF6, on loading of ionic liquid BMIMPF6 these films tend to become unstable. In the solution casting method, polymer PEO was dissolved in methanol with stirring at 40 °C and then requisite amount of LiClO4 was added and stirred until it appeared as a homogeneous solution. Subsequently, the required amount of BMIMPF6 was added to the abovementioned solution and stirred again for 2–4 h until a viscous solution was obtained. The viscous solution so obtained was poured into polypropylene Petri dishes. After the complete evaporation of the solvent, PEO + 10 wt% LiClO4 + x wt% BMIMPF6 polymer electrolyte films containing different amounts of BMIMPF6 were obtained. These films were vacuum dried before further use.
Three methods, viz., isothermal, non-isothermal and optical microscopic study, were used for studying the crystallization kinetics. The isothermal and non-isothermal crystallization kinetics of the prepared samples were studied using a differential scanning calorimeter (Mettler Toledo DSC1 system). All the DSC measurements were conducted under a nitrogen atmosphere. The detailed procedures of the isothermal and non-isothermal method are given in their respective sections where results are discussed.
For studying the crystalline morphology and spherulite growth rate, a Lietz DMR polarizing microscope was used. All polarizing optical microscopy (POM) studies were done at a magnification of 50×. For carrying out the POM-studies, the samples were first heated above the melting temperature (Tm) of the polymer electrolyte films and held there for some time until a completely isotropic amorphous phase was observed in POM. Then, the polymer films were quickly quenched to the desired temperature of crystallization (these temperature are less than Tm of the polymer electrolytes). The size of spherulites as a function of time elapsed after their initial appearance was monitored by POM for crystallization study.
Crystallization kinetics by isothermal method
For carrying out the actual experiment, the samples were heated to ∼75 °C (above the melting temperature of polymer Tm) and held there for 5 min to erase the thermal history, and then quickly cooled to the temperature at which crystallization was desired to be studied (note that these temperatures are less than (Tm)onset as determined by DSC thermograms). The samples were maintained at desired crystallization temperatures, viz., Tc = 44, 46, 48 and 50 °C, to crystallize, and DSC exothermic curves for heat flow vs. time were recorded. These DSC isothermal curves obtained for different samples were used for studying the crystallization kinetics by an isothermal method. The heat flow vs. time plot for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films (for x = 0, 10 and 20) are shown in Fig. 1. It can be seen that with increasing crystallization temperature, the crystallization exothermic peak flattens, possibly due to increase in the flexibility of polymer PEO backbone, which suggests that the samples take relatively longer time to crystallize as they approach their (Tm)onset. | ||
| Fig. 1 Heat Flow vs. time plots for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 for (a) x = 0, (b) x = 5 and (c) x = 10 during isothermal crystallization at different Tc. | ||
The crystallization kinetics of polymers under isothermal conditions is described by the well-known Avrami equation38 in terms of the dependence of relative crystallinity (Xt) on the crystallization time (t) as follows:
| Xt = 1 − exp(−Ktn) | (1) |
The relative crystallinity (Xt) generated at any time (t) can be obtained using the DSC exothermic isothermal curves, as illustrated in Fig. 1. The relative crystallinity (Xt), is defined as the ratio of crystallinity generated at any time t to the crystallinity when time approaches infinity. Xt has been evaluated using the following relation:
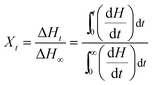 | (2) |
Obviously, the values of Xt at a given crystallization time t can be obtained by integrating the area of exothermic DSC isothermal curves between time t = 0 to t, divided by the entire area of the exothermic peaks.
Using eqn (2), the conversion curves of Xt (the crystallized fraction) versus t for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films (for x = 0, 5 and 10) at various crystallization temperatures (Tc = 44, 46, 48 and 50 °C) are shown in Fig. 2. All the curves show sigmoid characteristics with time and shift towards a higher time regime as the crystallization temperature (Tc) increases. These features indicate that at higher Tc, the crystallization rates become slow and samples take more time to crystallize because the system approaches (Tm)onset. Further, a comparison of the results of polymer electrolyte PEO + 10 wt% LiClO4 (see Fig. 2, curve ‘a’) with those polymer electrolyte films doped with ionic liquid, BMIMPF6 (see Fig. 2, curves b and c) showed that BMIMPF6-containing samples take a longer time to crystallize in comparison to pristine polymer electrolyte due to the increase in amorphicity of the samples.39 For example, we can see that the pristine polymer electrolyte PEO + 10 wt% LiClO4 took nearly 1.8 min to obtain complete crystallization at Tc = 44 °C, and the time required to finish crystallization for 5 wt% BMIMPF6-containing polymer electrolyte slightly increased. It was found to be ∼2 min at the same crystallization temperature, whereas at higher Tc (e.g. 50 °C), it takes ∼15 min to complete crystallization (for detail see Fig. 2c).
 | ||
| Fig. 2 The Xt vs. t plot for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 for (a) x = 0 (b) x = 5 and (c) x = 10 at different Tc, viz., 44, 46, 48, 50 °C. | ||
These observations clearly indicate that with increasing crystallization temperature or ionic liquid concentration, the samples take longer time to crystallize because of the plasticization effect of ionic liquid.40–42 In our previous studies43,44 on the crystallization kinetics of polymer PEO upon the inclusion of ionic liquid as well as lithium salt, it has been shown that the crystallization rate of PEO changes more on addition of BMIMPF6 alone rather than for (PEO + LiClO4) because the plasticization effect of ionic liquid is less experienced by the latter, which has already been amorphosized by the addition of LiClO4. This effect will become clearer while discussing the results of crystallization half time (t1/2) in the subsequent section.
The crystallization half time (t1/2), which is defined as the time necessary to attain 50% of the final crystallinity of the samples, is an important parameter for discussing the crystallization kinetics.45 The values of t1/2 are directly obtained from Fig. 2, from which the rate of crystallization (G = 1/t1/2) can be calculated. The greater the value of t1/2, the lower is the rate of crystallization. Fig. 3 shows the variation of G for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 at various crystallization temperatures (Tc). As illustrated in Fig. 3, the values of G decrease with increasing Tc for all the samples, indicating that the overall crystallization rate becomes slow at higher Tc because the nucleation process is more difficult at higher crystallization temperatures.46 Furthermore, it may be noted here that the values of G (=1/t1/2) obtained for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films are lower in comparison to that of pristine polymer electrolyte PEO + 10 wt% LiClO4 at a given Tc, as shown in Fig. 3, which clearly indicates the slowing of the crystallization rate by the incorporation of BMIMPF6. In addition, the reduction in overall crystallization rate G of polymer electrolytes with increasing BMIMPF6 concentration is attributed to the fact that the presence of ionic liquid BMIMPF6 exerts a dilution effect for the crystallizable polymer PEO due to the plasticization effect of the ionic liquid. Our previous studies47–49 also showed of the similar situation in which the effects of ionic liquid on the properties of polymers and polymer electrolytes were studied and it was found that the ionic liquid acts as an efficient plasticizer.
Avrami plots (i.e., double logarithmic graphic representations of crystallization data) are generally used to calculate the values of n and K. They can be obtained by taking the double logarithmic of eqn (1) and can be written as follows:
log[−ln(1 − Xt)] = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K + n K + n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t t
| (3) |
According to eqn (3), the plot of log[−ln(1 − Xt)] vs. log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t should be a straight line. In the present study, the Avrami plots for PEO + 10 wt% LiClO4 films containing different amounts of BMIMPF6 give rise to a series of parallel straight lines at different crystallization temperatures (viz., Tc = 44, 46, 48 and 50 °C), as shown in Fig. 4. After linear fitting of these plots by a straight line at different Tc, both the Avrami exponent ‘n’ and crystallization rate constant ‘K’ can be obtained by the slope and intercept of the straight line, respectively. The various Avrami parameters obtained from isothermal crystallization method for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 at various crystallization temperatures are given in Table 1.
t should be a straight line. In the present study, the Avrami plots for PEO + 10 wt% LiClO4 films containing different amounts of BMIMPF6 give rise to a series of parallel straight lines at different crystallization temperatures (viz., Tc = 44, 46, 48 and 50 °C), as shown in Fig. 4. After linear fitting of these plots by a straight line at different Tc, both the Avrami exponent ‘n’ and crystallization rate constant ‘K’ can be obtained by the slope and intercept of the straight line, respectively. The various Avrami parameters obtained from isothermal crystallization method for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 at various crystallization temperatures are given in Table 1.
| PEO + 10 wt% LiClO4 + x wt% BMIMPF6 | Tc (°C) | n | K (min−n) | t1/2 (min) |
|---|---|---|---|---|
| (a) x = 0 | 44 | 1.48 | 0.70 | 0.94 |
| 46 | 1.40 | 0.39 | 1.45 | |
| 48 | 1.65 | 0.07 | 3.85 | |
| 50 | 1.86 | 0.02 | 3.95 | |
| (b) x = 5 | 44 | 1.55 | 0.66 | 1.02 |
| 46 | 1.53 | 0.40 | 1.35 | |
| 48 | 1.63 | 0.10 | 4.09 | |
| 50 | 1.76 | 0.06 | 7.09 | |
| (c) x = 10 | 44 | 1.34 | 0.33 | 1.57 |
| 46 | 1.31 | 0.11 | 3.49 | |
| 48 | 1.70 | 0.02 | 10.69 | |
| 50 | 1.74 | 0.01 | 15.08 |
It is known that the values of Avrami exponent are the consequences of the geometry of specific crystal dimension and have been used to specify the dimension of growing crystals. The value of Avrami exponent n is assumed to lie in the range between 1 and 4 and is related to the geometry characteristics of the growing crystals: n = 1 is ascribed to the 1D structure, 2 is ascribed to the 2D structure, and 3 or 4 ascribed to the 3D structure.50 In the present study, the values of ‘n’ for all the polymer electrolyte PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films lie between 1 and 2 at all crystallization temperatures studied. This indicates that the 2D crystal growth morphology dominates at all temperatures at which crystallization has been studied.
The effect of the incorporation of ionic liquid into the polymer membrane, apart from decreasing the rate of crystallization, is also expected to reflect the values of activation energy for crystallization. To check this, we determined this activation energy for PEO + 10 wt% LiClO4 polymer electrolyte with BMIMPF6. The Avrami crystallization rate constant K can be assumed to be a consequence of a thermally activated process and has been used to determine the activation energy for crystallization.51 The crystallization rate constant K can be expressed by the Arrhenius equation as follows:
K1/n = Ko![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−ΔE/RTc) exp(−ΔE/RTc)
| (4) |
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K1/n = ln K1/n = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ko − ΔE/RTc Ko − ΔE/RTc
| (5) |
By plotting the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K1/n versus 1/Tc, ΔE can be obtained by the slope of these curves. Typical plots for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 are shown in Fig. 5. The slope of this curve gives the value of ΔE/R, from which the isothermal crystallization activation energy ΔE can be directly calculated. The value of isothermal crystallization activation energy ΔE for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 polymer electrolyte was found to be 97 kJ mol−1.
K1/n versus 1/Tc, ΔE can be obtained by the slope of these curves. Typical plots for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 are shown in Fig. 5. The slope of this curve gives the value of ΔE/R, from which the isothermal crystallization activation energy ΔE can be directly calculated. The value of isothermal crystallization activation energy ΔE for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 polymer electrolyte was found to be 97 kJ mol−1.
 | ||
Fig. 5 ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K1/n vs. 1/Tc plot for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 during isothermal crystallization. K1/n vs. 1/Tc plot for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 during isothermal crystallization. | ||
Crystallization kinetics by a non-isothermal method
Non-isothermal crystallization method using DSC is also one of the most convenient methods for studying the crystallization kinetic behavior of polymers. In this method, samples were heated above the melting temperature of the polymer PEO (∼75 °C), kept there for 5 min to erase the thermal history, and then cooled at different cooling rates (viz., 5, 10, 15 and 20 °C min−1). DSC exothermic curves for heat flow versus temperature were recorded to analyze the non-isothermal crystallization data. Samples used for non-isothermal crystallization were the same for which isothermal crystallization kinetic studies were carried out. The DSC exothermal curves for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films at different cooling rates 5, 10, 15 and 20 °C min−1 are shown in Fig. 6. It can be seen that the exothermic crystallization peaks were shifted to lower temperature side, which became broader with increasing cooling rates. To find the non-isothermal crystallization kinetics, relative crystallinity (Xt) versus temperature plots for these polymer electrolyte films were obtained using eqn (2) and are shown in Fig. 7 (the procedure is similar to that used for the isothermal crystallization method described earlier). Fig. 7 shows the plots of Xt versus temperature (T), which illustrate an anti-S-shaped characteristic. In non-isothermal crystallization process, temperature scale of Xt versus T plots could be converted into Xt versus t using the following relation:52
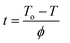 | (6) |
Several approaches have been used for describing the crystallization process involved in the non-isothermal crystallization kinetics, which are based on various models, including the modified Avrami equation by Jeziorny,53 Ozawa analysis54 and Mo's method.55 These approaches are discussed in the subsequent sections.
| Xt = 1 − exp(−Zttn′) | (7) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t. The log[−ln(1 − Xt)] vs. log
t. The log[−ln(1 − Xt)] vs. log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t plots for PEO +10 wt% LiClO4 + x wt% BMIMPF6 (for x = 0, 5, 10, 15 and 20) at different cooling rates (viz., 5, 10, 15 and 20 °C min−1) are shown in Fig. 9. From these plots, we can see that at initial stages, these curves are fitted well by eqn (7) but deviate from the linear relation on higher crystallization. Therefore, the Avrami equation cannot be accurately used for describing the entire non-isothermal crystallization kinetics process.
t plots for PEO +10 wt% LiClO4 + x wt% BMIMPF6 (for x = 0, 5, 10, 15 and 20) at different cooling rates (viz., 5, 10, 15 and 20 °C min−1) are shown in Fig. 9. From these plots, we can see that at initial stages, these curves are fitted well by eqn (7) but deviate from the linear relation on higher crystallization. Therefore, the Avrami equation cannot be accurately used for describing the entire non-isothermal crystallization kinetics process.
 | ||
| Fig. 9 Avrami plots using non-isothermal method of PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films for (a) x = 0 (b) x = 5 (c) x = 10 (d) x = 15 and (e) x = 20 at different cooling rates. | ||
Jeziorny53 modified the Avrami equation for non-isothermal crystallization processes; it assumes that curves with a fixed cooling rate represent a series of isothermal crystallization processes, and it gave a modified non-isothermal crystallization rate (Zc) in which the effect of cooling rate ‘ϕ’ on the value of Zt was included as follows:
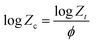 | (8) |
The values of the Avrami constants n′ and Zc obtained by the modified Avrami equation in a non-isothermal crystallization method are given in Table 2. It may be noted here that the values of n′ and Zc obtained in non-isothermal crystallization do not have the same magnitude as in the isothermal method due to different experimental conditions used in these two methods.
| PEO + 10 wt% LiClO4 + x wt% BMIMPF6 | Cooling rate ϕ (°C min−1) | n′ | Zt (min−n′) | Zc | t′1/2 (min) |
|---|---|---|---|---|---|
| (a) x = 0 | 5 | 2.29 | 0.139 | 0.674 | 1.96 |
| 10 | 2.27 | 0.516 | 0.936 | 1.13 | |
| 15 | 2.01 | 0.651 | 0.971 | 1.02 | |
| 20 | 2.05 | 0.717 | 0.986 | 0.88 | |
| (b) x = 5 | 5 | 2.539 | 0.096 | 0.626 | 2.16 |
| 10 | 2.225 | 0.067 | 0.961 | 1.04 | |
| 15 | 2.215 | 0.839 | 0.988 | 1.14 | |
| 20 | 1.742 | 0.900 | 0.994 | 0.85 | |
| (c) x = 10 | 5 | 2.260 | 0.138 | 0.673 | 1.96 |
| 10 | 2.242 | 0.516 | 0.936 | 1.13 | |
| 15 | 2.017 | 0.662 | 0.972 | 1.00 | |
| 20 | 1.893 | 0.751 | 0.985 | 0.91 | |
| (d) x = 15 | 5 | 2.374 | 0.106 | 0.638 | 2.17 |
| 10 | 2.107 | 0.311 | 0.889 | 1.42 | |
| 15 | 1.387 | 0.439 | 0.946 | 1.07 | |
| 20 | 1.642 | 0.608 | 0.975 | 0.84 | |
| (e) x = 20 | 5 | 2.199 | 0.095 | 0.625 | 2.47 |
| 10 | 1.913 | 0.289 | 0.883 | 1.57 | |
| 15 | 1.844 | 0.398 | 0.940 | 1.38 | |
| 20 | 1.503 | 0.599 | 0974 | 1.13 |
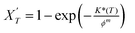 | (9) |
log[−ln(1 − X′T)] = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K*(T) − m K*(T) − m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ϕ ϕ
| (10) |
If the Ozawa theory is fully applicable for describing the non-isothermal crystallization process, then the plot of log[−ln(1 − X′T)] vs. log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ϕ should be a straight line, which was not the case for our samples, as shown in Fig. 10. Therefore, the Ozawa equation is not be fully applicable for describing the non-isothermal crystallization process of PEO + 10 wt% LiClO4 polymer electrolytes as well as PEO + 10 wt% LiClO4 + x wt% BMIMPF6 containing different amounts of BMIMPF6.
ϕ should be a straight line, which was not the case for our samples, as shown in Fig. 10. Therefore, the Ozawa equation is not be fully applicable for describing the non-isothermal crystallization process of PEO + 10 wt% LiClO4 polymer electrolytes as well as PEO + 10 wt% LiClO4 + x wt% BMIMPF6 containing different amounts of BMIMPF6.
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ϕ = log ϕ = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F(T) − b F(T) − b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t t
| (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F(T) = [K*(T)/Zt]1/m and b is the ratio between the Avrami and Ozawa exponents n and m. Here, the function F(T) refers to the value of cooling rate required to reach a defined degree of crystallinity at a certain temperature in unit crystallization time.58 The higher value of F(T) gives lower crystallization rate. The plot between log
F(T) = [K*(T)/Zt]1/m and b is the ratio between the Avrami and Ozawa exponents n and m. Here, the function F(T) refers to the value of cooling rate required to reach a defined degree of crystallinity at a certain temperature in unit crystallization time.58 The higher value of F(T) gives lower crystallization rate. The plot between log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ϕ and log
ϕ and log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films at different cooling rates and various crystallinity gives a straight line as shown in Fig. 11. The values of log
t for PEO + 10 wt% LiClO4 + x wt% BMIMPF6 films at different cooling rates and various crystallinity gives a straight line as shown in Fig. 11. The values of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F(T) and b can be obtained for the samples for different values of ‘x’ from the intercept and slope, respectively. These values are listed in Table 3. From this, we can see that for a given degree of crystallinity (e.g., 10%), the value of log
F(T) and b can be obtained for the samples for different values of ‘x’ from the intercept and slope, respectively. These values are listed in Table 3. From this, we can see that for a given degree of crystallinity (e.g., 10%), the value of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F(T) for PEO + 10 wt% LiClO4 polymer electrolyte is 0.607, which increases to 0.629, 0.634, 0.688 and 0.714, respectively, for the polymer electrolyte containing 5, 10, 15 and 20 wt% BMIMPF6. In general, the value of F(T) for all the polymeric films is found to increase with the increasing amount of BMIMPF6 in PEO + 10 wt% LiClO4. The abovementioned observation clearly indicates that the incorporation of ionic liquid into PEO + 10 wt% LiClO4 polymer electrolyte decreases the crystallization rate due to the plasticization effect of an ionic liquid.59–61 The addition of ionic liquid BMIMPF6 into PEO + LiClO4 polymer electrolyte hinders the crystallization of polymer and leads to slowing of the crystal growth rate, as confirmed by the morphological studies.
F(T) for PEO + 10 wt% LiClO4 polymer electrolyte is 0.607, which increases to 0.629, 0.634, 0.688 and 0.714, respectively, for the polymer electrolyte containing 5, 10, 15 and 20 wt% BMIMPF6. In general, the value of F(T) for all the polymeric films is found to increase with the increasing amount of BMIMPF6 in PEO + 10 wt% LiClO4. The abovementioned observation clearly indicates that the incorporation of ionic liquid into PEO + 10 wt% LiClO4 polymer electrolyte decreases the crystallization rate due to the plasticization effect of an ionic liquid.59–61 The addition of ionic liquid BMIMPF6 into PEO + LiClO4 polymer electrolyte hinders the crystallization of polymer and leads to slowing of the crystal growth rate, as confirmed by the morphological studies.
| PEO + 10 wt% LiClO4 + x wt% BMIMPF6 | X′T in % | F(T) | b |
|---|---|---|---|
| (a) x = 0 | 10 | 0.607 | 1.563 |
| 20 | 0.840 | 1.579 | |
| 30 | 0.983 | 1.662 | |
| 40 | 1.086 | 1.677 | |
| 50 | 1.171 | 1.671 | |
| 60 | 1.311 | 1.784 | |
| 70 | 1.504 | 1.504 | |
| 80 | 1.727 | 1.786 | |
| (b) x = 5 | 10 | 0.629 | 1.263 |
| 20 | 0.864 | 1.180 | |
| 30 | 0.957 | 1.245 | |
| 40 | 1.059 | 1.356 | |
| 50 | 1.172 | 1.172 | |
| 60 | 1.331 | 1.331 | |
| 70 | 1.570 | 1.570 | |
| 80 | 1.753 | 1.583 | |
| (c) x = 10 | 10 | 0.634 | 1.485 |
| 20 | 0.842 | 1.573 | |
| 30 | 0.973 | 1.635 | |
| 40 | 1.086 | 1.695 | |
| 50 | 1.204 | 1.774 | |
| 60 | 1.343 | 1.850 | |
| 70 | 1.529 | 1.529 | |
| 80 | 1.713 | 1.713 | |
| (d) x = 15 | 10 | 0.688 | 1.420 |
| 20 | 0.932 | 1.594 | |
| 30 | 1.078 | 1.714 | |
| 40 | 1.208 | 1.815 | |
| 50 | 1.334 | 1.936 | |
| 60 | 1.485 | 2.060 | |
| 70 | 1.672 | 2.149 | |
| 80 | 1.820 | 1.997 | |
| (d) x = 20 | 10 | 0.714 | 1.075 |
| 20 | 0.952 | 1.267 | |
| 30 | 1.113 | 1.473 | |
| 40 | 1.254 | 1.635 | |
| 50 | 1.397 | 1.818 | |
| 60 | 1.546 | 1.967 | |
| 70 | 1.722 | 2.121 | |
| 80 | 1.866 | 2.126 |
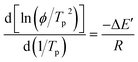 | (12) |
Crystalline morphology and spherulite growth by polarized optical microscopy
When polymers are crystallized from the melt, the most commonly observed structures are the spherulites. These spherulites are in the form of spherical aggregates of lamellae, characteristics of polymers crystallized isothermally in the absence of pronounced stress or flow.63–65 The typical dimensions of spherulites are of the order of microns, and sometimes even millimeters. Therefore, they can be easily viewed under a polarizing optical microscope. The spherulites continue to grow radially until they impinge upon one another. A measure of their growth until the time of impingement provides abundant information regarding the mechanism of crystallization in the polymer, and has been the focus of several investigations.66–68The number and size of spherulites growing in the polymer matrix controls the overall crystallinity of the polymer. Therefore, measuring the size of spherulites as a function of time can provide an estimate of the rate of crystallization. We used this technique for measuring the rate of crystallization of our polymer electrolyte membranes. The size of spherulites was measured by a polarizing optical microscope (POM). Typical growth of such spherulites appearing in PEO + 10 wt% LiClO4 and PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 polymer electrolyte films at different times, crystallized at 50 °C, are shown in Fig. 13. We can see that spherulite growth was very rapid for PEO + 10 wt% LiClO4 polymer electrolyte: within 10 seconds it acquired ∼100 μm diameter, and then its size increased to 310 μm at the elapsed time of 120 s, as shown in Fig. 13 curve a (i) and (v). As BMIMPF6 was added to PEO +10 wt% LiClO4 polymer electrolyte, the spherulite size was reduced and the number of nucleating sites increased. This is evident when we compare Fig. 13 curves a (i–v) with b (i–v).
In the present case, we can also see that the size of one particular spherulite at any particular snap increased with time, as indicated in rectangle of Fig. 13. Therefore, it is important to determine the spherulitic growth rate (and therefore crystallization rate) for deciding the effect of BMIMPF6 on the crystallization behavior of polymer electrolytes with and without ionic liquid. The spherulite size vs. time plot for PEO + 10 wt% LiClO4 polymer electrolyte and with added 10 wt% BMIMPF6 is shown in Fig. 14, whose slope gives the spherulites growth rate (Gs). The value of Gs for pure PEO + 10 wt% LiClO4 is found to be 1.77 μm s−1, which decreases to 0.675 μm s−1 for PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6. The abovementioned observation clearly indicates that the incorporation of BMIMPF6 in PEO +10 wt% LiClO4 polymer electrolyte hinders the spherulites growth rate of polymer PEO. This supports our previous results obtained from isothermal and non-isothermal DSC technique, which indicate the suppression in the crystallization rate of polymer PEO due to the incorporation of an ionic liquid (BMIMPF6) owing to its plasticization effect, as discussed earlier in this paper.
 | ||
| Fig. 14 Spherulite diameter vs. crystallization time plot for (a) PEO + 10 wt% LiClO4 (b) PEO + 10 wt% LiClO4 + 10 wt% BMIMPF6 films crystallized at 50 °C at different crystallization times. | ||
Conclusions
The crystallization kinetics behavior of PEO + 10 wt% LiClO4 + x wt% BMIMPF6 polymer electrolytes has been studied using three techniques, viz., isothermal and non-isothermal crystallization methods using DSC as well as by monitoring spherulite growth using a polarizing optical microscope. The well-known Avrami equation has been used to describe the isothermal crystallization process. The obtained crystallization parameters for isothermal crystallization, such as Avrami exponent (n), crystallization rate constant (K), crystallization half time (t1/2), crystallization rate (G) and isothermal crystallization activation energy (ΔE) suggested that the incorporation of the ionic liquid BMIMPF6 into PEO + 10 wt% LiClO4 polymer electrolyte hinders the crystallization of polymer PEO due to the plasticization effect. The value of Avrami exponent ‘n’ obtained for the isothermal crystallization process of PEO + 10 wt% LiClO4 + x wt% BMIMPF6 (for x = 0, 5 and 10) was found to be less than 2, indicating the 2D crystal growth geometry. It was also found that the non-isothermal crystallization data cannot be fully described by the modified Avrami equation (Jeziorny method) and Ozawa analysis. The method proposed by Mo and coworkers was employed to accurately analyze the non-isothermal crystallization process. The POM study shows that the incorporation of BMIMPF6 in PEO + 10 wt% LiClO4 polymer electrolyte slows down the spherulite growth rate, as observed by spherulite size vs. time plots. All the three techniques used by us to study the effect of ionic liquid BMIMPF6 on the crystallization behavior of polymer electrolytes have led to the conclusion that the presence of BMIMPF6 suppresses the crystallization rate. This will have an effect in decreasing the absolute crystallinity of PEO with increasing BMIMPF6 content.Acknowledgements
One of us (RKS) is grateful to BRNS-DAE Mumbai and DST New Delhi, India, for financial assistance for carrying out this work and SKC is thankful to UGC New Delhi, India, for providing the Dr D. S. Kothari Postdoctoral research fellowship.References
- J. R. MacCallum and C. A. Vincent, Polymer Electrolyte Reviews, Elsevier Applied Science, London, 1987, vol. 1 & 2 Search PubMed
.
- W. H. Meyer, Adv. Mater., 1998, 10, 439 CrossRef CAS
.
- Y. J. Wang, J. Qiao, R. Baker and J. Zhang, Chem. Soc. Rev., 2013, 42, 5768 RSC
.
- J. N. de Freitas, A. F. Nogueira and M. A. De Paoli, J. Mater. Chem., 2009, 19, 5279 RSC
.
- F. M. Gray, Polymer Electrolytes: Fundamental and Technological Applications, VCH Publishers, New York, 1997 Search PubMed
.
- W. Wieczorek, D. Raducha, A. Zalewska and J. R. Stevens, J. Phys. Chem. B, 1998, 102, 8725 CrossRef CAS
.
- A. Karmakar and A. Ghosh, J. Appl. Phys., 2010, 107, 104113 CrossRef PubMed
.
- E. Quartarone and P. Mustarelli, Chem. Soc. Rev., 2011, 40, 2525 RSC
.
- S. Seki, K. Takei, H. Miyashiro and M. Watanabe, J. Electrochem. Soc., 2011, 158, A769 CrossRef CAS PubMed
.
- J. J. Xu, H. Ye and J. Huang, Electrochem. Commun., 2005, 7, 1309 CrossRef CAS PubMed
.
- M. D. Slater, D. Kim, E. Lee and C. S. Johnson, Adv. Funct. Mater., 2013, 23, 947 CrossRef CAS
.
- S. Rajendran, M. Sivakumar and R. Subadevi, Mater. Lett., 2004, 58, 641 CrossRef CAS
.
- G. B. Appetecchi, F. Crore and B. Scrosati, Electrochim. Acta, 1995, 40, 991 CrossRef CAS
.
- F. Croce, G. B. Appetecchi, L. Persi and B. Scrosati, Nature, 1998, 394, 456 CrossRef CAS PubMed
.
- R. Haggenmueller, J. E. Fischer and K. I. Winey, Macromolecules, 2006, 39, 2964 CrossRef CAS
.
- J. Zhou and P. S. Fedkiw, Solid State Ionics, 2004, 166, 275 CrossRef CAS PubMed
.
- D. K. Pradhan, R. N. P. Choudhary, B. K. Samantaray, N. K. Karan and R. S. Katiyar, Int. J. Electrochem. Sci., 2007, 2, 861 CAS
.
- M. Armand, F. Endres, D. R. MacFarlane, H. Ohno and B. Scrosati, Nat. Mater., 2009, 621 CrossRef CAS PubMed
.
- Y. S. Ye, J. Rick and B. J. Hwang, J. Mater. Chem. A, 2013, 1, 2719 CAS
.
- J. Lu, F. Yana and J. Texter, Prog. Polym. Sci., 2009, 34, 431 CrossRef CAS PubMed
.
- H. Ohno, Electrochemical Aspects of Ionic Liquids, John Wiley & Sons, Inc., Hoboken, New Jersey, 2005 Search PubMed
.
- C. A. Angell, Y. Ansari and Z. Zhao, Faraday Discuss., 2012, 154, 9 RSC
.
- M. P. Singh, R. K. Singh and S. Chandra, Prog. Mater. Sci., 2014, 64, 73 CrossRef CAS PubMed
.
- Shalu, S. K. Chaurasia, R. K. Singh and S. Chandra, J. Phys. Chem. B, 2013, 117, 897–906 CrossRef CAS PubMed
.
- I. Tim, J. H. Lee, M. L. Shofner, K. Jacob and R. Tannenbaum, Polymer, 2013, 53, 2402 Search PubMed
.
- A. J. Muller, M. L. Arnal, A. L. Spinelli, E. Canizales, C. C. Puig, H. Wang and C. C. Han, Macromol. Chem. Phys., 2003, 204, 1497 CrossRef
.
- K. L. Singfield, R. A. Chisholm and T. L. King, J. Chem. Educ., 2012, 89, 159 CrossRef CAS
.
- Shalu, S. K. Chaurasia and R. K. Singh, RSC Adv., 2014, 4, 50914 CAS
.
- K. E. Strawhecker and E. Manias, Chem. Mater., 2003, 15, 844 CrossRef CAS
.
- A. Keller, G. G. Wood and M. Hikosaka, Faraday Discuss., 1993, 95, 109 RSC
.
- Y. Zhang, H. Huo, J. Li, Y. Shang, Y. Chen, S. S. Funari and S. Jiang, CrystEngComm, 2012, 14, 7972 RSC
.
- S. Acierno, N. Grizzuti and H. H. Winter, Macromolecules, 2002, 35, 5043 CrossRef CAS
.
- J. Jin, M. Song and F. Pan, Thermochim. Acta, 2007, 456, 25 CrossRef CAS PubMed
.
- V. Sencadas, P. Martins, A. Pitaes, M. Benelmekki, R. L. G. Ribelles and S. Lanceros-Mendez, Langmuir, 2011, 27, 7241 CrossRef CAS PubMed
.
- H. Wang, J. K. Keum, A. Hiltner and E. Baer, Macromolecules, 2010, 43, 3359 CrossRef CAS
.
- D. Zhou, X. Mei and J. Ouyang, J. Phys. Chem. C, 2011, 115, 16688 CAS
.
- K. Chirssopoulou, K. S. Andrikopoulos, S. Bollas, C. Karageorgaki, D. Chirstofilos, G. A. Voyiatzis and S. H. Anastasiadis, Macromolecules, 2011, 44, 9710 CrossRef
.
- M. J. Avrami, J. Chem. Phys., 1939, 7, 1103 CrossRef CAS PubMed
.
- S. K. Chaurasia, R. K. Singh and S. Chandra, J. Polym. Sci., Part B: Polym. Phys., 2011, 49, 291 CrossRef CAS
.
- M. P. Scott, C. S. Brazel, M. G. Benton, J. W. Mays, J. D. Holbrey and R. D. Rogers, Chem. Commun., 2002, 1370 RSC
.
- N. Winterton, J. Mater. Chem., 2006, 16, 4281 RSC
.
- S. K. Chaurasia, R. K. Singh and S. Chandra, J. Raman Spectrosc., 2011, 42, 2168 CrossRef CAS
.
- S. K. Chaurasia, R. K. Singh and S. Chandra, CrystEngComm, 2013, 15, 6022 RSC
.
- S. K. Chaurasia, R. K. Singh and S. Chandra, Solid State Ionics, 2014, 262, 790 CrossRef CAS PubMed
.
- J. B. Zeng, F. Wu, C. L. Huang, Y. S. He and Y. Z. Wang, ACS Macro Lett., 2012, 1, 965 CrossRef CAS
.
- H. Zhao, Y. Bian, M. Xu, C. Han, Y. Li, Q. Dong and L. Dong, CrystEngComm, 2014, 16, 3896 RSC
.
- S. K. Chaurasia, R. K. Singh and S. Chandra, Solid State Ionics, 2011, 183, 32 CrossRef CAS PubMed
.
- Shalu, S. K. Chaurasia, R. K. Singh and S. Chandra, J. Appl. Polym. Sci., 2014 DOI:10.1002/app.41456
.
- A. L. Saroj and R. K. Singh, J. Phys. Chem. Solids, 2012, 73, 162 CrossRef CAS PubMed
.
- J. Maiz, J. Martin and C. Mijangos, Langmuir, 2012, 28, 12296 CrossRef CAS PubMed
.
- J. Li, C. Zhou, G. Wang, Y. Tao, Q. Liu and Y. Li, Polym. Test., 2002, 21, 583 CrossRef CAS
.
- J. B. Zeng, M. Srivanasan, S. L. Li, R. Narayan and Y. Z. Wang, Ind. Eng. Chem. Res., 2011, 50, 4471 CrossRef CAS
.
- A. Jeziorny, Polymer, 1978, 19, 1142 CrossRef CAS
.
- T. Ozawa, Polymer, 1971, 12, 150 CrossRef CAS
.
- T. Liu, Z. Mo, S. Wang and H. Zhang, Polym. Eng. Sci., 1997, 37, 568 CAS
.
- L. Wang, G. Li, Y. Chen and S. Li, Polym. Eng. Sci., 2012, 52, 1621 CAS
.
- K. Cao, Q. Wang, Y. C. Zhou, J. M. Sun and Z. Yao, Ind. Eng. Chem. Res., 2012, 51, 5461 CrossRef CAS
.
- M. L. Di Lorenzo and C. Silvestre, Prog. Polym. Sci., 1999, 24, 917 CrossRef CAS
.
- A. S. Fischer, M. B. Khalid, M. Widstrom and P. Kofinas, J. Power Sources, 2011, 196, 9767 CrossRef PubMed
.
- S. K. Chaurasia and R. K. Singh, Phase Trans., 2010, 83, 457 CrossRef CAS
.
- T. Wei, S. Pang, N. Xu, L. Pan, Z. Zhang, R. Xu, N. Ma and Q. Lin, J. Appl. Polym. Sci., 2014 DOI:10.1002/app.41308
.
- H. E. Kissinger, Anal. Chem., 1957, 29, 1702 CrossRef CAS
.
- Y. Zheng, M. L. Bruening and G. L. Baker, J. Polym. Sci., Part B: Polym. Phys., 2010, 48, 1955 CrossRef CAS
.
- A. G. Shtukenberg, Y. O. Punin, E. Gunn and B. Kahr, Chem. Rev., 2012, 112, 1805 CrossRef CAS PubMed
.
- J. Xi, X. Qiu, S. Zheng and X. Tang, Polymer, 2005, 46, 5702 CrossRef CAS PubMed
.
- B. K. Choi, Solid State Ionics, 2004, 168, 123 CrossRef CAS PubMed
.
- D. F. Miranda, T. P. Russell and J. J. Watkins, Macromolecules, 2010, 43, 10528 CrossRef CAS
.
- Z. B. Zeng, F. Wu, C. L. Huang, Y. S. He and Y. Z. Wang, ACS Macro Lett., 2012, 1, 965 CrossRef
.
| This journal is © The Royal Society of Chemistry 2015 |









