Iron chelation and liver disease healing activity of edible mushroom (Cantharellus cibarius), in vitro and in vivo assays
Abstract
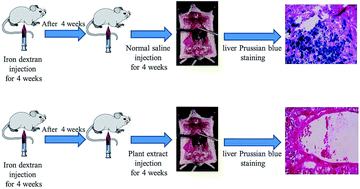
Cantharellus cibarius, an edible mushroom, has been reported to display a wide variety of biological properties, including iron-chelation activity. In the current study we evaluated the chelating capacity of ethyl acetate and methanolic extracts of Cantharellus cibarius on iron-overloaded mice. Extracts and defroxamine were injected for a frequency of 5 times a week for 4 weeks. Total iron and Fe3+ content of plasma was determined by atomic absorption spectroscopy and kit respectively. Liver sections were stained by haematoxylin and eosin and Perls' stain. Iron-overloaded animals treated with the extract, showed a dramatic decrease in plasma iron content when compared with the control group. The highest activity was observed in the methanolic extract. High-performance liquid chromatography was performed to simultaneously separate 5 phenolic acids and 2 flavonoids in extracts. p-Coumaric acid and ferulic acid were discovered to be major phenolic acids in ethyl acetate extract and methanol extract, respectively. Both methanolic and ethyl acetate derived extracts of mushroom Cantharellus cibarius exhibit satisfactory potency to chelate excessive iron in mice.

 Please wait while we load your content...
Please wait while we load your content...