Monitoring analgesic drug using sensing method based on nanocomposite
Abstract
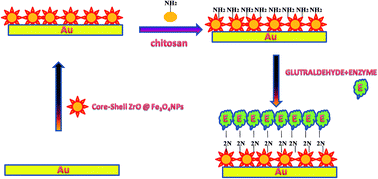
This paper reports a rapid, reliable and sensitive electrochemical method for the determination of acetaminophen, a safe analgesic drug. Most methods currently used for therapeutic drug monitoring require a pre-treatment of the sample. Biosensors avoid this kind of drawbacks. A horseradish peroxidase (HRP) was immobilized using core–shell ZrO@Fe3O4 nanoparticles on chitosan hybrid film electrodeposited on the surface of an Au electrode. The surface functionalization of core–shell ZrO@Fe3O4NPs on a chitosan hybrid film was characterized by cyclic voltammetry (CV), scanning electron microscopy (SEM), and electrochemical impedance spectroscopy (EIS). The experimental variables that can affect the acetaminophen amperometric response, such as the pH, temperature and applied potential, have been optimized to perform a selective determination of acetaminophen. An average limit of detection of 0.01 μM (S/N = 3) was obtained. The biosensor was finally applied to the determination of acetaminophen in complex matrices, such as pharmaceutical drugs.

 Please wait while we load your content...
Please wait while we load your content...