Reactive force field simulation on polymerization and hydrolytic reactions in calcium aluminate silicate hydrate (C–A–S–H) gel: structure, dynamics and mechanical properties
Dongshuai Hou*a,
Zongjin Li*b and
Tiejun Zhaoc
aQingdao Technological University, Cooperative Innovation Center of Engineering Construction and Safety in Shandong Blue Economic Zone, Qingdao, China. E-mail: dhou@ust.hk; monkeyphil@126.com; Tel: +86267636710
bQingdao Technological University, Cooperative Innovation Center of Engineering Construction and Safety in Shandong Blue Economic Zone, Qingdao, China. E-mail: zongjin@ust.hk
cQingdao Technological University, Cooperative Innovation Center of Engineering Construction and Safety in Shandong Blue Economic Zone, Qingdao, China. E-mail: ztjgp@263.net
First published on 19th November 2014
Abstract
The reactive force field method was first utilized to characterize the structure, dynamics and mechanical properties of calcium–aluminate–silicate–hydrate, which is essential in the chemistry of high alumina layered gel in Portland cement. In order to study the role of Al atoms, the properties of Al atoms located in the calcium silicate sheet and the interlayer region have been investigated. The Si–Al substitution in the calcium silicate sheet has not changed the layered structure of the C–A–S–H gel. On the other hand, the presence of Al atoms in the interlayer region improves the structure and mechanical performance significantly. The connectivity factor, Q, species evolution indicates that the aluminate species in the interlayer region play an essential role in bridging the defective silicate chains and transforming the layered C–A–S–H gel at low Al/Ca levels to the branch network structure at high Al/Ca levels. The structural transition is partly attributed to the aluminate–silicate connection by the NBO sites and is partly caused by the polymerization reaction between the aluminate species, both of which can be described by the reactive force field. Additionally, the polymerization reaction by the aluminate species also leads to a hydrolytic reaction. In this way, a lot of water molecules are transformed to hydroxyls, even bridging oxygen atoms. Dynamically, due to the high strength of the Al–O bond, the aluminate–silicate network in the C–A–S–H gel has a better stability at higher Al/Ca ratios. Furthermore, uniaxial tension tests on the C–A–S–H gels demonstrate the mechanical behavior and large structural deformation of the gel. Both the Young’s modulus and tensile strength are improved significantly with increasing aluminum content, indicating a good loading resistance ability in the aluminate–silicate network. The tensile deformation, simulated by the reactive force field, is also coupled with de-polymerization of the aluminate species and the water dissociation reaction, which shows good plasticity due to the Al atom addition.
1. Introduction
Concrete is the most widely utilized construction and building material in modern cities, and the manufacturing process of concrete is closely related with the environment. Unfortunately, the production of traditional Portland cement results in significant release of CO2 and other greenhouse gases.1 Hence, environmental friendliness acts as the leading issue in the sustainable development of the cement and concrete industry. In particular, mineral admixtures, partially substituting the cement materials, provide a promising way to reuse industrial waste and reduce energy consumption. Due to their pozzolanic properties, furnace slag and fly ash can be used as a replacement for some of the Portland cement content of concrete.2,3 Since the content of the furnace slag and fly ash includes some aluminate phases, the hydration reaction produces calcium–aluminate–silicate–hydrate (C–A–S–H) gel, like the calcium–silicate–hydrate (C–S–H) gel in OPC cement.4–6C–A–S–H gel, as the main hydration product, is responsible for the mechanical properties and durability of the materials. The introduction of aluminate species can effectively improve the chemical stability, mechanical and transport properties of the traditional binding phase, C–S–H gel. Recently, archaeologists excavated old concrete constructed in the Roman period from the ocean near Naples.7,8 Despite thousands of years of immersion in sea water, the integrity of the structure of the concrete remained, exhibiting very good durability. It is worth noting that the main hydration product in the old concrete is C–A–S–H gel.
Great effort has been devoted to investigating the morphology and properties of C–A–S–H gel by means of experimental studies and simulation techniques. Experimental results have provided significant insights into the structural mechanisms of Al-substitution in the C–S–H phase. NMR tests show some local structural information on the aluminate atoms.9–11 Tetrahedral, pentahedral and octahedral aluminate species coexist in the C–A–S–H gel. The multiple roles of the Al atoms are due to the complicated environment of the Al species in the C–A–S–H gel; while AlIV occurs on the bridging tetrahedra of the drierkette Al–silicate chains, AlV and AlVI occur in the interlayer and perhaps on particle surfaces. In addition, with increasing Ca/Si ratio for the C–S–H gel, the location of the incorporation of the aluminate species also varied significantly.12 It is also worth noting that the aluminate species are coordinated by the highly adsorbed water molecules and hydroxyls.13 Furthermore, experimental results also demonstrated that aluminate species can enhance polymerization due to their tendency to form a bridging tetrahedron between silicate units.14,15 By using NMR testing,16,17 it is widely accepted that the C–A–S–H gel has longer silicate chains than in the C–S–H gel in the OPC cement paste. Additionally, the signal of the Q3 species in the spectrum indicates the appearance of network structures,11 which has not been observed in the C–S–H gel.
Based on the findings from experiments, molecular simulation has been utilized to give a more comprehensive understanding of the structure of C–A–S–H gel. Puertasa16 constructed a model by replacing the silicon with aluminum atoms in tobermorite 11 Å and 14 Å (the mineral analogue of the C–S–H gel). Even though the model can predict the mechanical properties of the C–A–S–H gel, the tobermorite structure is not able to represent the real structure of the C–S–H gel with respect to the chemical composition and density.18 Recently, by using empirical force field simulation, the C–A–S–H model has been constructed by substituting the atoms in the “realistic model” of C–S–H with aluminum atoms.19 This work can be considered as a new application for a realistic model and provides new insights on the local structure, the connectivity of the Q species as well as the deformation of the C–A–S–H gel. However, the C–A–S–H gel model should be further improved in two respects: the empirical force field cannot describe the dissociation of water molecules so that no hydroxyl is present in the C–A–S–H gel, and the Q species distribution in the realistic model overestimates the Q0 species so that the silicate skeleton role is weakened.20
In this work, reactive force field molecular simulation is employed to investigate the structural, dynamical and mechanical properties of the C–A–S–H gel. The model of the C–A–S–H gel is constructed by replacing the Si atoms and interlayer Ca atoms in the improved “realistic model” of the C–S–H gel. The Si–Al substitution in the silicate chains has been widely confirmed by experimental findings.21 However, the Ca–Al substitution can be understood as the presence of Al atoms in the interlayer region. In this way, the complicated local environment of the Al atoms can be investigated. With different substitution ratios of aluminum atoms, the role of the aluminum atoms in the structure and dynamic properties can be investigated. Additionally, the uniaxial tension test simulates the deformation of the C–A–S–H gel and provides the stiffness and cohesive force in the gel.
2. Method
2.1 Reactive force field
The reactive force field (ReaxFF) method, developed by van Duin et al.,22 is utilized to simulate the chemical reactions for both atomic structure construction and uniaxial tensile testing. The reactive force field provides an advanced description of the interaction between Ca, Si, Al, O and H atoms in the C–S–H gel. The short-range interactions for the ReaxFF are determined by a bond length–bond order scheme so that the bonds can be broken and formed, with the potential energy transforming into a smooth state.23 On the other hand, the long-range coulombic interactions are determined by a 7th order taper function, with an outer cut off radius of 10 Å. The reactive force field has been widely utilized in silica–water interfaces,24 calcium silicate hydrate gel25 and nano-crystals.26 The parameters of the force field for Ca, Si, Al, O and H can be directly obtained from previous published reference data.22,27,282.2 Model construction
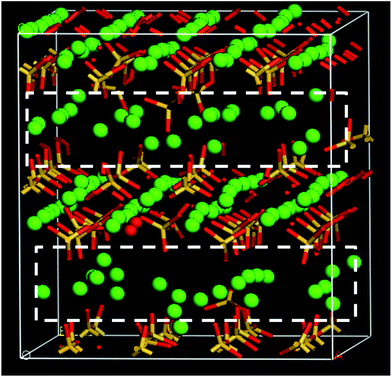
For the C–A–S–H gel, the model construction in the present study is based on the procedures that combine the methods proposed by Hou,29 Pellenq,18 Manzano30 and Qomi.19 Firstly, the layered analogue mineral of C–S–H, tobermorite 11 Å without water, was taken as the initial configuration for the C–S–H model.31,32 Silicate chains were then broken to match the Q species distribution with Q1 = 73.9%, Q2 = 21.4% and Q0 = 4.7%. The mean silicate chain length (MCL = 2(Q2/Q1 + 1) = 2.58) is consistent with the results obtained both from NMR testing33 and molecular dynamics simulation.34 It is worth noting that the Q0 percentage is controlled to less than 5%, also matching well with experimental results.35 Meanwhile, the Ca/Si ratio ranges from 0.7 to 2.3 with an average value of 1.7. In order to increase the Ca/Si ratio, some of the bridging SiO2 units are first removed as proposed by Pellenq et al.18 More importantly, some dimer structures (Si2O4) are also removed to satisfy the Q species distribution, especially for low Q0 percentages. After omission of the dimer structure, to maintain charge neutrality, some oxygen atoms in the silicate chains have to remain in the form of dangling atoms. It should be noted that the dangling atoms are not the final state for the oxygen atoms. After water adsorption and reactive force field MD in the following procedures, the dangling oxygen atoms can react with neighboring water molecules and transform to hydroxyl groups. In this way, the dry calcium silicate skeleton is obtained, as shown in Fig. 1.Subsequently, the silicon atoms in the chains and interlayer Ca atoms are gradually replaced by Al atoms. Considering the charge balance, the Si–Al substitution requires including some H atoms by transforming the Si–O bond to Si–OH. For the Ca–Al substitution, two Al3+ atoms replace three Ca2+ atoms to maintain the charge neutrality. For the former case, five dry C–A–S–H samples with Al/Si ratios of 0, 0.075, 0.133, 0.3 and 0.88 can be constructed. For the latter case, five dry C–A–S–H samples with Al/Ca ratios of 0, 0.075, 0.16, 0.26 and 0.38 can be obtained (Al/Ca = 0 is the C–S–H gel).
After the dry samples were constructed, the Grand Canonical Monte Carlo (GCMC) method was utilized to investigate the structure of the dry calcium aluminate silicate skeleton immersed in water solution.36 The dry sample obtained by the temperature quenching method is utilized for simulation. GCMC simulations determine the properties of the water molecules confined in the calcium aluminate silicate system at constant volume V, in equilibrium with a fictitious infinite reservoir of liquid bulk water solution that imposes its chemical potential μ = 0 eV and its temperature T = 300 K.18 The simulation process is analogous to water adsorption in micro-porous phases such as calcium silicate hydrate and zeolites.37 The simulation included 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles for the system to reach equilibrium, followed by 100
000 cycles for the system to reach equilibrium, followed by 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles for the production run. For each cycle, it is attempted to insert, delete, displace and rotate water molecules 1000 times in the constant volume calcium aluminate silica hydrate system.
000 cycles for the production run. For each cycle, it is attempted to insert, delete, displace and rotate water molecules 1000 times in the constant volume calcium aluminate silica hydrate system.
Finally, the chemical formula of the saturated C–S–H structure in the current simulation is given by (CaO)1.69(SiO2)·(H2O)1.82, which is quite close to (CaO)1.7(SiO2)·1.8H2O obtained by the SANS test.38 The reactive force field molecular dynamic simulations under constant pressure and temperature (NPT) for 300 ps give the structures of the C–S–H gel at the equilibrium states. A further 1000 ps NPT run is employed to achieve the equilibrium configuration for structural and dynamic analysis.
2.3 Uniaxial tension test
Uniaxial tension testing is employed to investigate the mechanical behavior of the tobermorite and the C–S–H gel. Super-cells, obtained by periodically extending the simulated model in Section 2.2 by a factor of two, underwent uniaxial tension in the y and z direction. The super-cells were composed of 7744 to 8064 atoms, with the size around 40 Å × 40 Å × 40 Å. It should be noted that using a large number of atoms in this study can give stable statistical simulation results, especially with regard to reliable failure modes. In order to explore the failure mechanism of the layered structure, the stress–strain relation and the deformation of the molecular structure were investigated in the loading process.To obtain the stress–strain relation, the structure was subjected to uniaxial tensile loading through gradual elongation at constant strain rates of 0.08 per ps. In the whole simulation process, NPT ensembles are defined for the system. Take the tension along the y direction for example: firstly, the super-cells were relaxed at 300 K and coupled to zero external pressure in the x, y, z dimensions for 500 ps. Then, after the pressures in the three directions reached equilibrium, the C–S–H structure would be elongated in the y direction. Meanwhile, the pressure in the x, z directions was kept at zero. Pressure evolution in the y direction was taken as the internal stress σyy. Setting the pressure perpendicular to the tension direction to zero can allow the normal direction to relax un-isotropically without any restriction. The setting, considering Poisson’s ratio, can eliminate the artificial constraint for the deformation.
3. Results and discussion
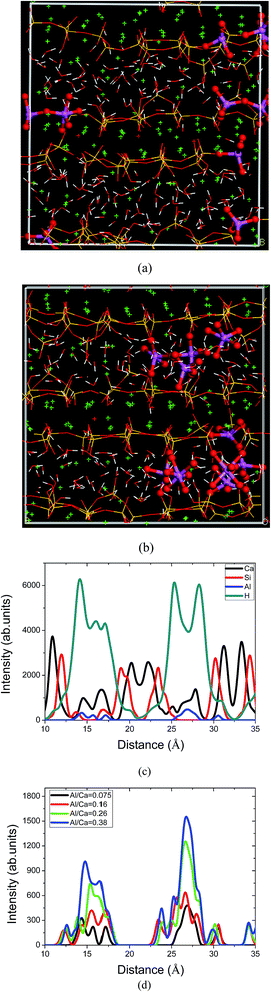
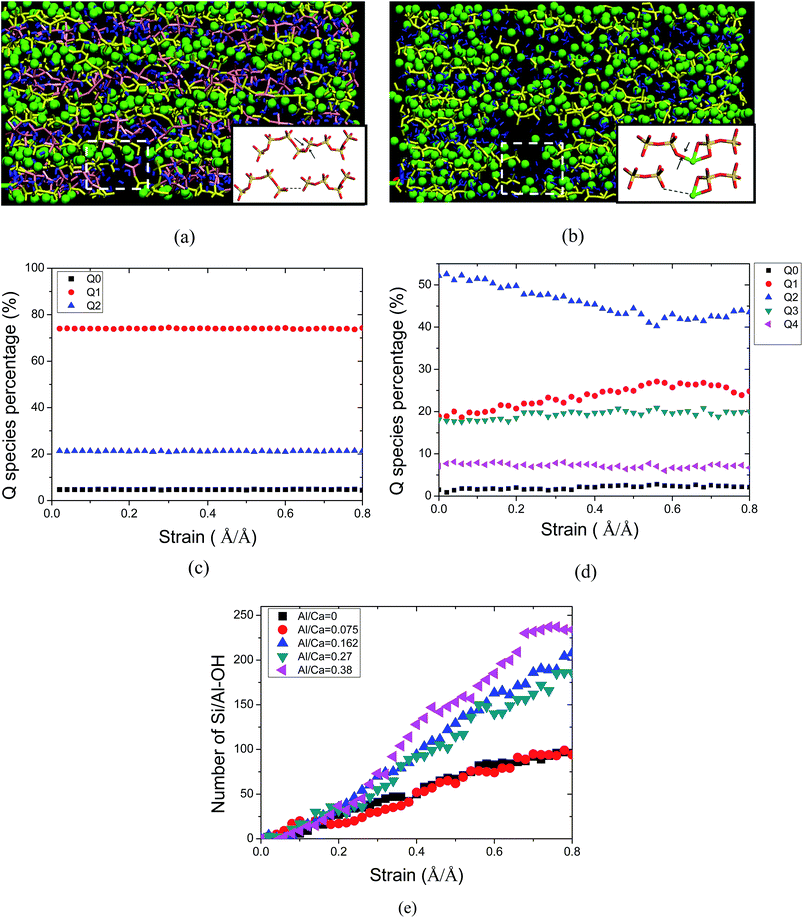
For the samples with Si–Al substitution, the simulated molecular structure of C–A–S–H at the Al/Si ratio of 0.075 is shown in Fig. 2a. It shows that the incorporation of aluminate atoms has not changed the silicate skeleton to a great extent. The aluminate atoms present in the bridging and pairing sites of the chains grows along the y direction. No further polymerization and de-polymerization can be observed in the silicate–aluminate chains, indicating the chemical stability of the Al–Si substitution. The Si substitution for Al is possible due the similar size and charge of these atoms. This matches well with NMR findings that the Al substitution is in the silicate chains in many synthesized C–S–H gels.10,11 In addition, five-coordinated aluminate species are widely found in the aluminate silicate chains. The aluminate atoms, closed to the interlayer water molecules and hydroxyl groups, are likely to have a strong Al–Ow connection.On the other hand, for aluminate atoms present in the interlayer region, the molecular structure of the C–A–S–H gel has been changed significantly. The simulated molecular structure of C–A–S–H at an Al/Ca ratio of 0.075 is shown in Fig. 2b; correspondingly, the intensity profiles of different atoms at equilibrium state are plotted in Fig. 2c versus the distance in the z direction. It can be clearly observed in Fig. 2b that calcium atoms and the surrounding oxygen atoms form a Ca–O octahedron, constructing the Ca sheet. Defective silicate chains graft on both sides of the Ca sheets in the C–S–H gels. Between the neighboring calcium silicate sheets locate the interlayer calcium atoms (Caw), aluminum atoms and water molecules. As shown in Fig. 2c, the alternative maxima of Ca, Si, Al and H atoms in the density profiles indicate that the C–A–S–H gel has a sandwich-like structure. In the intra-layer, Al atoms grow into the deficient region of the broken silicate chains and elongate the silicate chain length in the y direction. Besides, the healing effect of the Al atoms in molecular structure reconstruction can enhance the interlayer connections. As shown in Fig. 2d, Al atoms are distributed continuously across the interlayer region, with increasing Al content. It is valuable to note that the Al atoms associated with the non-bridging oxygen atoms in the defective silicate chains bridge the neighboring calcium silicate sheets. In this way, the aluminum–silicate network is gradually constructed in the interlayer region. Hence, the addition of Al atoms can transform the C–S–H gel from a layered crystal to a glassy-like structure.
On the other hand, irregular distribution of the H atoms in the interlayer region implies that the structure of the water molecules, highly confined by the nano-pores, is significantly distorted by the complicated local calcium aluminate silicate environment. The large intensity peaks of the H atoms also reflect that the defective silicate chains, with large numbers of NBO atoms, have a good ability to adsorb water molecules. In Fig. 2b, the intensity distributions of the H atoms and Si atoms overlap, which means that water molecules are not only present in the interlayer regions but also diffuse into the defective region of the calcium silicate sheet. Additionally, the aluminate hydroxyls observed in the interlayer region imply that the addition of Al atoms has a great influence on the hydrolytic reaction of the water molecules.
In order to give more quantitative insights on the molecular structure of the C–A–S–H gel, the local structure of the Al atoms is further investigated by the Q species distribution, the radial distribution function, the angle distribution function and the coordination number.
3.1 Connectivity factor
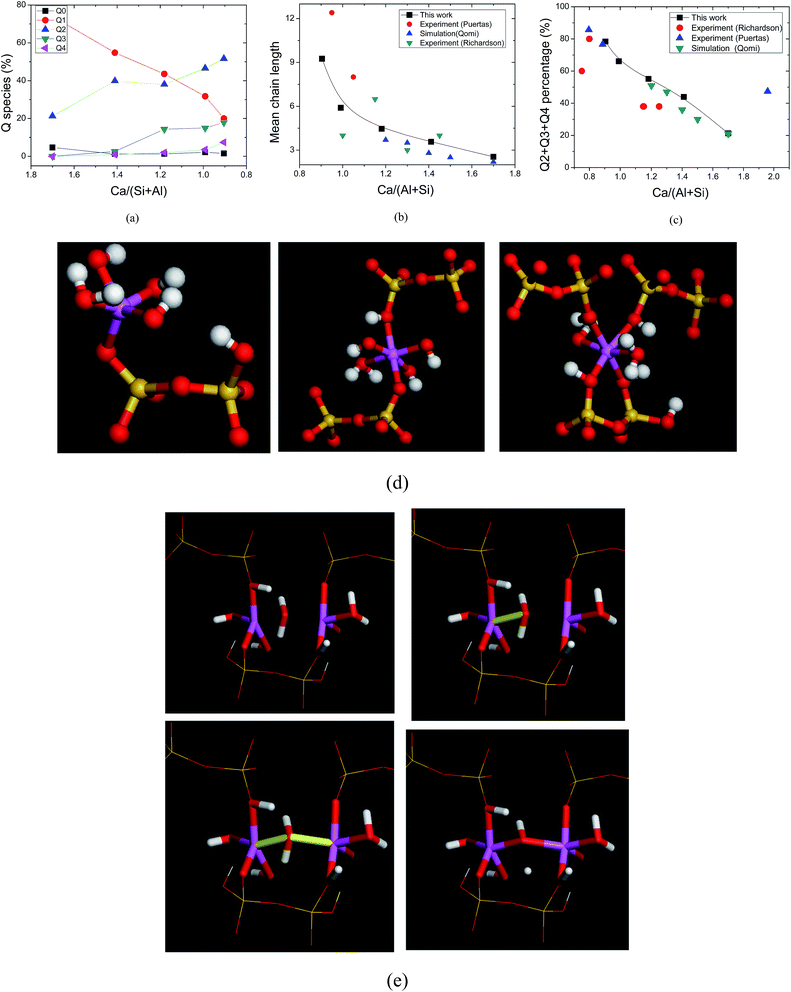
As in the silicate composite structure, the connectivity factor, Qn (n = 0, 1, 2, 3, 4), is an important parameter to estimate the silicate connection, where n is defined as the number of connected neighboring silicate tetrahedrons. Q0 is the monomer, Q1 represents the dimer structure (two connected silicate tetrahedrons), Q2 is the long chain, Q3 is the branch structure and Q4 is the network structure (Feuston and Garofalini, 1990; ref. 39). For the aluminate–silicate substitution, the Q species distribution remains unchanged. At a Ca/(Al + Si) ratio of 1.7, the dimer structure has the predominant percentage. On the other hand, the aluminate species in the interlayer region change the Q species distribution significantly. As shown in Fig. 3a, with a decreasing Ca/(Al + Si) ratio from 1.7 to 0.9, the percentage of the Q1 species is reduced significantly from around 74% to 20%. The Q0 species completely disappeared when the Ca/(Al + Si) ratio reached 0.9. On the contrary, the ratios of the Q2, Q3 and Q4 species grew to 51.6%, 17.9% and 7.4%, respectively. It is worth noting that the growth of the Q species follows the sequence: Q2, Q3 and Q4. This trend indicates that the silicate–aluminate structures first transform from the short dimer structure to long chains at low aluminum content, and subsequently turn to branches and network structures at high aluminum content. The Q species distribution in the current simulation confirms the observation by NMR experiment that the signal of the Q2 species is intensified and the Q3 species appears.11 The mean chain length, defined as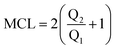 , is calculated as a function of the Ca/(Al + Si) ratio. As shown in Fig. 3b, the tendency for MCL to decrease with increasing Ca/(Al + Si) ratio matches well with the results obtained from previous simulation by the empirical force field method19 and experimental work by NMR.16,40 Additionally, as shown in Fig. 3c, the percentage of Q2, Q3 and Q4 species, reflecting the polymerization degree, reduces with increasing Ca/(Si + Al) ratio. However, compared with the percentages obtained from previous simulations by the empirical force field method,19 the current simulation work demonstrates higher values. In particular, at a Ca/(Si + Al) ratio of around 1.2, the percentage of Q3 and Q4 species is 16%, twice as large as the value from previous simulations. The large discrepancy reflects different polymerization mechanisms predicted by the reactive force field and the empirical force field.
, is calculated as a function of the Ca/(Al + Si) ratio. As shown in Fig. 3b, the tendency for MCL to decrease with increasing Ca/(Al + Si) ratio matches well with the results obtained from previous simulation by the empirical force field method19 and experimental work by NMR.16,40 Additionally, as shown in Fig. 3c, the percentage of Q2, Q3 and Q4 species, reflecting the polymerization degree, reduces with increasing Ca/(Si + Al) ratio. However, compared with the percentages obtained from previous simulations by the empirical force field method,19 the current simulation work demonstrates higher values. In particular, at a Ca/(Si + Al) ratio of around 1.2, the percentage of Q3 and Q4 species is 16%, twice as large as the value from previous simulations. The large discrepancy reflects different polymerization mechanisms predicted by the reactive force field and the empirical force field.
The connectivity transition is attributed to the polymerization role of the aluminum atoms in the network construction. On the one hand, as shown in Fig. 3d, the Al atoms connect with the NBO atoms in the defective silicate chains so a long aluminate–silicate chain is formed. Since the number of NBO atoms coordinated with the Al atom ranges from 1 to 4, the aluminate structure varies from Q1 to Q4. This reaction between Al and the silicate chains can be described by both the reactive and empirical force fields. On the other hand, when the NBO sites in the silicate chains are occupied by the Al atoms at high Al/Ca ratios, polymerization reactions between different Al species occur. Fig. 3e shows the reaction process: one water molecule diffuses to one nearby Al atom and forms a Al–Ow (Ow: oxygen atom in water) connection; subsequently another Al–Ow bond is also constructed; finally water dissociates one hydrogen atom and a stable Al–Oh–Al bond is formed (Oh: oxygen atom in hydroxyl). Water molecules play an essential role in bridging the two neighboring Al species during the polymerization process. Meanwhile, the Ow atoms transform to the bridging oxygen atoms (BO) in the aluminate–silicate skeleton. Since the empirical force field can only simulate Al–Ow rather than further water dissociation processes, the Q3 species formation is restricted to a great extent.
3.2 Local structure of Al atoms
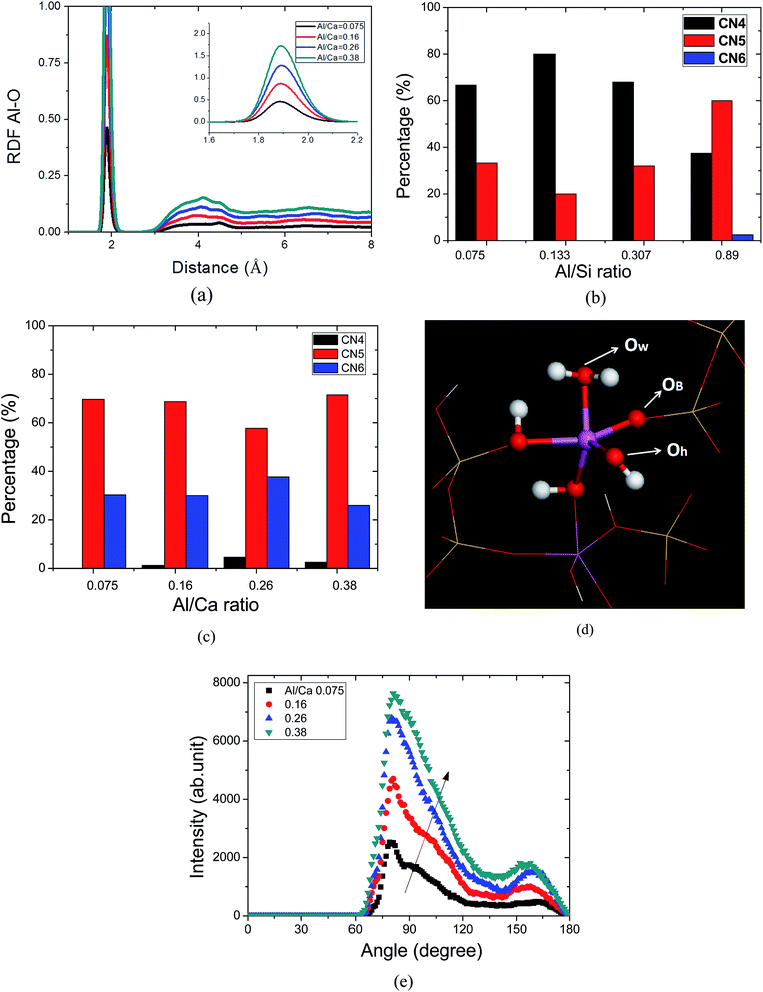
RDF curves of Al–O bonds in the C–A–S–H gel with different Al/Ca ratios are plotted in Fig. 4a. For bonded atoms, the relevant bond distances can be readily determined by the positions of the peaks in the corresponding RDF. In the C–A–S–H gels, the reactive force field yielded a Al–O bond distance from 1.88 Å to 1.89 Å, which is close to the value of 1.9 Å obtained from previous simulations involving the empirical force field.19 In the medium range, the Al–O spatial correlation in C–A–S–H is extended to 4 Å, at which point the intensity peaks gradually increase with Al concentration. The longer distance correlation in the C–A–S–H gel at high Al/Ca ratios can be interpreted as a stronger connection attributed to the branch structure formation.For the Al–Si substitution, as shown in Fig. 4b, the coordinated number (CN) of aluminum atoms, describing the amount of neighboring oxygen atoms, ranges from 4 to 6, with 4 occupying the predominant percentage. This means that the aluminate species in the silicate chains are mainly in the form of tetrahedrons. On the other hand, as shown in Fig. 4c, the CN distribution indicates that the structures of the aluminate atoms are pentahedrons and octahedrons. It should be noted that in NMR testing, the relative concentration of visible AlV and AlVI species accounts for 85% of the hydrated aluminate cement products, with 15% AlIV species.9 In addition to confirming the coexistence of AlIV, AlV and AlVI species in the C–A–S–H gel, the simulation results also suggest that the aluminate species located in the calcium silicate sheet and interlayer region demonstrate different local structures.
In previous experiments,9 2D 29Si{1H} and 27Al{1H} NMR spectra revealed that heterogeneous distributions of hydroxyl species and strongly adsorbed water are present in the hydrated aluminate products. MD simulations can quantitatively access those hydroxyl and water molecules associated with Al atoms. As shown in Fig. 4d, the coordinated oxygen atoms of the Al species are decomposed into hydroxyl oxygen (Oh), bridging oxygen (Ob) and water oxygen (Ow) to analyze the local structural difference. The hydroxyl groups in the C–A–S–H gel are produced by the hydrolytic reaction. The water molecules dissociate into H+ and OH− ionic pairs; the former diffuse near the ONB sites in the defective silicate chain, forming silicate hydroxyl. The remaining OH− group can either associate with Caw atoms or Al atoms in the interlayer region. It is interesting to note that Ob in the Al–Ob–Al and Si–Ob–Al can further react with water, forming an Ob–H bond. This is quite different to the siloxane bond (Si–O–Si) in the C–S–H gel in that the Ob atoms have no reactivity with water molecules.30 The reactivity discrepancy indicates that the Al–O–Al bond has a hydrophilic feature, contrary to the hydrophobic nature of bridging oxygen atoms in the siloxane bond. Fundamentally, the electronic attraction of Al–Ob is slightly weaker than that of Si–Ob, which prevents additional H+ from associating.
Table 1 lists the average coordination number (CN) of aluminate atoms at different Al/Ca ratios. In all the C–A–S–H gels, the CN value of the Ob and Oh atoms is more than 4.6, which is more than 85% of the total CN. The small ratio of coordinated Ow atoms implies that the water molecules strongly absorbed by the Al atoms probably dissociate into hydroxyl and H+. Besides, as shown in Table 1, with increasing Al/Ca ratios, the reduction of the CN value for Oh is accompanied by the increase of Ob. The transition from Oh atoms to Ob atoms is attributed to the polymerization of the aluminate species, as discussed in the previous section.
| Al/Ca | Oh | Ob | Ow | Total |
|---|---|---|---|---|
| 0.075 | 2.3 | 2.5 | 0.6 | 5.4 |
| 0.16 | 2.05 | 2.55 | 0.7 | 5.3 |
| 0.26 | 2.03 | 2.63 | 0.8 | 5.47 |
| 0.38 | 1.8 | 3 | 0.6 | 5.4 |
The O–Al–O angle distribution, exhibited in Fig. 4e, provides geometric information on the aluminum-oxide local structure. The angle distribution for C–A–S–H is characterized by one intense and narrow peak at around 80°, and a small and broad peak at 160°, that correspond to the distorted Al–O octahedral or pentahedral arrangement. It should be noted that the peak at 80° has also been observed in calcium aluminate glass41 and silicate–aluminate glass42 at high aluminum concentration, implying an amorphous glassy nature for the local structure of the Al atoms. Besides, as shown in Fig. 4e, a shoulder located at around 90° for the C–A–S–H gel gradually disappears with increasing Al/Ca ratio and the second peak at 160° become more pronounced at high Al/Ca ratios. The angle distribution evolution reflects the interaction between aluminate species and silicate chains at different Al/Ca ratios. At low Al/Ca ratio, the defective silicate chains provide a large amount of NBO sites for the Al–O bond to restrict the geometric arrangement of the aluminate octahedron. Since 109° is a typical value for the silicate tetrahedron structure, the aluminate octahedron is significantly influenced, resulting in a large shoulder in the angle distribution. On the other hand, at high Al/Ca ratio, the interplay between neighboring aluminate species dominates the angle distribution, which weakens the effect of the silicate chains. Apart from the influence of the defective silicate chains, the highly solvated calcium atoms confined in the interlayer region and the two dimensional confined geometry of the layered structure all determine the distorted octahedral structure of the aluminate species.
3.3 Dynamic properties
The previous discussion describes the molecular structure of the C–A–S–H gel at different Al/Ca ratios. The chemical bonds of Si–O, Ca–O, Al–O, H–O and the H-bonds play roles in bridging the neighboring calcium silicate sheets. The strengths of the bonds depend on the stability of the chemical bonds to a large extent. As in previous research studying the strength of the H-bonds,43 the time correlated function (TCF) is utilized to describe the dynamical properties of various chemical bonds.The TCF C(t) of a bond is described in eqn (1):
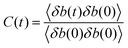 | (1) |
Fig. 5a demonstrates the evolution of the TCFs of Si–O, Ca–O, Al–O, H–O and H-bonds of C–A–S–H at an Al/Ca ratio of 0.26 over 10 ps in the equilibrium state. According to the reduction extent of C(t), the strength of the chemical bonds is ranked in the following order: Al–O, Si–O > O–H, Ca–O > H-bonds. The C(t) values of Si–O and Al–O almost remain at one without any reduction with time. This implies that the silicate–aluminate network, having good stability, acts as the skeleton of the C–A–S–H gel.
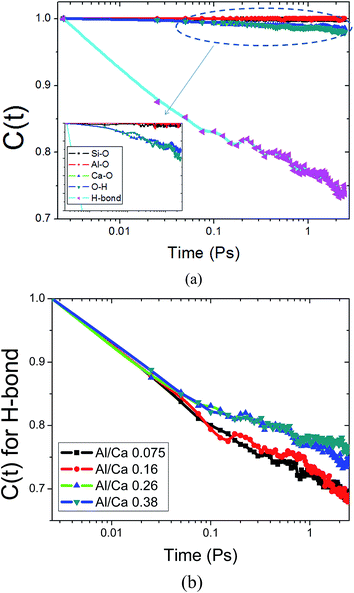 | ||
| Fig. 5 (a) Time correlated function (TCF or C(t)) of different bonds in the C–A–S–H gel; (b) TCF for H-bonds at different Al/Ca ratios. | ||
On the other hand, the slight reduction of C(t) for the O–H bond and Ca–O bond indicates a weaker connection than the silicate–aluminate skeleton. Water molecules and hydroxyl groups continuously dissociate and associate to maintain the hydrolytic equilibrium. The variation for the Ca–O bonds is mainly attributed to the frequent breakage of the connection between the water molecules and the interlayer Ca atoms. Caw atoms form clusters with water in the interlayer region, and the diffusion rate is thus accelerated to some extent. With respect to the chemical bonds, substitution of the interlayer Ca atoms with Al atoms transforms unstable Ca–Ow bonds to the high strength Al–O skeleton, which improves the stability of the interlayer region.
The lowest C(t) value of the H-bonds is partly attributed to the high diffusion rate of water molecules and is partly caused by the water reaction with neighboring Caw atoms and Al atoms. Hence, the stability of the H-bonds depends greatly on the local environment in the interlayer region. As shown in Fig. 5b, the C(t) value for the H-bonds gradually increases as the Al/Ca ratio becomes higher. Since Ow atoms in the water molecules transform to the Oh and OB atoms due to the polymerization role of the Al atoms, the percentage of H-bonds accepted by the structural oxygen atoms increases significantly. According to H-bond strength analysis by Youssef,43 the H-bonds formed by neighboring water molecules have lower strength than those constructed between structural oxygen and water molecules. Therefore, the stability of the H-bonds is indirectly enhanced due to the addition of Al atoms. The dynamic properties of the Si–O bonds, Al–O bonds and Ca–O bonds match well with previous findings by ab initio methods.44,28 The findings prove that Si–O bonds and Al–O bonds in the calcium silicate aluminate minerals have more binding energy than Ca–O bonds and are hard to break.
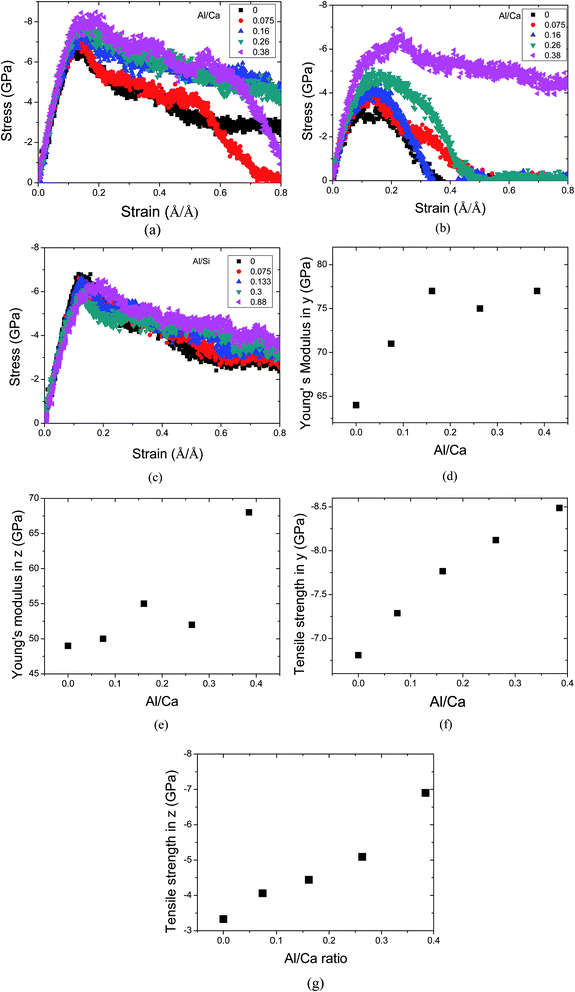
3.4 Stress–strain relation
The stress–strain curve can characterize the mechanical behavior of a layered structure during the tensile process and help gain insights into the constitutive relation between stress and strain. Different stress–strain relations of tensile loading in the y and z directions indicate the heterogeneous nature of the layered structures. In the y direction, as shown in Fig. 6a, during the tensile process, stress first increases linearly in the elastic stage and subsequently slowly increases to a maximum of 6.5 GPa at a strain of around 0.17 Å Å−1. After the maximum, the stress directly reduces slowly without obvious yield behavior. Even at a strain of 0.8 Å Å−1, the stress does not drop to zero, implying good plasticity along the y direction. On the other hand, the C–S–H gel is more likely to suffer breakage in the z direction, with the strain at the fracture state 0.4 Å Å−1, indicating that the interlayer structure shows more brittle features. The mechanical performance discrepancy in different directions confirms that the ionic–covalent bonds in the calcium silicate sheet are quite a bit stronger than the H-bonds in tensile loading resistance.For the Si–Al substitution, as shown in Fig. 6c, in the y direction, the stress–strain curves of C–A–S–H resemble that of C–S–H. The Young’s modulus and the tensile strength have not been changed to a great extent. This can be explained by the structural and dynamical role of Al atoms in the silicate chains. On one hand, the Si–Al substitution has not improved the polymerization degree in the silicate skeleton. With respect to the bond stability, as discussed in the previous section, the Al–O and Si–O bonds demonstrate equivalent bond strength. Hence, mechanical performance is not improved due to the Si–Al substitution.
On the contrary, for the Ca–Al substitution, as shown in Fig. 6d and e, with progressively increasing Al/Ca ratio from 0 to 0.38, Young’s modulus grows from 64 GPa to 75 GPa along the y direction and from 50 to 68 GPa along the z direction. The stiffness improvement is greatly related to the polymerization changes for the C–S–H gel with Al addition, as discussed in the connectivity section. Al species play a critical role in connecting the defective silicate chains and in transforming the C–S–H gel from a layered structure to a 3 dimensional network. While Ey increases significantly as the Al/Ca ratio is less than 0.2, the pronounced enhancement for Ez is from Al/Ca 0.26 to 0.38. The moduli evolution trend reflects the growth order for the aluminate network: at low Al/Ca ratios, the Al atoms mainly heal the defective calcium silicate sheet; at high Al/Ca ratios, the branches are constructed in the interlayer region. Young’s moduli obtained in the current simulations are consistent with the results from nano-indentation testing (60 GPa),45 previous ab initio calculations (55–68 Gpa)18 and molecular mechanical calculation (56–61.6 GPa).
Similar evolution trends in the tensile strength can be observed in Fig. 6f and g. The tensile strength increases from 6.5 GPa for the C–S–H gel to 8.5 GPa for the C–A–S–H gel with Al/Ca equal to 0.38 in the y direction, and from 3.25 GPa to 7 GPa in the z direction. The failure stresses obtained here are consistent with the rupture strength (∼3 GPa) by shear simulation18 and fluid pressure (2.75 GPa) calculation.36 The huge strength discrepancy between the C–S–H gel and C–A–S–H gel indicates different failure mechanisms. The chemical bonds determine the mechanical performance for different Al concentrations to a large extent. When Caw atoms are substituted by Al atoms, the unstable Ca–Ow bonds and H-bonds are partially substituted by the stable and high strength Al–Ob bonds, while the structure transforms from a layered structure to a network structure. The variations in the chemical bonds and structure that play important roles in bridging the calcium silicate sheets result in the strengthening of the cohesive force. In the aspect of “hydrolytic weakening”, Bonnaud proposed the disjoining effect of water molecules in C–S–H grains36 in which the fluid pressure in the C–S–H gel is contributed to by the repulsive force of the water molecules. However, in the C–A–S–H gel, water molecules, absorbed by the Al atoms, are further dissociated and transformed to bridging oxygen atoms or hydroxyls that are connected firmly with the calcium–aluminate–silicate structure. Hence, there is less chance for the water molecules to attack the ionic–covalent bonds.
3.5 Deformation of the structure
More information can be obtained from examining the deformed molecular structure of the C–A–S–H gel and C–S–H gel in resisting tensile loading. As shown in Fig. 7b, Ca–O bond breakage can be widely observed in the damaged C–S–H gel. Since the short silicate chains are separately distributed in the calcium silicate sheet, the mechanical contribution of the silicate chain, such as the siloxane bond stretching and the enlargement of the Si–O–Si angle, is limited to a great extent. On the contrary, in the C–A–S–H gel, the Al atoms heal the defective silicate chains and construct a relatively integral network structure. Hence, the changes of the Si–O–Al bonds and angles carry a major percentage of the tensile loading. It can be observed in Fig. 7a that the breakage of Si–O or Al–O bonds results in the de-polymerization of the aluminate–silicate network during the tensile process.To quantitatively access the morphology variation, the Q species distribution is monitored as a function of the strain. As shown in Fig. 7c, the percentages of the Q0, Q1 and Q2 species maintain constant values, implying that the short silicate chains are hard to be stretched to breaking. On the other hand, as shown in Fig. 7d, the Q2 species continuously decrease from 52.5% to 45%, while the Q1 species increase from less than 20% to 25%. Additionally, the total variation of Q0, Q3 and Q4 species is less than 2.5%. The transformation from Q2 to Q1 species implies that the long silicate chain is stretch broken into short silicate chains. Meanwhile, the newly formed short chains can be reconstructed with the neighboring aluminate–silicate network, which contributes to a small amount of other Q species. In this respect, the structural evolution due to tensile stretching can be interpreted as the de-polymerization of the silicate–aluminate network. The aluminate branched (Q3 and Q4) species, strongly restricted by the Al–O and Si–O bonds, can give a great mechanical contribution by local structural rearrangement. By previous ab initio calculation,44 the presence of the Q3 species can improve the interlayer connection of tobermorite46 (a kind of calcium silicate crystal) by forming a hinge mechanism.
Furthermore, as shown in Fig. 7a and b, in both the deformed C–S–H gel and C–A–S–H gel, the broken calcium silicate aluminate bonds are terminated by forming hydroxyls. As shown in Fig. 7e, the number of Si–OH and Al–OH bonds continuously increase during the tensile process, implying a progressive dissociation of water molecules. The mechanism of the hydrolytic reaction is that the tensile loading results in local bond elongation or even breakage, which lowers the energy barrier for the water dissociation.47,48 As compared with BO sites, water molecules are more likely to react with the NBO sites in aluminate–silicate chains. It also should be noted that at higher Al/Ca ratios, the hydroxyl number has a larger value in Fig. 7e. Since the de-polymerization reaction widely occurs at high Al/Ca ratios, more BO sites in the aluminate–silicate chains transform to NBO sites. In this way, water dissociation, accompanied by aluminate–silicate de-polymerization, produces large amounts of hydroxyl bonds.
4. Conclusions
Reactive force field molecular dynamic simulation was employed to investigate the molecular characteristics of the C–A–S–H gel, including the structural, dynamical and mechanical properties. The following conclusions can be made from this work.(1) The Si–Al substitution in the calcium silicate sheet has not changed the layered structure of the C–A–S–H gel. On the other hand, the presence of the Al atoms in the interlayer region improves the structure and mechanical performance significantly.
(2) The Q species evolution trend indicates that the silicate–aluminate structure first transforms from the short dimer structure to long chains at low aluminum content and subsequently turn to branches and a network structure at high aluminum content.
(3) The structural transition is partly attributed to the aluminate–silicate connection by the NBO sites and is partly caused by the polymerization reaction between the aluminate species, both of which can be described by the reactive force field.
(4) The coordinated oxygen atoms of aluminum atoms include water, hydroxyl and bridging oxygen atoms. Due to the polymerization reaction, partial hydroxyl groups transform to bridging oxygen atoms.
(5) Dynamically, due to the high strength of the Al–O bond, the aluminate–silicate network in the C–A–S–H gel has a better stability at higher Al/Ca ratios. The stable aluminate species also strengthens the neighboring H-bond network significantly.
(6) Uniaxial tension testing, performed on C–A–S–H gels, demonstrate the mechanical behavior and large structural deformation of the gel. Both Young’s modulus and the tensile strength are improved significantly with increasing aluminum content, indicating the good loading resistance ability of aluminate–silicate networks.
(7) Tensile deformation, simulated by the reactive force field, is also coupled with the de-polymerization of aluminate species and the water dissociation reaction, which shows good plasticity due to the addition of Al atoms.
Acknowledgements
Financial support from the China Ministry of Science and Technology under Grant 2015CB655100 and Major International Joint Research Project under Grant 51420105015 are gratefully acknowledged.References
- Z. Li, Advanced Concrete Technology, John Wiley & Sons Inc, Hoboken, N.J., 2011 Search PubMed.
- A. Scott and M. Thomas, Evaluation of Fly Ash From Co-Combustion of Coal and Petroleum Coke for Use in Concrete, ACI Mater. J., 2007, 104(1), 62–70 CAS.
- C. Hu and Z. Li, Micromechanical investigation of Portland cement paste, Construct. Build. Mater., 2014, 71, 44–52 CrossRef PubMed.
- I. Richardson, The nature of calcium silicate hydrate, Cem. Concr. Res., 1999, 29, 1131–1147 CrossRef CAS.
- H. Ma, D. Hou, J. Liu and Z. Li, Estimate the relative electrical conductivity of C–S–H gel from experimental results, Construct. Build. Mater., 2014, 71, 392–396 CrossRef PubMed.
- L. Zhang, Z. Lin and Z. Li, The structure of silicate ions in CSH discussed from chemical composition, Adv. Cem. Res., 2012, 24(5), 263–281 CrossRef CAS.
- A. Emwas, P. Monteiro, M. Jackson, S. Chae, S. Mulcahy, C. Meral, R. Taylor, P. Li, J. Moon, G. Vola and H.-R. Wenk, Unlocking the secrets of Al–tobermorite in Roman seawater concrete, Am. Mineral., 2013, 98, 1669–1687 CrossRef.
- M. D. Jackson, J. Moon, E. Gotti, R. Taylor, S. R. Chae, M. Kunz, A.-H. Emwas, C. Meral, P. Guttmann, P. Levitz, H.-R. Wenk and P. J. M. Monteiro, Material and Elastic Properties of Al–Tobermorite in Ancient Roman Seawater Concrete, J. Am. Ceram. Soc., 2013, 96, 2598–2606 CrossRef CAS PubMed.
- A. Rawal, B. Smith, G. Athens, C. Edwards, L. Roberts, V. Gupta and B. Chmelka, Molecular Silicate and Aluminate Species in Anhydrous and Hydrated Cements, J. Am. Chem. Soc., 2010, 132(21), 7321–7337 CrossRef CAS PubMed.
- M. D. Andersen, H. J. Jakobsen and J. Skibsted, Incorporation of aluminum in the calcium silicate hydrate (CSH) of hydrated Portland cements: A high-field 27Al and 29Si MAS NMR investigation, Inorg. Chem., 2003, 42(7), 2280–2287 CrossRef CAS PubMed.
- G. Sun, J. Young and R. Kirkpatrick, The Role of Al in C–S–H: NMR, XRD, and Compositional Results for Precipitated Samples, Cem. Concr. Res., 2006, 36(1), 18–29 CrossRef CAS PubMed.
- P. Faucon, A. Delagrave, J. C. Petit, C. Richet, J. M. Marchand and H. Zanni, Aluminum incorporation in calcium silicate hydrates (CSH) depending on their Ca/Si ratio, J. Phys. Chem. B, 1999, 103(37), 7796–7802 CrossRef CAS.
- M. Andersen, H. Jakobsen and J. Skibsted, A new aluminium-hydrate species in hydrated Portland cements characterized by 27Al and 29Si MAS NMR spectroscopy, Cem. Concr. Res., 2005, 36, 3–17 CrossRef PubMed.
- S. Komarneni, R. Roy, D. Roy, C. Fyfe, G. Kennedy, A. Bothner-By, J. Dadok and A. Chesnic, 27Al and 29Si magic angle spinning nuclear magnetic resonance spectroscopy of Al-substituted tobermorites, J. Mater. Sci., 1985, 20, 4209–4214 CrossRef CAS.
- M. Tsuji, S. Komarneni and P. Malla, Substituted tobermorites: 27Al and 29Si MAS NMR cation exchange, and water sorption studies, J. Am. Ceram. Soc., 1991, 74, 274–279 CrossRef CAS PubMed.
- F. Puertas, M. Palacios, H. Manzano, J. S. Dolado, A. Rico and J. Rodríguez, A model for the C–A–S–H gel formed in alkali-activated slag cements, J. Eur. Ceram. Soc., 2011, 31(12), 2043–2056 CrossRef CAS PubMed.
- R. Taylor, I. Richardson and R. Brydson, Composition and microstructure of 20-year-old ordinary Portland cement–ground granulated blast-furnace slag blends containing 0 to 100% slag, Cem. Concr. Res., 2010, 40(7), 971–983 CrossRef CAS PubMed.
- R. J. Pellenq, A. Kushima, R. Shahsavari, K. J. Van Vliet, M. J. Buehler and S. Yip, A realistic molecular model of cement hydrates, Proc. Natl. Acad. Sci. U. S. A., 2009, 106(38), 16102–16107 CrossRef CAS PubMed.
- M. Qomi, F. Ulm and R. Pellenq, Evidence on the Dual Nature of Aluminum in the Calcium–Silicate–Hydrates Based on Atomistic Simulations, J. Am. Ceram. Soc., 2012, 95(3), 1128–1137 CAS.
- I. Richardson, The importance of proper crystal-chemical and geometrical reasoning demonstrated using layered single and double hydroxides, Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater., 2013, 69, 150–162 CrossRef CAS PubMed.
- P. Faucon, J. C. Petit, T. Charpentier, J. F. Jacquinot and F. Adenot, Silicon substitution for aluminum in calcium silicate hydrates, J. Am. Ceram. Soc., 1999, 82(5), 1307–1312 CrossRef CAS PubMed.
- A. van Duin, A. Strachan, S. Stewman, Q. Zhang, X. Xu and W. Goddard, ReaxFFsio Reactive Force Field for Silicon and Silicon Oxide Systems, J. Phys. Chem. A, 2003, 107(19), 3803–3811 CrossRef CAS.
- D. W. Brenner, O. A. Shenderova, J. A. Harrison, S. J. Stuart, B. Ni and S. B. Sinnott, A second-generation reactive empirical bond order (REBO) potential energy expression for hydrocarbons, J. Phys.: Condens. Matter, 2002, 14(4), 783–802 CrossRef CAS.
- S. Leroch and M. Wendland, Simulation of Forces between Humid Amorphous Silica Surfaces: A Comparison of Empirical Atomic Force Fields, J. Phys. Chem. C, 2012, 116, 26247–26261 CAS.
- H. Manzano, E. Masoero, I. Arbeloa and H. Jennings, Molecular modelling of shear deformations in ordered and disordered calcium silicate hydrates, Soft Matter, 2013, 9(30), 7333–7341 RSC.
- T. Lau, A. Kushima and S. Yip, Atomistic Simulation of Creep in a Nanocrystal, Phys. Rev. Lett., 2010, 104(17), 175501 CrossRef.
- H. Manzano, R. Pellenq, F. Ulm and M. Buehler, Hydration of Calcium Oxide Surface Predicted by Reactive Force Field Molecular Dynamics, Langmuir, 2012, 28, 4187–4197 CrossRef CAS PubMed.
- L. Liu, A. Botero, W. Goddard and H. Sun, Development of a ReaxFF Reactive Force Field for Ettringite and Study of its Mechanical Failure Modes from Reactive Dynamics simulations, J. Phys. Chem. A, 2012, 116, 3918–3925 CrossRef CAS PubMed.
- D. Hou, H. Ma, Y. Zhu and Z. Li, Calcium silicate hydrate from dry to saturated state: structure, dynamics and mechanical properties, Acta Mater., 2014, 67, 81–94 CrossRef CAS PubMed.
- H. Manzano, S. Moeini, F. Marinelli, A. van Duin, F. Ulm and R. Pellenq, Confined Water Dissociation in Microporous Defective Silicates: Mechanism, Dipole Distribution, and Impact on Substrate Properties, J. Am. Chem. Soc., 2011, 134(4), 2208–2215 CrossRef PubMed.
- S. J. Murray, V. J. Subramani, R. P. Selvam and K. D. Hall, Molecular Dynamics to Understand the Mechanical Behavior of cement Paste, J. Transp. Res. Board, 2010, 2142(11), 75–82 CrossRef CAS PubMed.
- R. P. Selvam, V. G. Subramani, S. Murray and K. Hall, Potential Application of Nanotechnology on Cement Based materials, 2009 Search PubMed.
- X. Cong and R. Kirkpatrick, 29Si MAS NMR study of the structure of calcium silicate hydrate, Adv. Cem. Based Mater., 1996, 3(3–4), 144–156 CrossRef CAS.
- J. S. Dolado, M. Griebel and J. Hamaekers, A Molecular Dynamic Study of Cementitious Calcium Silicate Hydrate (C–S–H) gels, J. Am. Ceram. Soc., 2007, 90, 3938–3942 CAS.
- A. Brough, C. Dobson, I. Richardson and G. Groves, In situ solid-state NMR studies of Ca3SiO5: hydration at room temperature and at elevated temperatures using 29Si enrichment, J. Mater. Sci., 1994, 29(15), 3926–3940 CrossRef CAS.
- P. A. Bonnaud, Q. Ji, B. Coasne, R. J.-M. Pellenq and K. J. Van Vliet, Thermodynamics of Water Confined in Porous Calcium–Silicate–Hydrates, Langmuir, 2012, 28(31), 11422–11432 CrossRef CAS PubMed.
- J. Puibasset and R. Pellenq, Grand Canonical Monte Carlo Simulation Study of Water Adsorption in Silicalite at 300 K, J. Phys. Chem. B, 2008, 112(20), 6390–6397 CrossRef CAS PubMed.
- A. J. Allen, J. J. Thomas and H. M. Jennings, Composition and density of nanoscale calcium silicate hydrate in cement, Nat. Mater., 2007, 6, 311–316 CrossRef CAS PubMed.
- B. P. Feuston and S. H. Garofalini, Onset of polymerization in silica sols, Chem. Phys. Lett., 1990, 170(2), 264–270 CrossRef CAS.
- I. Richardson and G. Groves, The Structure of the Calcium Silicate Hydrate Phases Present in Hardened Pastes of White Portland Cement Blast-Furnace Slag Blends, J. Mater. Sci., 1997, 32(18), 4793–4802 CrossRef CAS.
- E. Kang, S. Lee and A. Hannon, Molecular dynamics simulations of calcium aluminate glasses, J. Non-Cryst. Solids, 2006, 352, 725–736 CrossRef CAS PubMed.
- A. Winkler, J. Horbach, W. Kob and K. Binder, Structure and diffusion in amorphous aluminum silicate: a molecular dynamics computer simulation, J. Chem. Phys., 2004, 120(1), 384–393 CrossRef CAS PubMed.
- M. Youssef, R. J. M. Pellenq and B. Yildiz, Glassy Nature of Water in an Ultraconfining Disordered Material: The Case of Calcium silicate hydrate, J. Am. Chem. Soc., 2011, 133(8), 2499–2510 CrossRef CAS PubMed.
- R. Shahsavari, M. J. Buechler, R. J. M. Pellenq and F. J. Ulm, First-principles study of elastic constants and interlayer interactions of complex hydrated oxides: case study of tobermorite and jennite, J. Am. Ceram. Soc., 2009, 92(10), 2323–2330 CrossRef CAS PubMed.
- G. Costantinide and F. Ulm, The nanogranular nature of C–S–H, J. Mech. Phys. Solids, 2006, 55(1), 64–90 CrossRef PubMed.
- S. Merlino, E. Bonnacorsi and T. Armbruster, The real structure of tobermorite 11 Å: normal and anomalous forms, OD character and polyptic modifications, Eur. J. Mineral., 2001, 13(3), 577–590 CrossRef CAS.
- D. Hou, H. Ma, Z. Li and Z. Jin, Molecular simulation of “hydrolytic weakening”: a case study on silica, Acta Mater., 2014, 80, 264–277 CrossRef CAS PubMed.
- T. Zhu, J. Li, X. Lin and S. Yip, Stress-dependent molecular pathways of silica–water reaction, J. Mech. Phys. Solids, 2005, 53(7), 1597–1623 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |