Synergistic effect of carbon fibers and carbon nanotubes on improving thermal stability and flame retardancy of polypropylene: a combination of a physical network and chemical crosslinking†
Abstract
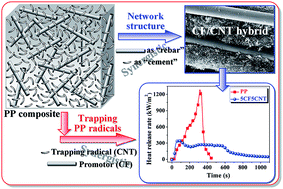
Recently, an intensive research effort has been devoted to the fabrication of polymer composites with enhanced physical and chemical properties. In this work, the combination of carbon fibers (CFs) and carbon nanotubes (CNTs) was demonstrated to show a synergistic effect on improving the thermal stability and flame retardancy of polypropylene (PP). The results of morphology characterization indicated that both CFs and CNTs were well dispersed in the PP matrix. The temperature at the maximum weight loss rate of PP under an air atmosphere was dramatically increased by 93.4 °C, and the peak value of the heat release rate measured by a cone calorimeter was significantly reduced by 71.7%. The remarkably improved thermal stability and flame retardancy of PP were partially owing to the accelerated oxidation crosslinking reaction of PP radicals by CNTs (chemical effect), and partially to the in situ formation of a dense and continuous CF/CNT hybrid protective layer (physical effect). This was because the CF/CNT hybrid protective layer not only hindered the diffusion of oxygen into PP and the migration of volatile decomposition products out of PP, but also acted as a thermal shield for energy feedback from the flame.

 Please wait while we load your content...
Please wait while we load your content...