Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Short and efficient synthesis of fluorinated δ-lactams†
Thomas J.
Cogswell
a,
Craig S.
Donald
b,
De-Liang
Long‡
a and
Rodolfo
Marquez§
*a
aSchool of Chemistry, University of Glasgow, Glasgow, G12 8QQ, UK. E-mail: rudi.marquez@glasgow.ac.uk; Fax: +44 (0)141 330 4888; Tel: +44 (0)141 330 5953
bOncology iMed, AstraZeneca, Alderley Park, Macclesfield, Cheshire SK10 4TG, UK
First published on 29th August 2014
Abstract
The diastereoselective synthesis of fluorinated δ-lactams has been achieved through an efficient five step process. The route can tolerate a range of functionalities, and provides a quick route for the generation of new fluorinated medicinal building blocks.
Introduction
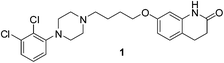
Fluorination has been used to increase the potency and ‘drug-like’ nature of active compounds for many years, making organofluorine compounds a cornerstone in medicinal chemistry.1 Fluorination has also been extensively used to alter the basicity of N-heterocycles, and as a tool through which hydrogen bonding can be probed and assessed.2 As such, new ways to selectively incorporate fluorine atoms into different substructures is an ever expanding area of research.1,2δ-Lactams on the other hand are also present widely in medicinal chemistry and natural systems. For instance, aripiprazole 1 is used currently for the treatment of schizophrenia and bipolar disorders (Fig. 1).3
Thus, it is not surprising that considerable time and efforts have been directed towards the synthesis of fluorinated δ-lactams.4–6 However to date, there are a relatively small number of examples and few general routes available to enable synthesis of compound libraries to drive structure–activity relationship understanding in medicinal chemistry programmes.
Herein, we would like to report a short and efficient route to the synthesis of fluorinated δ-lactams starting from commercial aldehydes. The methodology is amenable for the generation of a wide range of δ-lactams, and has the potential to allow entry into several different classes of N-heterocyclic compounds.
Results and discussion
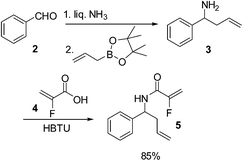
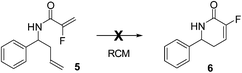
Our initial studies towards the synthesis of fluorinated δ-lactams began with benzaldehyde, which upon amino allylation under Kobayashi conditions afforded amine 3.7 Amide coupling of amine 3 with 2-fluoroacrylic acid 4 then afforded the desired diene 5 in 85% yield over the 2 steps (Scheme 1).Amide 5 was then treated with a range of metathesis catalysts in an attempt to induce ring closure (Scheme 2), however only a complex mixture of cross metathesis products resulted, with no desired cyclised product 6 being detected.
The lack of cyclisation could be attributed to the interaction of the ruthenium catalyst with the amide group which prevents the ring closing metathesis from taking place, in a situation similar to that reported by Vilar and co-workers.8
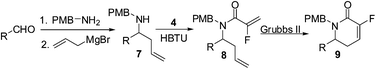
To test this hypothesis, the route was altered to incorporate a protecting group on the amide to prevent the amide from interacting with the Ru catalyst. The protecting group of choice was the p-methoxybenzyl group due to its electron donating properties, and the ease of removal via oxidative cleavage.9
Thus, reaction of p-methoxybenzylamine with benzaldehyde yielded the corresponding imine which upon allyl Grignard addition produced the secondary amine 7a in near quantitative yield. HBTU promoted coupling between the amine 7a and 2-fluoroacrylic acid 4 and then proceeded to generate the desired amide 8a in good yield.
Gratifyingly, PMB protection of the amide unit allowed the ring closing metathesis reaction to proceed smoothly to yield the unsaturated lactam 9a in quantitative yield (Table 1).
| R | Yield | Yield | Yield | |||
|---|---|---|---|---|---|---|
| Ph | 95% | 7a | 63% | 8a | Quant | 9a |
| 4-MeOPh | 80% | 7b | 66% | 8b | Quant | 9b |
| 4-CF3Ph | 66% | 7c | 45% | 8c | Quant | 9c |
| 1-Naphthyl | 99% | 7d | 31% | 8d | Quant | 9d |
| 4-BrPh | 97% | 7e | 54% | 8e | 99% | 9e |
| i-Butyl | 76% | 7f | 74% | 8f | 85% | 9f |
| Cyclohexyl | 74% | 7g | 70% | 8g | 89% | 9g |
| 2-Furyl | 83% | 7h | 28% | 8h | 64% | 9h |
| N-Ts 2-pyrrole | 85% | 7i | 50% | 8i | 77% | 9i |
| 2-Pyridyl | 73% | 7j | 68% | 8j | — | 9j |
With the benzaldehyde example working efficiently, the scope of the methodology was explored (Table 1). Electron donating and electron withdrawing substituents on the aromatic ring were both tested with good results in all cases through the four step sequence. The electron withdrawing analogue being slightly less efficient in the first three steps, however, the RCM reaction produced the desired unsaturated lactam 9c in quantitative yield.
Aliphatic substrates are also well tolerated, and afforded the corresponding fluorinated lactams 9f, 9g cleanly and in good yield over the 4 step sequence.
Finally, furan, pyrrole and pyridine frameworks were also explored due to their widespread use in medicinal and biological chemistry. Furfural and N-tosylpyrrole carboxaldehyde proceeded to generate the structurally interesting fluorinated δ-lactams 9h, 9i in good yield. In the case of the pyridine unit, the amino allylation and amide coupling proceeded in reasonable yields, affording intermediates 7j and 8j respectively. Unfortunately, the ring closing metathesis failed to generate the desired fluorinated δ-lactam 9j under any of the conditions attempted. This was rationalised in the same manner as the previous example (Scheme 2), where an unprotected nitrogen atom was thought to bind to the Ru catalyst.
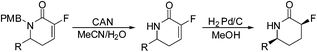
With the fluorinated ring system in place, the removal of the p-methoxybenzyl protecting group and reduction of the olefin unit were explored. Removal of the PMB group was achieved with cerium ammonium nitrate to yield the desired fluoro-pyridones 10a–g in variable yields (Table 2). Unfortunately, treatment of the furyl and pyrrole substituted pyridones 9h–i with CAN failed to yield the desired unprotected pyridones, resulting instead in substrate decomposition.
Hydrogenation of the fluoro-olefin unit, on the other hand, proceeded in near quantitative yield in most cases, and with complete diastereoselectivity to yield the desired fluorinated δ-lactams 11a–g. The only exception was the brominated analogue 10e, which unsurprisingly yielded the unbrominated lactam 11a upon hydrogenation in excellent yield.
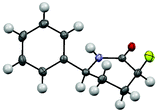
The syn selectivity of hydrogenation, and hence the stereochemistry of the newly formed C3 stereocentre, is dictated by the C6 substituent. The relative stereochemistry was corroborated by X-ray crystallography (Fig. 2).10
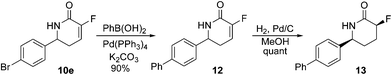
The bromo-lactam intermediate 10e, on the other hand, presented us with an opportunity to further explore the scope of the fluoro-lactam intermediates as medicinal chemistry building blocks. Thus treatment of fluoro-lactam 10e under Suzuki–Miyaura conditions with phenylboronic acid generated the tricyclic unit 12 in near quantitative yield.11
Alkene reduction under standard conditions then generated the desired fluoro-lactam unit 13 in excellent yield and with complete diastereocontrol (Scheme 3).
Conclusions
In conclusion, we have developed a fast and reliable approach to the synthesis of a number of fluorinated δ-lactams in an efficient 5 step process. Work is now underway to expand the methodology to allow access to other fluorinated ring systems including pyridines and pyrroles as well as developing an enantioselective variant of this methodology.Experimental
All reactions were performed in oven-dried glassware under an inert argon atmosphere unless otherwise stated. Tetrahydrofuran (THF), diethyl ether, toluene and dichloromethane were purified through a solvent purification system. Petroleum ether refers to the fraction boiling between 40 and 60 °C. All reagents were used as received, unless otherwise stated. Solvents were evaporated under reduced pressure at 40 °C unless otherwise stated. IR spectra were recorded as thin films on NaCl plates using a Fourier Transform spectrometer. Only significant absorptions (νmax) are reported in wavenumbers (cm−1). Proton magnetic resonance spectra (1H NMR), fluorine magnetic resonance spectra (19F NMR) and carbon magnetic resonance spectra (13C NMR) were recorded at 400 MHz, 377 MHz and 100 MHz or at 500 MHz, 470 MHz and 125 MHz respectively. 13C NMR spectra were recorded with 1H noise decoupling. Chemical shifts (δ) are reported in parts per million (ppm) and are referenced to the residual solvent peak. The order of citation in parentheses is (1) the number of equivalent nuclei (by integration), (2) multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, quint = quintet, sext = sextet, sept = septet, m = multiplet, b = broad), (3) and the coupling constant (J) quoted in hertz to the nearest 0.1 Hz. High resolution mass spectra were obtained by electrospray (EI) chemical ionisation (CI) mass spectrometry operating at a resolution of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 full widths at half height. Flash chromatography was performed using silica gel (40–63 micron) as the stationary phase. TLC was performed on aluminium sheets pre-coated with silica (Silica Gel 60 F254) unless otherwise stated. The plates were visualised by the quenching of UV fluorescence (λmax 254 nm) and/or by staining with either anisaldehyde, potassium permanganate, iodine or cerium ammonium molybdate followed by heating.
000 full widths at half height. Flash chromatography was performed using silica gel (40–63 micron) as the stationary phase. TLC was performed on aluminium sheets pre-coated with silica (Silica Gel 60 F254) unless otherwise stated. The plates were visualised by the quenching of UV fluorescence (λmax 254 nm) and/or by staining with either anisaldehyde, potassium permanganate, iodine or cerium ammonium molybdate followed by heating.
1-Phenylbut-3-en-1-ylamine, 312
Benzaldehyde (0.21 g, 2.0 mmol) was dissolved in methanol (4 mL) and the resulting solution was cooled to −78 °C. NH3 (ca. 4 mL) was condensed into the solution, and the resulting reaction mixture was warmed to −10 °C and stirred until the excess ammonia had evaporated (3 h). Allylboronic pinacol ester (0.76 mL, 4.0 mmol) was added and the reaction was stirred for 2 h. The reaction vessel was then allowed to warm up to room temperature and stirred for a further 1 h. Aq. HCl (6 M) was added slowly to the solution until pH 1 and the mixture was extracted with diethyl ether (3 × 20 mL). The aqueous phase was collected, and aq. NaOH (2 M) was added slowly until pH 14. The aqueous solution was extracted with CH2Cl2 (3 × 20 mL). The combined CH2Cl2 phases were then dried (Na2SO4), filtered and evaporated in vacuo to yield amine 3 as a colourless oil (0.28 g, 1.9 mmol, 95%) which was of sufficient purity to enable the substance to be used without further purification.1H (CDCl3, 400 MHz) δ: 7.27–7.15 (5H, m), 5.68–5.63 (1H, m), 5.06–5.00 (2H, m), 3.91 (1H, dd, JHH = 8.0, 5.2 Hz) 2.40–2.27 (2H, m), 1.60 (2H, br s). 13C (CDCl3, 125 MHz) δ: 145.8, 135.4, 128.4, 127.9, 126.4, 117.7, 55.4, 44.2.
2-Fluoroacrylic acid, 413
Commercially available 2-fluoroacrylic acid methyl ester (0.87 mL, 9.6 mmol) was dissolved in EtOH–H2O (8.7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.3, 10 mL). Aq. NaOH (2 M) was then added dropwise until pH 11 was reached, and the resulting mixture was stirred for 30 min. After which, the solution was evaporated to dryness under vacuum to yield the sodium salt as a white solid. Diethyl ether (20 mL) was added to the salt, followed by aq. HCl (6 M) dropwise until the solid dissolved. The layers were then separated, and the aqueous layer was extracted with diethyl ether (10 mL). The organics were combined, dried over sodium sulfate and evaporated in vacuo to yield the desired acid 4 as a white solid (0.74 g, 8.0 mmol, 87%) with no need for further purification.
1.3, 10 mL). Aq. NaOH (2 M) was then added dropwise until pH 11 was reached, and the resulting mixture was stirred for 30 min. After which, the solution was evaporated to dryness under vacuum to yield the sodium salt as a white solid. Diethyl ether (20 mL) was added to the salt, followed by aq. HCl (6 M) dropwise until the solid dissolved. The layers were then separated, and the aqueous layer was extracted with diethyl ether (10 mL). The organics were combined, dried over sodium sulfate and evaporated in vacuo to yield the desired acid 4 as a white solid (0.74 g, 8.0 mmol, 87%) with no need for further purification.
1H (CDCl3, 400 MHz) δ: 11.51 (1H, br s), 5.92 (1H, dd, JHF = 42.8, JHH = 3.6 Hz), 5.52 (1H, dd, JHF = 12.4, JHH = 3.2 Hz). 19F (CDCl3, 470 MHz) δ: −118.3. 13C (CDCl3, 125 MHz) δ: 165.4 (d, JCF = 46.3 Hz), 151.3 (d, JCF = 323.8 Hz), 105.2 (d, JCF = 18.8 Hz).
2-Fluoro-N-(1-phenyl-but-3-enyl)-propenamide, 5
A solution of 2-fluoroacrylic acid 4 (0.41 g, 4.6 mmol) in CH2Cl2 (25 mL) was treated with HBTU (2.59 g, 6.5 mmol) and was then cooled down to 0 °C. DIPEA (1.19 mL, 6.5 mmol) and amine 3 (0.80 g, 5.4 mmol) were sequentially added, and the reaction was stirred at room temperature until completion by TLC analysis (1 h). The solvent was removed under reduced pressure, and the resulting residue was purified by flash column chromatography (0–5% EtOAc in petroleum ether) to yield amide 5 as a white solid (0.89 g, 4.0 mmol, 89%). m.p. 63–65 °C.1H (CDCl3, 400 MHz) δ: 7.28–7.16 (5H, m), 6.48 (1H, br s), 5.66–5.55 (2H, m), 5.09–5.03 (4H, m), 2.55 (2H, t, JHH = 6.8 Hz). 19F (CDCl3, 470 MHz) δ: −121.3. 13C (CDCl3, 125 MHz) δ: 158.8 (d, JCF = 30.0 Hz), 156.2 (d, JCF = 268.8 Hz) 140.8, 133.4, 128.8, 127.7, 126.5, 118.3, 99.1 (d, JCF = 15.0 Hz), 52.6, 40.3. m/z [CI (+ve)] 220.3 [M + H]+, HRMS found [M + H]+ 220.1135, C13H15FNO requires 220.1138. IR (thin film) νmax = 3338, 1651, 1529, 1190 cm−1.
1-(Toluene-4′-sulfonyl)-1H-pyrrol-2-carboxaldehyde14
Pyrrole-2-carboxyaldehyde (1.00 g, 10.5 mmol) was dissolved in THF (20 mL) and the solution was cooled down to 0 °C. NaH (0.62 g, 60% in oil, 15.0 mmol) was added slowly, and the resulting mixture was stirred at 0 °C for 10 min. Tosyl chloride (3.0 g, 15.0 mmol) was added, and the reaction mixture was stirred for a further 15 min at 0 °C before being allowed to warm up to room temperature and stirred overnight. The reaction mixture was quenched with H2O (20 mL), and extracted with CH2Cl2 (2 × 25 mL). The combined organics were then dried (Na2SO4) and the solvent was removed in vacuo. The resulting crude residue was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the tosylated pyrrole as a white solid (2.47 g, 9.9 mmol, 94%).1H (CDCl3, 400 MHz) δ: 10.01 (1H, s), 7.83 (2H, d, JHH = 8.3 Hz), 7.65 (1H, dd, JHH = 3.0, 1.8 Hz), 7.35 (2H, d, JHH = 8.3 Hz), 7.12 (1H, dd, JHH = 3.7, 1.8 Hz), 6.43 (1H, br appt, JHH = 3.4 Hz), 2.45 (3H, s). 13C (CDCl3, 125 MHz) δ: 178.9, 145.9, 135.3, 133.6, 130.1, 129.5, 127.6, 124.4, 112.4, 21.7.
General procedure A: synthesis of PMB protected allylic amines from aldehydes
Na2SO4 (2 g) was dried under vacuum in a round bottom flask for 10 min. The aldehyde (1 eq.) was then added, followed by toluene (15 mL) and 4-methoxybenzylamine (1.1 eq.). The resulting reaction mixture was then heated to reflux for 3 h. The reaction was then cooled down to room temperature, and the solid residue was filtered off. The solution was concentrated under vacuum and the residue was re-dissolved in anhydrous diethyl ether (20 mL). The solution was placed under argon and was cooled down to 0 °C. The solution was then treated dropwise with allylmagnesium bromide (1.5 eq.) and the resulting mixture was allowed to warm up to room temperature overnight. The reaction was quenched with water (20 mL) and extracted with diethyl ether (3 × 20 mL). The combined organic extracts were dried over sodium sulfate, and evaporated under reduced pressure. The crude residue was purified by flash column chromatography to afford the corresponding allylic amine.4′-Methoxy-N-(1-phenyl-3-butenyl)benzylamine, 7a15
Following general procedure A, benzaldehyde (0.95 mL, 9.4 mmol) was reacted with 4-methoxybenzylamine (1.4 mL, 10.4 mmol) and allylmagnesium bromide (14.3 mL 1.0 M in THF, 14.3 mmol). The crude residue was purified by flash column chromatography (0–2.5% EtOAc in petroleum ether) to yield the expected amine 7a (2.39 g, 8.9 mmol, 95% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.40–7.36 (4H, m,), 7.30–7.28 (1H, m), 7.20 (2H, d, JHH = 8.6 Hz), 6.88 (2H, d, JHH = 8.6 Hz), 5.75–5.70 (1H, m), 5.11–5.70 (2H, m), 3.83 (3H, s), 3.71 (1H, dd, JHH = 7.8, 5.9 Hz), 3.64 (1H, d, JHH = 13.2 Hz), 3.49 (1H, d, JHH = 13.2 Hz,), 2.44–2.41 (2H, m), 1.73 (1H, br s). 13C (CDCl3, 125 MHz) δ: 158.5, 143.9, 135.5, 132.8, 129.3 (2C), 128.4 (2C), 127.3 (2C), 127.0 (2C), 117.5, 113.7, 61.5, 55.3, 50.8, 43.1.
4′-Methoxy-N-[1-(4′′-methoxyphenyl)-3-butenyl]benzylamine, 7b16
Following general procedure A, 4-methoxybenzaldehyde (0.84 mL, 7.3 mmol) was reacted with 4-methoxybenzylamine (1.1 mL, 8.0 mmol) and allylmagnesium bromide (11.0 mL 1.0 M in THF, 11.0 mmol). The crude residue was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the expected amine 7b (1.67 g, 5.6 mmol, 77% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.30 (2H, d, JHH = 8.8 Hz), 7.19 (2H, d, JHH = 8.6 Hz), 6.92 (2H, d, JHH = 8.8 Hz), 6.87 (2H, d, JHH = 8.6 Hz), 5.75–5.68 (1H, m), 5.12–5.04 (2H, m), 3.85 (3H, s) 3.82 (3H, s), 3.68–3.60 (2H, m), 3.47 (1H, d, JHH = 12.9 Hz), 2.43–2.39 (2H, m). 13C (CDCl3, 125 MHz) δ: 158.6, 158.5, 135.9, 135.6 (2C), 132.8, 129.3 (2C), 128.3 (2C), 117.4, 113.7 (2C), 60.81, 55.3, 55.2, 50.7, 43.2.
4′-Methoxy-N-[1-(4′′-trifluoromethanephenyl)-3-butenyl]benzylamine, 7c
Following general procedure A, 4-(trifluoromethyl)benzaldehyde (0.78 mL, 5.7 mmol) was reacted with 4-methoxybenzylamine (0.83 mL, 6.3 mmol) and allylmagnesium bromide (8.6 mL 1.0 M in THF, 8.6 mmol). The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the expected amine 7c (1.27 g, 3.8 mmol, 66% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.62 (2H, d, JHH = 8.2 Hz), 7.51 (2H, d, JHH = 8.2 Hz), 7.18 (2H, d, JHH = 8.8 Hz), 6.88 (2H, d, JHH = 8.8 Hz), 5.75–5.65 (1H, m), 5.12–5.08 (2H, m), 3.83 (3H, s), 3.77 (1H, dd, JHH = 7.6, 5.6 Hz), 3.64 (1H, d, JHH = 13.2 Hz), 3.45 (1H, d, JHH = 13.2 Hz), 2.45–2.34 (2H, m). 19F (CDCl3, 377 MHz) δ: −62.3. 13C (CDCl3, 125 MHz) δ: 158.7, 148.2, 134.8, 132.4, 129.8 (2C), 127.7 (2C), 125.7, 125.4 (2C), 125.3, 118.2, 113.8 (2C), 61.2, 55.3, 44.2, 33.6. m/z [CI (+ve)] 336.1 [M + H]+. HRMS found [M + H]+ 336.1572, C19H21F3NO requires 336.1575. IR (thin film) νmax = 2935, 1612, 1512, 1323, 1246, 1120, 1066 cm−1.
4′-Methoxy-N-[1-(naphthalen-1′′-yl)-3-butenyl]benzylamine, 7d
Following general procedure A, 1-naphthaldehyde (0.86 mL, 6.40 mmol) was reacted with 4-methoxybenzylamine (0.84 mL, 7.0 mmol) and allylmagnesium bromide (9.6 mL 1.0 M in THF, 9.6 mmol). The crude product was purified by flash column chromatography (0–10% diethyl ether in petroleum ether) to yield the desired amine 7d (2.03 g, 6.3 mmol, 99% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 8.10 (1H, appd, JHH = 7.4 Hz), 7.81–7.79 (1H, m), 7.72–7.67 (2H, m), 7.44–7.38 (3H, m), 7.09 (2H, d, JHH = 8.6 Hz), 6.76 (2H, d, JHH = 8.6 Hz), 5.75–5.67 (1H, m), 5.05–4.97 (2H, m), 3.71 (3H, s), 3.60 (1H, d, JHH = 13.0 Hz), 3.44 (1H, d, JHH = 13.0 Hz), 2.59–2.54 (1H, m), 2.41–2.35 (1H, m). 13C (CDCl3, 125 MHz) δ: 158.7, 139.7, 138.9, 135.8, 134.1, 132.9, 131.7, 129.4 (2C), 129.0, 127.4, 125.7, 125.3, 123.9, 123.1, 117.7, 113.8 (2C), 56.9, 55.4, 51.1, 42.2. m/z [CI (+ve)] 318.2 [M + H]+, HRMS found [M + H]+ 318.1862, C22H24NO requires 318.1858. IR (thin film) νmax = 2960, 1511, 1246, 1035 cm−1.
4′-Methoxy-N-[1-(4′′-bromophenyl)-3-butenyl]benzylamine, 7e
Following general procedure A, 4-bromobenzaldehyde (1 g, 5.4 mmol) was reacted with 4-methoxybenzylamine (0.74 mL, 5.4 mmol) and allylmagnesium bromide (8.1 mL 1.0 M in THF, 8.1 mmol). The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired amine 7e (1.82 g, 5.25 mmol, 97% yield) as a pale yellow oil.1H (CDCl3, 500 MHz) δ: 7.48 (2H, d, JHH = 8.4 Hz), 7.27 (2H, d, JHH = 8.4 Hz), 7.15 (2H, d, JHH = 9.1 Hz), 6.87 (2H, d, JHH = 9.1 Hz), 5.72–5.63 (1H, m), 5.10–5.05 (2H, m), 3.82 (3H, s), 3.66 (1H, dd, JHH = 7.8, 6.3 Hz), 3.61 (1H, d, JHH = 13.1 Hz), 3.46 (1H, d, JHH = 13.1 Hz), 2.40–2.35 (2H, m). 13C (CDCl3, 125 MHz) δ: 158.7, 143.3, 135.1, 132.6, 131.6 (2C), 129.3 (2C), 129.1 (2C), 120.4, 117.9, 113.9 (2C), 60.7, 55.3, 50.9, 42.7. m/z [CI (+ve)] 345.8 M+, HRMS found [M + H]+ 346.0804, C18H2179BrNO requires 346.0807. IR (thin film) νmax = 2945, 2835, 1511, 1245, 1035, 1009 cm−1.
4′-Methoxy-N-(1-isobutyl-3-butenyl)benzylamine, 7f
Following general procedure A, isovaleraldehyde (1.25 mL, 11.6 mmol) was reacted with 4-methoxybenzylamine (1.7 mL, 12.7 mmol) and allylmagnesium bromide (17.4 mL 1.0 M in THF, 17.4 mmol). The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield 7f (2.06 g, 8.4 mmol, 76% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.26 (2H, d, JHH = 8.8 Hz), 6.86 (2H, d, JHH = 8.8 Hz), 5.82–5.70 (1H, m), 5.14–5.00 (2H, m), 3.81 (3H, s), 3.73 (1H, quint, JHH = 6.4 Hz), 2.70–2.65 (1H, m), 2.25–2.03 (2H, m), 1.62 (1H, appsept, JHH = 6.8 Hz), 1.46–1.23 (2H, m), 0.90 (3H, d, JHH = 6.6 Hz), 0.87 (3H, d, JHH = 6.6 Hz). 13C (CDCl3, 100 MHz) δ: 158.5, 135.8, 133.0, 129.3 (2C), 118.7, 113.7 (2C), 55.3, 54.0, 50.5, 41.2, 38.6, 24.7, 22.5. m/z [CI (+ve)] 248.2 [M + H]+, HRMS found [M + H]+ 248.2018, C16H26NO requires 248.2014. IR (thin film) νmax = 2953, 2906, 1612, 1512, 1464, 1246 cm−1.
4′-Methoxy-N-(1-cyclohexyl-3-butenyl)benzylamine, 7g
Following general procedure A, cyclohexanecarboxaldehyde (1.08 mL, 8.87 mmol) was reacted with 4-methoxybenzylamine (1.2 mL, 9.8 mmol) and allylmagnesium bromide (13.3 mL 1.0 M in THF, 13.3 mmol). The crude product was purified by flash column chromatography (0–2.5% diethyl ether in petroleum ether) to yield the desired amine 7g (1.81 g, 6.6 mmol, 74% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.26 (2H, d, JHH = 8.6 Hz), 6.87 (2H, d, JHH = 8.6 Hz), 5.85–5.75 (1H, m), 5.11–5.06 (2H, m), 3.82 (3H, s), 3.71 (2H, s), 2.42–2.38 (1H, m), 2.32–2.26 (1H, m), 2.16–2.09 (1H, m), 1.81–1.70 (4H, m), 1.47–1.43 (1H, m), 1.31–1.18 (4H, m,), 1.10–1.00 (2H, m). 13C (CDCl3, 125 MHz) δ: 158.5, 136.8, 133.3, 129.3 (2C), 116.8, 113.7 (2C), 61.1, 55.3, 51.3, 40.6, 35.3, 29.4, 28.9, 26.8, 26.7, 26.7. m/z [CI (+ve)] 274.2 [M + H]+, HRMS found [M + H]+ 274.2171, C18H28NO requires 274.2168. IR (thin film) νmax = 2924, 1511, 1246, 1037 cm−1.
4′-Methoxy-N-[1-(furan-2′′-yl)-3-butenyl]benzylamine, 7h
Following general procedure A, 2-furaldehyde (0.86 mL, 10.4 mmol) was reacted with 4-methoxybenzylamine (1.3 mL, 11.4 mmol) and allylmagnesium bromide (15.6 mL 1.0 M in THF, 15.6 mmol). The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired amine 7h (2.21 g, 8.59 mmol, 83% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.42 (1H, dd, JHH = 1.8, 0.8 Hz), 7.23 (2H, d, JHH = 8.7 Hz), 6.88 (2H, d, JHH = 8.7 Hz), 6.36 (1H, dd, JHH = 3.7, 1.8 Hz), 6.21 (1H, d, JHH = 3.7 Hz), 5.79–5.62 (1H, m), 5.14–5.06 (2H, m), 3.83 (3H, s), 3.79 (1H, t, JHH = 6.8 Hz), 3.72 (1H, d, JHH = 13.0 Hz), 3.56 (1H, d, JHH = 13.0 Hz), 2.58–2.53 (2H, m). 13C (CDCl3, 125 MHz) δ: 158.6, 156.3, 141.5, 134.9, 132.4, 129.4 (2C), 117.5, 113.8 (2C), 109.9, 106.6, 55.3, 54.7, 50.5, 39.3. m/z [CI (+ve)] 258.2 [M + H]+, HRMS found [M + H]+ 258.1492, C16H20NO2 requires 258.1494. IR (thin film) νmax = 2930, 2850, 1511, 1441, 1246, 1035 cm−1.
4′-Methoxy-N-[1-(1′′-(toluene-4′′′-sulfonyl)-1H-pyrrol-2′′-yl)-3-butenyl]benzylamine, 7i
Following general procedure A, 1-(toluene-4′-sulfonyl)-1H-pyrrol-2-carboxaldehyde (1 g, 4.02 mmol) was reacted with 4-methoxybenzylamine (0.53 mL, 4.4 mmol) and allylmagnesium bromide (6.0 mL 1.0 M in THF, 6.0 mmol). The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired amine 7i (1.4 g, 3.42 mmol, 85% yield) as a brown oil.1H (CDCl3, 500 MHz) δ: 7.62 (2H, d, JHH = 8.3 Hz), 7.36 (1H, dd, JHH = 3.2, 1.7 Hz), 7.25 (2H, d, JHH = 8.3 Hz), 7.15 (2H, d, JHH = 8.6 Hz), 6.84 (2H, d, JHH = 8.6 Hz), 6.35–6.33 (1H, m), 6.30 (1H, t, JHH = 3.3 Hz), 5.73–5.63 (1H, m), 5.05–4.98 (2H, m), 4.22 (1H, dd, JHH = 7.3, 5.1 Hz), 3.84 (3H, s), 3.43 (1H, d, JHH = 12.6 Hz), 3.25 (1H, d, JHH = 12.6 Hz), 2.47–2.30 (2H, m), 2.41 (3H, s). 13C (CDCl3, 125 MHz) δ: 158.6, 144.9, 137.6, 136.7, 135.0, 132.6, 129.9 (2C), 129.1 (2C), 126.7 (2C), 123.1, 117.5, 114.1 (2C), 112.7, 111.6, 55.3, 54.0, 50.1, 40.7, 21.7. m/z [ESI] 433.1 [M + Na]+, HRMS found [M + Na]+ 433.1539, C23H26N2O3SNa requires 433.1556. IR (thin film) νmax = 2975, 1512, 1247, 1172 cm−1.
4′-Methoxy-N-[1-(pyridin-2′′-yl)-3-butenyl]benzylamine, 7j
Following general procedure A, pyridine-2-carboxaldehyde (0.89 mL, 9.30 mmol) was reacted with 4-methoxybenzylamine (1.2 mL, 10.2 mmol) and allylmagnesium bromide (14.0 mL 1.0 M in THF, 14.0 mmol). The crude product was purified by flash column chromatography (0–25% diethyl ether in petroleum ether) to yield the desired amine 7j (1.82 g, 6.78 mmol, 73% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 8.51 (1H, dq, JHH = 4.8, 0.9 Hz), 7.58 (1H, td, JHH = 7.7, 1.8 Hz), 7.30 (1H, d, JHH = 7.7 Hz), 7.12 (2H, d, JHH = 8.6 Hz), 7.08 (1H, ddd, JHH = 7.4, 4.8, 1.1 Hz), 6.76 (2H, d, JHH = 8.6 Hz), 5.70–5.60 (1H, m), 4.99–4.92 (2H, m), 3.75 (1H, dd, JHH = 7.8, 5.9 Hz), 3.71 (3H, s), 3.54 (1H, d, JHH = 12.9 Hz), 3.43 (1H, d, JHH = 12.9 Hz), 2.50–2.43 (1H, m), 2.39–2.32 (1H, m). 13C (CDCl3, 125 MHz) δ: 163.3, 158.5, 149.4, 136.3, 135.3, 132.6, 129.3 (2C), 121.9 (2C), 117.5, 113.7 (2C), 62.8, 55.3, 51.1, 41.6. m/z [CI (+ve)] 269.1 [M + H]+, HRMS found [M + H]+ 269.1653, C17H21N2O requires 269.1654. IR (thin film) νmax = 2836, 1512, 1247, 905 cm−1.
General procedure B: amide coupling of allylic amine
2-Fluoroacrylic acid (1.5 eq.) and HBTU (1.5 eq.) were dry mixed and then dissolved in CH2Cl2 (20 mL). DIPEA (1.5 eq.) was added followed by the corresponding amine (1 eq.). The resulting solution was stirred and refluxed for 17 h. The reaction was cooled down to room temperature and the solvent was then evaporated under reduced pressure. The crude material was purified by flash column chromatography.2′-Fluoro-N-(4′′-methoxybenzyl)-N-(1-phenyl-3-butenyl)acrylamide, 8a
Amine 7a (0.5 g, 1.8 mmol) was coupled with 2-fluoroacrylic acid (0.25 g, 2.8 mmol) using HBTU (1.1 g, 2.8 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired dialkene 8a (0.38 g, 1.10 mmol, 63% yield) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.36–7.32 (5H, m), 6.97 (2H, d, JHH = 8.6 Hz), 6.76 (2H, d, JHH = 8.3 Hz), 5.75–5.66 (1H, m), 5.35 (1H, br s), 5.26 (1H, br s), 5.08–4.99 (3H, m), 4.47 (1H, d, JHH = 15.7 Hz), 4.16 (1H, br s), 3.78 (3H, s), 2.74 (2H, br s). 19F (CDCl3, 470 MHz) δ: −102.4, −114.2. 13C (CDCl3, 125 MHz) δ: 163.3 (d, JCF = 30.0 Hz), 158.7, 158.2 (d, JCF = 271.3), 138.1, 134.2, 134.1 (2C), 129.2 (2C), 128.6 (2C) 128.1 (2C), 118.1, 113.6 (2C), 99.4 (d, JCF = 16.3 Hz), 55.2 (2C), 35.7, 33.5. m/z [EI (+ve)] 339.2 [M]+, HRMS found [M]+ 339.1639, C21H22FNO2 requires 339.1635. IR (thin film) νmax = 2937, 1637, 1512, 1417, 1246, 1176, 1033 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(4′′′-methoxyphenyl)-3-butenyl]acrylamide, 8b
Amine 7b (0.55 g, 1.8 mmol) was coupled with 2-fluoroacrylic acid (0.25 g, 2.8 mmol) using HBTU (1.1 g, 2.8 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired dialkene 8b (0.44 g, 1.2 mmol, 66%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.27 (2H, d, JHH = 8.6 Hz), 6.98 (2H, d, JHH = 8.6 Hz), 6.89 (2H, d, JHH = 8.4 Hz), 6.77 (2H, d, JHH = 8.4 Hz), 5.74–5.64 (1H, m), 5.35 (1H, br s), 5.23 (1H, br s), 5.07–4.92 (3H, m), 4.46 (1H, d, JHH = 15.5 Hz), 4.14 (1H, br s), 3.83 (3H, s), 3.78 (3H, s), 2.69 (2H, br s). 19F (CDCl3, 470 MHz) δ: −102.1, −104.2. 13C (CDCl3, 125 MHz) δ: 163.2 (d, JCF = 30.0 Hz), 159.4, 159.3, 158.7, 158.5 (d, JCF = 271.3), 134.2, 134.1, 129.8 (2C), 129.2 (2C), 118.0, 113.9 (2C), 113.6 (2C), 99.2 (d, JCF = 15.0 Hz), 65.9, 55.3, 55.2, 36.04, 33.5. m/z [EI (+ve)] 369.2 [M]+, HRMS found [M]+ 369.1743, C22H24FNO3 requires 369.1740. IR (thin film) νmax = 2933, 2837, 1635, 1512, 1246, 1178, 1033 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(4′′′-trifluoromethanephenyl)-3-butenyl]acrylamide, 8c
Amine 7c (0.64 g, 1.8 mmol) was coupled with 2-fluoroacrylic acid (0.25 g, 2.8 mmol) using HBTU (1.1 g, 2.8 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired dialkene 8c (0.33 g, 0.80 mmol, 45%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.57 (2H, d, JHH = 8.0 Hz), 7.44 (2H, d, JHH = 8.0 Hz), 6.98 (2H, d, JHH = 8.4 Hz), 6.76 (2H, d, JHH = 8.4 Hz), 5.76–5.66 (1H, m), 5.41 (1H, d, JHH = 3.2 Hz), 5.29 (1H, d, JHH = 3.2 Hz), 5.14–5.09 (3H, m), 4.43 (1H, d, JHH = 15.6 Hz), 4.34 (1H, br. s), 3.80 (3H, s), 2.79 (2H, br. s). 19F (CDCl3, 377 MHz) δ: −62.3, −102.5, −104.4. 13C (CDCl3, 125 MHz) δ: 163.4 (d, JCF = 29.8 Hz), 159.0, 158.0 (d, JCF = 273.0 Hz), 142.5, 139.0, 137.8, 133.9, 129.9, 129.2 (2C), 128.8 (2C), 125.3 (2C), 125.0, 122.9, 118.5, 113.7 (2C), 99.8 (d, JCF = 12.5 Hz), 59.8, 55.2, 35.4. m/z [EI (+ve)] 407.1 [M]+, HRMS found [M]+ 407.1504, C22H21F4NO2 requires 407.1508. IR (thin film) νmax = 2939, 1639, 1514, 1415, 1325, 1246, 1120, 1068 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(naphthalen-1′′′-yl)-3-butenyl]acrylamide, 8d
Amine 7d (0.5 g, 1.5 mmol) was coupled with 2-fluoroacrylic acid (0.21 g, 2.3 mmol) using HBTU (0.89 g, 2.3 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired dialkene 8d (0.21 g, 0.53 mmol, 36%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 8.07 (1H, d, JHH = 8.4 Hz), 7.87 (1H, br d, JHH = 8.4 Hz), 7.82 (1H, t, JHH = 4.6 Hz), 7.59–7.51 (2H, m), 7.44 (2H, d, JHH = 5.0 Hz), 6.76 (2H, d, JHH = 8.3 Hz), 6.63 (2H, d, JHH = 8.3 Hz), 6.57 (1H, br s), 5.87–5.76 (1H, m), 5.32 (1H, br d, JHF = 47.8 Hz), 5.14–5.05 (3H, m), 4.40 (1H, br d, JHH = 16.2 Hz), 3.91 (1H, dd, JHH = 16.2, 1.7 Hz), 3.75 (3H, s), 2.77 (2H, appt, JHH = 6.7 Hz). 19F (CDCl3, 377 MHz) δ: −103.7, −103.8. 13C (CDCl3, 100 MHz) δ: 162.9 (d, JCF = 30.4 Hz), 158.7, 158.2 (d, JCF = 263.3 Hz), 134.7, 133.9, 132.6, 132.5, 129.2, 128.8, 128.6 (2C), 126.9, 126.5, 124.7, 124.6, 123.4, 117.7, 114.4, 113.4 (2C), 99.9 (d, JCF = 15.8 Hz), 55.2 (2C), 54.1, 36.3. m/z [EI (+ve)] 389.2 [M]+. HRMS found [M]+ 389.1794, C25H24FNO2 requires 389.1791. IR (thin film) νmax = 2970, 1632, 1513, 1246, 1176 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(4′′′-bromophenyl)-3-butenyl]acrylamide, 8e
Amine 7e (0.50 g, 1.4 mmol) was coupled with 2-fluoroacrylic acid (0.19 g, 2.2 mmol) using HBTU (0.82 g, 2.2 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–5% diethyl ether in petroleum ether) to yield the desired dialkene 8e (0.24 g, 0.57 mmol, 41%) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.47 (2H, d, JHH = 8.1 Hz), 7.21 (2H, d, JHH = 8.1 Hz), 6.99 (2H, d, JHH = 8.6 Hz), 6.79 (2H, d, JHH = 8.6 Hz), 5.74–5.63 (1H, m), 5.40–5.25 (2H, m,), 5.19–5.05 (3H, m), 4.44 (1H, d, JHH = 16.0 Hz), 4.25 (1H, m), 3.81 (3H, s), 2.74–2.69 (2H, m). 19F (CDCl3, 470 MHz) δ: −102.3, −104.2. 13C (CDCl3, 125 MHz) δ: 163.3 (d, JCF = 29.4 Hz), 158.9, 157.9 (d, JCF = 273.3 Hz), 137.3, 133.9, 131.6 (2C), 130.3 (2C), 129.1 (2C), 129.0, 122.0, 118.3, 113. 7 (2C), 99.6 (d, JCF = 15.6 Hz), 55.3 (2C), 35.6, 23.9. m/z [EI (+ve)] 417.0 [M]+, HRMS found [M]+ 417.0739, C21H2179BrFNO2 requires 417.0740. IR (thin film) νmax = 2940, 1639, 1513, 1247, 1176 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-(1-isobutyl-3-butenyl)acrylamide, 8f
Amine 7f (0.27 g, 1.1 mmol) was coupled with 2-fluoroacrylic acid (0.25 g, 2.8 mmol) using HBTU (1.1 g, 2.8 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–2.5% diethyl ether in petroleum ether) to yield the desired dialkene 8f (0.26 g, 0.81 mmol, 74%) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.27 (2H, d, JHH = 8.0 Hz), 6.85 (2H, d, JHH = 7.2 Hz), 5.73–5.62 (1H, m), 5.26 (1H, br s), 5.15 (1H, br s), 5.13–5.02 (2H, m), 4.54–4.39 (2H, m), 4.19–4.01 (1H, m), 3.81 (3H, s), 2.33–2.19 (2H, m), 1.52–1.16 (3H, m), 0.85 (3H, d, JHH = 6.4 Hz,), 0.74 (3H, d, JHH = 6.0 Hz). 19F (CDCl3, 377 MHz) δ: −102.4 (dd, JFH = 47.5, 16.2 Hz), −103.6 (dd, JFH = 46.5, 15.1 Hz). 13C (CDCl3, 125 MHz) δ: 163.6 (d, JCF = 30.1 Hz), 158.7, 158.3 (d, JCF = 270.0 Hz), 135.4, 134.3, 130.3, 129.2 (2C), 118.0, 113.8 (2C), 98.5, 57.2, 55.3, 44.1, 42.0, 38.9, 24.6, 22.7. m/z [CI (+ve)] 320.2 [M + H]+, HRMS found [M + H]+ 320.2025, C19H27FNO2 requires 320.2026. IR (thin film) νmax = 2958, 1637, 1514, 1246 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-(1-cyclohexyl-3-butenyl)acrylamide, 8g
Amine 7g (0.48 g, 1.8 mmol) was coupled with 2-fluoroacrylic acid (0.24 g, 2.6 mmol) using HBTU (1.0 g, 2.6 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–2.5% diethyl ether in petroleum ether) to yield the desired dialkene 8g (0.44 g, 1.3 mmol, 70%) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.32 (2H, d, JHH = 8.6 Hz), 6.84 (2H, d, JHH = 8.6 Hz), 5.63–5.56 (1H, m), 5.27–4.91 (4H, m), 4.43 (2H, s), 3.81 (3H, s), 3.69 (1H, br t, JHH = 9.3 Hz), 2.51–2.44 (1H, m), 2.33–2.25 (1H, m), 1.85–1.51 (5H, m), 1.20–1.05 (2H, m), 0.97–0.81 (4H, m). 19F (CDCl3, 470 MHz) δ: −102.7. 13C (CDCl3, 125 MHz) δ: 164.6 (d, JCF = 30.0 Hz), 158.7, 158.2 (d, JCF = 270.0 Hz), 134.6, 130.1, 129.8 (2C), 117.7, 113.7 (2C), 98.5 (d, JCF = 15.0 Hz), 64.7, 55.3, 44.8, 40.9, 35.1, 30.5, 30.4, 26.3, 26.1, 26.0. m/z [CI (+ve)] 346.3 [M + H]+, HRMS found [M + H]+ 346.2176, C21H29FNO2 requires 346.2182. IR (thin film) νmax = 2924, 2852, 1635, 1513, 1442, 1246 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(furan-2′′′-yl)-3-butenyl]acrylamide, 8h
Amine 7h (0.5 g, 1.9 mmol) was coupled with 2-fluoroacrylic acid (0.26 g, 2.9 mmol) using HBTU (1.1 g, 2.9 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–10% diethyl ether in petroleum ether) to yield the desired dialkene 8h (0.18 g, 0.54 mmol, 28%) as a yellow oil.1H (CDCl3, 400 MHz) δ: 7.36 (1H, br s), 7.00 (2H, d, JHH = 8.6 Hz), 6.77 (2H, d, JHH = 8.6 Hz), 6.35–6.25 (2H, m), 5.72–5.64 (1H, m), 5.38–5.24 (2H, m), 5.10–5.05 (3H, m), 4.55 (1H, d, JHH = 15.7 Hz), 4.27 (1H, m), 3.80 (3H, s), 2.65–2.59 (2H, m). 19F (CDCl3, 470 MHz) δ: −103.5, −105.3. 13C (CDCl3, 125 MHz) δ: 163.1 (d, JCF = 29.4 Hz), 158.6, 157.6 (d, JCF = 267.8 Hz), 151.9, 142.4, 133.1, 129.2 (2C), 118.7, 114.2, 113.6 (2C), 110.3, 109.2, 99.3, 57.7, 55.2, 44.4, 23.8. m/z [EI (+ve)] 329.2 [M]+, HRMS found [M]+ 329.1427, C19H20FNO3 requires 329.1428. IR (thin film) νmax = 2956, 1699, 1513, 1246, 1117 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(1′′′-(toluene-4′′′′-sulfonyl)-1H-pyrrol-2′′′-yl)-3-butenyl]acrylamide, 8i
Amine 7i (0.66 g, 1.6 mmol) was coupled with 2-fluoroacrylic acid (0.22 g, 2.4 mmol) using HBTU (0.92 g, 2.4 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–15% EtOAc in petroleum ether) to yield the desired dialkene 8i (0.39 g, 0.81 mmol, 50%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.72 (2H, br s), 7.39 (1H, br s), 7.31 (2H, d, JHH = 8.3 Hz), 7.00 (2H, d, JHH = 8.6 Hz), 6.81 (2H, d, JHH = 8.6 Hz), 6.26–6.12 (2H, m), 5.75–5.65 (1H, m), 5.50–5.08 (3H, m), 4.81–4.53 (3H, m), 4.09 (1H, d, JHH = 6.0 Hz), 3.81 (3H, s), 2.49–2.41 (5H, m). 19F (CDCl3, 470 MHz) δ: −103.9. 13C (CDCl3, 125 MHz) δ: 162.4 (d, JCF = 29.4 Hz), 159.3, 157.9 (d, JCF = 276.8 Hz), 145.2, 143.4, 137.0, 135.9, 133.8, 130.0 (2C), 129.7 (2C), 129.3, 127.2 (2C), 117.8, 117.4, 114.0, 113.5 (2C), 98.9, 60.4, 55.2, 51.8, 46.7, 21.6. m/z [EI (+ve)] 482.1 [M]+, HRMS found [M]+ 482.1676, C26H27FN2O2S requires 482.1676. IR (thin film) νmax = 2975, 1652, 1511, 1247 cm−1.
2′-Fluoro-N-(4′′-methoxybenzyl)-N-[1-(pyridin-2′′′-yl)-3-butenyl]acrylamide, 8j
Amine 7j (0.5 g, 1.86 mmol) was coupled with 2-fluoroacrylic acid (0.25 g, 2.8 mmol) using HBTU (1.1 g, 2.8 mmol) following general procedure B. The crude product was purified by flash column chromatography (0–25% diethyl ether in petroleum ether) to yield the desired dialkene 8j (0.42 g, 1.24 mmol, 67%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 8.55 (1H, s), 7.63 (1H, dt, JHH = 7.7, 1.8 Hz), 7.37 (1H, br s), 7.19 (1H, br s), 6.93 (2H, d, JHH = 8.6 Hz), 6.70 (2H, d, JHH = 8.6 Hz), 5.76–5.65 (1H, m), 5.47–5.02 (5H, m), 4.59 (2H, s), 3.76 (3H, s), 3.04–2.95 (1H, m), 2.89–2.80 (1H, m). 19F (CDCl3, 377 MHz) δ: −102.5, −105.1. 13C (CDCl3, 125 MHz) δ: 163.4 (d, JCF = 29.4 Hz), 158.5, 158.0 (d, JCF = 271.5 Hz), 157.7, 149.0, 136.6, 134.4, 129.7 (2C), 128.7, 124.1, 122.9, 118.3, 113.5 (2C), 99.5 (d, JCF = 16.5 Hz), 62.5, 59.5, 55.1, 35.0. m/z [CI (+ve)] 341.1 [M + H]+, HRMS found [M + H]+ 341.1669, C20H22FN2O2 requires 341.1665. IR (thin film) νmax = 1638, 1513, 1415, 1207, 1176 cm−1.
General procedure C: ring-closing metathesis of fluorinated dialkene
A solution of the dialkene (1 eq.) in toluene (0.0025 g ml−1) was treated with Grubbs 2nd generation catalyst (2.5 mol%) and the resulting mixture was heated to 100 °C until completion as indicated by TLC analysis (1–4 hours). The reaction was cooled down to room temperature, the solvent was removed under reduced pressure and the crude material was purified by flash column chromatography.3-Fluoro-1-(4′-methoxybenzyl)-6-phenyl-5, 6-dihydro-1H-pyridin-2-one, 9a
Dialkene 8a (0.11 g, 0.32 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9a (0.10 g, 0.32 mmol, quantitative yield) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.40–7.34 (3H, m), 7.19 (2H, d, JHH = 7.2 Hz), 7.15 (2H, d, JHH = 8.6 Hz), 6.86 (2H, d, JHH = 8.6 Hz), 5.77 (1H, m), 5.54 (1H, d, JHH = 14.8 Hz), 4.57 (1H, dd, JHH = 7.7, 2.6 Hz), 3.83 (3H, s), 3.51 (1H, d, JHH = 14.8 Hz), 2.99–2.93 (1H, m), 2.50–2.44 (1H, m). 19F (CDCl3, 470 MHz) δ: −126.8. 13C (CDCl3, 125 MHz) δ: 159.8 (d, JCF = 30.0 Hz), 159.2, 149.4 (d, JCF = 251.1 Hz), 139.4, 129.6 (2C), 129.0, 128.9 (2C), 128.0, 126.4 (2C), 114.1 (2C), 109.5 (d, JCF = 14.6 Hz), 57.0, 55.3, 47.2, 29.4 (d, JCF = 6.0 Hz). m/z [EI (+ve)] 311.2 [M]+. HRMS found [M]+ 311.1318, C19H18FNO2 requires 311.1322. IR (thin film) νmax = 2933, 2837, 1651, 1512, 1247, 1176, 1031 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-(4′′-methoxyphenyl)-5,6-dihydro-1H-pyridin-2-one, 9b
Dialkene 8b (0.22 g, 0.58 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9b (0.19 g, 0.58 mmol, quantitative yield) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.05 (2H, d, JHH = 8.8 Hz), 7.00 (2H, d, JHH = 8.8 Hz), 6.80 (2H, d, JHH = 8.8 Hz), 6.77 (2H, d, JHH = 8.8 Hz), 5.70–5.66 (1H, m), 5.66 (1H, d, JHH = 14.8 Hz), 4.41 (1H, dd, JHH = 7.6, 3.2 Hz), 3.74 (3H, s), 3.84 (3H, s), 3.39 (1H, d, JHH = 14.8 Hz), 2.84–2.79 (1H, m), 2.37–2.32 (1H, m). 19F (CDCl3, 470 MHz) δ: −126.7. 13C (CDCl3, 125 MHz) δ: 159.7 (d, JCF = 30.2 Hz), 159.4, 159.2, 149.4 (d, JCF = 252.5 Hz), 131.2, 129.6 (2C), 129.0, 127.6 (2C), 114.2 (2C), 114.1 (2C), 109.6 (d, JCF = 13.8 Hz), 56.6, 55.4, 55.3, 47.1, 29.6 (d, JCF = 5.0 Hz). m/z [EI (+ve)] 341.1 [M]+. HRMS found [M]+ 341.1420, C20H20FNO3 requires 341.1427. IR (thin film) νmax = 2951, 2837, 1651, 1512, 1462, 1247, 1178, 1033 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-(4′′-trifluoromethanephenyl)-5,6-dihydro-1H-pyridin-2-one, 9c
Dialkene 8c (0.20 g, 0.49 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9c (0.18 g, 0.49 mmol, quantitative yield) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.64 (2H, d, JHH = 8.0 Hz), 7.30 (2H, d, JHH = 8.4 Hz), 7.14 (2H, d, JHH = 8.6 Hz), 6.85 (2H, d, JHH = 8.6 Hz), 5.78–5.75 (1H, m), 5.52 (1H, d, JHH = 14.8 Hz), 4.62 (1H, dd, JHH = 7.6, 2.0 Hz), 3.82 (3H, s), 3.53 (1H, d, JHH = 14.8 Hz), 3.05–2.98 (1H, m), 2.48–2.43 (1H, m). 19F (CDCl3, 470 MHz) δ: −62.3, −126.1. 13C (CDCl3, 125 MHz) δ: 159.6 (d, JCF = 30.1 Hz), 159.4, 149.4 (d, JCF = 253.8 Hz), 143.5, 130.6, 130.3, 129.6 (2C), 128.5, 126.8 (2C), 124.9 (2C), 114.2 (2C), 109.3 (d, JCF = 15.0 Hz), 56.7, 55.3, 47.5, 29.2 (d, JCF = 6.3 Hz). m/z [EI (+ve)] 379.0 [M]+, HRMS found [M]+ 379.1198, C20H17F4NO2 requires 379.1195. IR (thin film) νmax = 2970, 1737, 1654, 1512, 1413, 1327, 1249, 1112, 1068 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-(naphthalen-1′′-yl)-5,6-dihydro-1H-pyridin-2-one, 9d
Dialkene 8d (0.14 g, 0.36 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–15% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9d (0.13 g, 0.36 mmol, quantitative yield) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.97–7.94 (1H, m), 7.88 (1H, d, JHH = 8.2 Hz), 7.78–7.75 (1H, m), 7.56–7.53 (2H, m), 7.49 (1H, br t, JHH = 7.7 Hz), 7.34 (1H, d, JHH = 7.2 Hz), 7.15 (2H, d, JHH = 8.5 Hz), 6.86 (2H, d, JHH = 8.5 Hz), 5.74–7.69 (1H, m), 5.61 (1H, d, JHH = 14.8 Hz), 5.39 (1H, br d, JHH = 8.3 Hz), 3.83 (3H, s), 3.47 (1H, d, JHH = 14.8 Hz), 3.12–3.04 (1H, m), 2.70–2.62 (1H, m). 19F (CDCl3, 377 MHz) δ: −127.5. 13C (CDCl3, 125 MHz) δ: 160.5 (d, JCF = 31.2 Hz), 159.2, 149.1 (d, JCF = 255.0 Hz), 134.4, 133.5, 130.2, 129.7, 129.5 (2C), 129.1, 128.8, 126.7, 125.8, 125.3, 123.9 121.9, 114.1 (2C), 109.9 (d, JCF = 14.7 Hz), 55.3, 53.7, 47.4, 27.8 (d, JCF = 5.5 Hz). m/z [EI (+ve)] 361.2 [M]+. HRMS found [M]+ 361.1480, C23H20FNO2 requires 361.1478. IR (thin film) νmax = 2932, 1652, 1511, 1244, 1200 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-(4′′-bromophenyl)-5,6-dihydro-1H-pyridin-2-one, 9e
Dialkene 8e (0.17 g, 0.41 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–15% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9e (0.15 g, 0.39 mmol, 96%) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.51 (2H, d, JHH = 8.5 Hz), 7.14 (2H, d, JHH = 8.6 Hz), 7.05 (2H, d, JHH = 8.5 Hz), 6.87 (2H, d, JHH = 8.6 Hz), 5.79–5.74 (1H, m), 5.50 (1H, d, JHH = 14.6 Hz), 4.52 (1H, dd, JHH = 7.6, 2.4 Hz), 3.83 (3H, s), 3.51 (1H, d, JHH = 14.6 Hz), 3.00–2.91 (1H, m), 2.46–2.38 (1H, m). 19F (CDCl3, 377 MHz) δ: −126.3. 13C (CDCl3, 100 MHz) δ: 159.6 (d, JCF = 31.0 Hz), 159.3, 149.3 (d, JCF = 253.0 Hz), 138.5, 132.1 (2C), 129.6 (2C) 128.6, 128.1 (2C), 122.0, 114.2 (2C), 109.4 (d, JCF = 15.0 Hz), 56.5, 55.3, 47.3, 29.3 (d, JCF = 6.0 Hz). m/z [CI (+ve)] 391.7 [M + H]+, HRMS found [M + H]+ 390.0489, C19H1879BrFNO2 requires 390.0505. IR (thin film) νmax = 2950, 1653, 1512, 1247, 1217 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-isobutyl-5, 6-dihydro-1H-pyridin-2-one, 9f
Dialkene 8f (0.23 g, 0.71 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9f (0.17 g, 0.60 mmol, 85%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.14 (2H, d, JHH = 8.4 Hz), 6.79 (2H, d, JHH = 8.8 Hz), 5.77–5.71 (1H, m), 5.27 (1H, d, JHH = 14.8 Hz), 3.73 (3H, s), 3.66 (1H, d, JHH = 14.8 Hz), 3.31–3.26 (1H, m), 2.48–2.44 (1H, m), 2.14–2.07 (1H, m), 1.72–1.65 (1H, m), 1.43–1.39 (1H, m), 1.27–1.17 (1H, m), 0.85 (3H, d, JHH = 6.8 Hz), 0.76 (3H, d, JHH = 6.8 Hz). 19F (CDCl3, 470 MHz) δ: −127.6. 13C (CDCl3, 125 MHz) δ: 159.2, 158.7 (d, JCF = 31.3 Hz), 149.6 (d, JCF = 252.5 Hz), 129.5, 129.4 (2C), 114.1 (2C), 109.8 (d, JCF = 13.8 Hz), 55.3, 52.1, 46.9, 39.6, 25.1, 24.6 (d, JCF = 5.0 Hz), 23.6, 21.5. m/z [EI (+ve)] 291.2 [M]+. HRMS found [M]+ 291.1629, C17H22FNO2 requires 291.1635. IR (thin film) νmax = 2955, 1651, 1512, 1249, 1201 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-cyclohexane-5, 6-dihydro-1H-pyridin-2-one, 9g
Dialkene 8g (0.35 g, 1.06 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9g (0.30 g, 0.94 mmol, 89%) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.23 (2H, d, JHH = 8.8 Hz), 6.87 (2H, d, JHH = 8.8 Hz), 5.83–5.79 (1H, m), 5.49 (1H, d, JHH = 14.9 Hz), 3.83 (3H, s), 3.79 (1H, d, JHH = 14.9 Hz), 3.18 (1H, br t, JHH = 6.8 Hz), 2.54–2.45 (1H, m), 2.36–2.28 (1H, m), 1.86–1.63 (6H, m), 1.29–1.07 (4H, m), 1.00–0.94 (1H, m). 19F (CDCl3, 470 MHz) δ: −128.2. 13C (CDCl3, 125 MHz) δ: 159.2, 159.0 (d, JCF = 31.1 Hz), 149.5 (d, JCF = 253.2 Hz, CF), 129.7, 129.3 (2C), 114.1 (2C), 110.6 (d, JCF = 14.2 Hz), 59.0, 55.3, 48.7, 40.8, 30.3, 30.2, 26.4, 26.3, 26.2, 22.8 (d, JCF = 5.5 Hz). m/z [CI (+ve)] 318.2 [M + H]+, HRMS found [M + H]+ 318.1871, C19H25FNO2 requires 318.1869. IR (thin film) νmax = 2925, 2850, 1645, 1511, 1247, 1198 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-(furan-2′′-yl)-5,6-dihydro-1H-pyridin-2-one, 9h
Dialkene 8h (0.37 g, 1.1 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9h (0.21 g, 0.69 mmol, 62%) as a colourless oil.1H (CDCl3, 400 MHz) δ: 7.40 (1H, dd, JHH = 1.7, 0.6 Hz), 7.22 (2H, d, JHH = 8.6 Hz), 6.90 (2H, d, JHH = 8.6 Hz), 6.36 (1H, dd, JHH = 3.2, 1.8 Hz), 6.19 (1H, br d, JHH = 3.2 Hz), 5.91–5.86 (1H, m), 5.48 (1H, d, JHH = 14.8 Hz), 4.59 (1H, dd, JHH = 7.0, 2.5 Hz), 3.84 (3H, s), 3.75 (1H, d, JHH = 14.8 Hz), 2.84–2.75 (1H, m), 2.67–2.59 (1H, m). 19F (CDCl3, 470 MHz) δ: −126.3. 13C (CDCl3, 125 MHz) δ: 159.4 (d, JCF = 30.0 Hz), 152.2, 149.1 (d, JCF = 252.5 Hz), 142.5, 131.0, 129.6 (2C), 128.9, 114.2 (2C), 110.3, 110.1 (d, JCF = 16.5 Hz), 107.5, 55.3, 51.6, 47.4, 26.2 (d, JCF = 5.9 Hz). m/z [EI (+ve)] 301.2 [M]+. HRMS found [M]+ 301.1111, C17H16FNO3 requires 301.1114. IR (thin film) νmax = 2957, 2364, 1654, 1513, 1415, 1248, 1117 cm−1.
3-Fluoro-1-(4′-methoxybenzyl)-6-(1′′-(toluene-4′′′-sulfonyl)-1H-pyrrol-2′′-yl)-5,6-dihydro-1H-pyridin-2-one, 9i
Dialkene 8i (0.23 g, 0.47 mmol) was subjected to general procedure C. The crude product was purified by flash column chromatography (0–30% EtOAc in petroleum ether) to yield the desired α,β-unsaturated lactam 9i (0.16 g, 0.36 mmol, 77%) as a pale yellow oil.1H (CDCl3, 400 MHz) δ: 7.50 (2H, d, JHH = 8.5 Hz), 7.37 (1H, dd, JHH = 1.7, 1.5 Hz), 7.30 (2H, d, JHH = 8.5 Hz), 6.89 (2H, d, JHH = 8.6 Hz), 6.83 (2H, d, JHH = 8.6 Hz), 6.28 (1H, t, JHH = 3.3 Hz), 6.16–6.15 (1H, m), 5.76–5.72 (1H, m), 5.30 (1H, d, JHH = 15.0 Hz), 4.95 (1H, d, JHH = 7.2 Hz), 3.85 (3H, s), 3.12 (1H, d, JHH = 15.0 Hz), 2.94–2.87 (1H, m), 2.71–2.65 (1H, m), 2.47 (3H, s). 19F (CDCl3, 377 MHz) δ: −127.9. 13C (CDCl3, 125 MHz) δ: 159.6 (d, JCF = 30.0 Hz), 159.1, 148.8 (d, JCF = 252.5 Hz), 145.4, 136.1, 132.5, 130.3 (2C), 128.9 (2C), 128.8, 126.4 (2C), 124.8, 114.8, 114.0 (2C), 112.0, 109.8 (d, JCF = 15.0 Hz), 55.3, 51.9, 47.4, 27.0 (d, JCF = 6.3 Hz), 21.7. m/z [ESI (+ve)] 477.1 [M + Na]+, HRMS found [M + Na]+ 477.1259, C24H23FN2O4SNa requires 477.1255. IR (thin film) νmax = 2955, 1630, 1515, 1447, 1276, 1205 cm−1.
General procedure D: removal of the p-methoxybenzyl protecting group
The cyclic amide (1 eq.) was dissolved in a MeCN–H2O (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 4 mL) mixture and ceric ammonium nitrate was added portionwise. The resulting solution was stirred at room temperature until completion indicated by TLC analysis (7 h). The reaction was quenched with aq. sat. NaHCO3 (10 mL) and extracted with diethyl ether (3 × 10 mL). The organics were combined, dried (Na2SO4) and evaporated in vacuo. The crude material was purified by flash column chromatography.
2, 4 mL) mixture and ceric ammonium nitrate was added portionwise. The resulting solution was stirred at room temperature until completion indicated by TLC analysis (7 h). The reaction was quenched with aq. sat. NaHCO3 (10 mL) and extracted with diethyl ether (3 × 10 mL). The organics were combined, dried (Na2SO4) and evaporated in vacuo. The crude material was purified by flash column chromatography.
3-Fluoro-6-phenyl-5,6-dihydro-1H-pyridin-2-one, 10a
α,β-Unsaturated lactam 9a (96 mg, 0.31 mmol) was subjected to general procedure D using 0.45 g of ceric ammonium nitrate (2.7 eq., 0.86 mmol). The crude product was purified by flash column chromatography (0–30% EtOAc in petroleum ether) to yield the desired dihydropyridone 10a (60 mg, 0.29 mmol, 94%) as a white solid. m.p. 109–111 °C.1H (CDCl3, 400 MHz) δ: 7.45–7.37 (5H, m), 6.09 (1H, ddd, JHF = 11.1 Hz, JHH = 5.9, 3.3 Hz), 5.62 (1H, br s), 4.82 (1H, dd, JHH = 11.6, 5.8 Hz), 2.74–2.60 (2H, m). 19F (CDCl3, 470 MHz) δ: −129.9. 13C (CDCl3, 125 MHz) δ: 161.2 (d, JCF = 32.8 Hz), 149.1 (d, JCF = 253.2 Hz), 139.9, 129.2 (2C), 128.8, 126.4 (2C), 113.5 (d, JCF = 13.8 Hz), 56.1, 31.2 (d, JCF = 5.0 Hz). m/z [EI (+ve)] 191.1 [M]+, HRMS found [M]+ 191.0748, C11H10FNO requires 191.0746. IR (thin film) νmax = 2356, 1705, 1670, 1248 cm−1.
3-Fluoro-6-(4′-methoxyphenyl)-5,6-dihydro-1H-pyridin-2-one, 10b
α,β-Unsaturated lactam 9b (0.14 g, 0.42 mmol) was subjected to general procedure D using 1.3 g of ceric ammonium nitrate (5.5 eq., 2.3 mmol). The crude product was purified by flash column chromatography (0–25% EtOAc in petroleum ether) to yield the desired dihydropyridone 10b (37 mg, 0.17 mmol, 40%) as a yellow solid. m.p. 133–135 °C.1H (CDCl3, 400 MHz) δ: 7.21 (2H, d, JHH = 8.6 Hz), 6.84 (2H, d, JHH = 8.6 Hz), 6.01–6.97 (1H, m), 5.57 (1H, br s), 4.67 (1H, dd, JHH = 12.1, 5.6 Hz,), 3.81 (3H, s), 2.60–2.46 (2H, m). 19F (CDCl3, 470 MHz) δ: −130.0. 13C (CDCl3, 125 MHz) δ: 161.1 (d, JCF = 31.3 Hz), 159.8, 149.8 (d, JCF = 253.8 Hz), 131.9, 127.6 (2C), 114.4 (2C), 113.6 (d, JCF = 13.8 Hz), 55.6, 55.4, 31.2 (d, JCF = 5.3 Hz). m/z [CI (+ve)] 222.1 [M + H]+, HRMS found [M + H]+ 222.0929, C12H13FNO2 requires 222.09230. IR (thin film) νmax = 1695, 1630, 1250 cm−1.
3-Fluoro-6-(4′-trifluoromethanephenyl)-5,6-dihydro-1H-pyridin-2-one, 10c
α,β-Unsaturated lactam 9c (0.16 g, 0.42 mmol) was subjected to general procedure D using 1.1 g of ceric ammonium nitrate (4.9 eq., 2.1 mmol). The crude product was purified by flash column chromatography (0–20% EtOAc in petroleum ether) to yield the desired dihydropyridone 10c (50 mg, 0.20 mmol, 47%) as a white solid. m.p. 104–105 °C.1H (CDCl3, 500 MHz) δ: 7.60 (2H, d, JHH = 8.2 Hz), 7.43 (2H, d, JHH = 8.2 Hz), 5.99 (1H, ddd, JHF = 11.8 Hz, JHH = 5.5, 3.6 Hz), 5.93 (1H, br s), 4.80 (1H, dd, JHH = 10.5, 6.1 Hz), 2.67–2.53 (2H, m). 19F (CDCl3, 470 MHz) δ: −62.4, −129.1. 13C (CDCl3, 125 MHz) δ: 161.1 (d, JCF = 31.3 Hz), 149.6 (d, JCF = 253.8 Hz), 144.0, 131.1, 130.9, 126.8 (2C), 126.1 (2C), 113.2 (d, JCF = 13.8 Hz), 55.4, 30.9 (d, JCF = 5.0 Hz). m/z [EI (+ve)] 259.1 [M]+, HRMS found [M]+ 259.0623, C12H9F4NO requires 259.0620. IR (thin film) νmax = 1720, 1705, 1680, 1305, 1180 cm−1.
3-Fluoro-6-(naphthalen-1′-yl)-5,6-dihydro-1H-pyridin-2-one, 10d
α,β-Unsaturated lactam 9d (0.11 g, 0.30 mmol) was subjected to general procedure D using 0.97 g of ceric ammonium nitrate (5.9 eq., 1.8 mmol). The crude product was purified by flash column chromatography (0–15% EtOAc in petroleum ether) to yield the desired dihydropyridone 10d (37 mg, 0.15 mmol, 51%) as a yellow solid. m.p. 135–137 °C.1H (CDCl3, 400 MHz) δ: 7.96 (1H, d, JHH = 8.1 Hz), 7.85 (1H, br d, JHH = 7.9 Hz), 7.79 (1H, d, JHH = 8.1 Hz), 7.55–7.41 (4H, m), 6.04 (1H, dt, JHF = 11.0 Hz, JHH = 4.6 Hz), 5.74 (1H, br s), 5.56 (1H, t, JHH = 8.5 Hz), 2.81–2.76 (2H, m). 19F (CDCl3, 470 MHz) δ: −129.6. 13C (CDCl3, 125 MHz) δ: 161.2 (d, JCF = 33.0 Hz), 149.6 (d, JCF = 252.5 Hz), 135.3, 134.1, 130.0, 129.4, 129.2, 126.9, 126.2, 125.5, 123.9, 122.1, 113.7 (d, JCF = 12.5 Hz), 52.3, 29.7 (d, JCF = 5.0 Hz). m/z [EI (+ve)] 241.1 [M]+, HRMS found [M]+ 241.0902, C15H12FNO requires 241.0903. IR (thin film) νmax = 1715, 1797, 1320, 1180 cm−1.
3-Fluoro-6-(4′-bromophenyl)-5,6-dihydro-1H-pyridin-2-one, 10e
α,β-Unsaturated lactam 9e (0.19 g, 0.47 mmol) was subjected to general procedure D using 0.98 g of ceric ammonium nitrate (3.8 eq., 1.8 mmol). The crude product was purified by flash column chromatography (0–20% EtOAc in petroleum ether) to yield the desired dihydropyridone 10e (92 mg, 0.34 mmol, 72%) as a white solid. m.p. 199–200 °C.1H (CDCl3, 400 MHz) δ: 7.56 (2H, d, JHH = 8.1 Hz), 7.27 (2H, d, JHH = 8.1 Hz), 6.09 (1H, dt, JHF = 10.9 Hz, JH = 4.7 Hz), 5.58 (1H, br s), 4.79 (1H, t, JHH = 8.5 Hz), 2.67–2.62 (2H, m). 19F (CDCl3, 470 MHz) δ: −129.6. 13C (CDCl3, 125 MHz) δ: 161.0 (d, JCF = 31.2 Hz), 149.6 (d, JCF = 253.2 Hz), 138.9, 132.3 (2C), 128.0 (2C), 122.7, 113.3 (d, JCF = 13.8 Hz), 55.5, 31.0 (d, JCF = 5.0 Hz). m/z [CI (+ve)] 269.8 [M + H]+, HRMS found [M + H]+ 269.9945, C11H1079BrFNO requires 269.9930. IR (thin film) νmax = 1705, 1685, 1205, 1010 cm−1.
3-Fluoro-6-isobutyl-5,6-dihydro-1H-pyridin-2-one, 10f
α,β-Unsaturated lactam 9f (0.13 g, 0.44 mmol) was subjected to general procedure D using 0.91 g of ceric ammonium nitrate (3.8 eq., 1.7 mmol). The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired dihydropyridone 10f (70 mg, 0.4 mmol, 92%) as a white solid. m.p. 61–63 °C.1H (CDCl3, 500 MHz) δ: 6.07 (1H, ddd, JHF = 11.1 Hz, JHH = 5.9, 3.3 Hz), 5.59 (1H, br s, NH), 3.76–3.72 (1H, m), 2.49–2.40 (1H, m), 2.34–2.25 (1H, m), 1.74–1.63 (1H, m), 1.59–1.51 (1H, m), 1.45–1.36 (1H, m), 0.97 (3H, d, JHH = 6.6 Hz), 0.96 (3H, d, JHH = 6.6 Hz). 19F (CDCl3, 470 MHz) δ: −130.1. 13C (CDCl3, 100 MHz) δ: 161.2 (d, JCF = 28.0 Hz), 149.7 (d, JCF = 253.0 Hz), 113.7 (d, JCF = 13.0 Hz), 49.3, 44.0, 28.3 (d, JCF = 5.0 Hz), 24.4, 22.6, 22.2. m/z [CI (+ve)] 172.1 [M + H]+, HRMS found [M + H]+ 172.1144, C9H15FNO requires 172.1138. IR (thin film) νmax = 3219, 2934, 2906, 1696, 1669, 1264, 1206 cm−1.
3-Fluoro-6-cyclohexane-5,6-dihydro-1H-pyridin-2-one, 10g
α,β-Unsaturated lactam 9g (0.22 g, 0.70 mmol) was subjected to general procedure D using 1.3 g of ceric ammonium nitrate (3.3 eq., 2.3 mmol). The crude product was purified by flash column chromatography (0–10% EtOAc in petroleum ether) to yield the desired dihydropyridone 10g (0.11 g, 0.55 mmol, 79%) as a white solid. m.p. 108–110 °C.1H (CDCl3, 400 MHz) δ: 5.98 (1H, dt, JHF = 11.4 Hz, JHH = 4.3 Hz), 5.54 (1H, br s), 3.35 (1H, br q, JHH = 7.7 Hz), 2.32–2.29 (2H, m), 1.74–1.61 (5H, m), 1.43–1.32 (1H, m), 1.19–0.97 (3H, m), 0.95–0.92 (2H, m). 19F (CDCl3, 470 MHz) δ: −130.5. 13C (CDCl3, 125 MHz) δ: 161.3 (d, JCF = 31.3 Hz), 149.6 (d, JCF = 252.5 Hz), 114.0 (d, JCF = 13.8 Hz), 56.0, 41.4, 28.8, 28.7, 26.1, 25.9 (d, JCF = 5.0 Hz), 24.9, 24.8. m/z [CI (+ve)] 198.1 [M + H]+, HRMS found [M + H]+ 198.1295, C11H17FNO requires 198.1294. IR (thin film) νmax = 2927, 2855, 1691, 1652, 1208, 1199 cm−1.
General procedure E: hydrogenation of α,β-unsaturated lactams
A solution of dihydropyridone (1 eq.) in MeOH (2 mL) was treated with palladium activated charcoal (10% by weight) and the suspension was stirred under a H2 atmosphere until completion indicated by TLC analysis (1–4 hours). The resulting mixture was filtered through celite, dried (Na2SO4) and the solvent was removed under reduced pressure. The crude product was purified by flash column chromatography.3-Fluoro-6-phenyl-piperidin-2-one, 11a
Dihydropyridone 10a (43 mg, 0.22 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–30% EtOAc in petroleum ether) to yield the desired δ-lactam 11a (32 mg, 0.17 mmol, 75%) as a white solid. m.p. 149–151 °C.1H (CDCl3, 400 MHz) δ: 7.45–7.32 (5H, m), 5.93 (1H, br s), 4.95 (1H, dt, JHF = 46.27 Hz, JHH = 5.28 Hz), 4.61–4.60 (1H, m), 2.28–2.24 (1H, m), 2.17–2.03 (3H, m). 19F (CDCl3, 470 MHz) δ: −180.3. 13C (CDCl3, 125 MHz) δ: 167.5 (d, JCF = 20.1 Hz), 141.4, 129.0 (2C), 128.4, 126 1 (2C), 85.7 (d, JCF = 175.1 Hz), 57.3, 27.3 (d, JCF = 3.7 Hz), 26.2 (d, JCF = 20.0 Hz). m/z [EI (+ve)] 193.1 [M]+. HRMS found [M]+ 193.0904, C11H12FNO requires 193.0903. IR (thin film) νmax = 3194, 2066, 2958, 1666, 1329 cm−1.
3-Fluoro-6-(4′-methoxyphenyl)-piperidin-2-one, 11b
Dihydropyridone 10b (24 mg, 0.11 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–50% EtOAc in petroleum ether) to yield the desired δ-lactam 11b (14 mg, 0.06 mmol, 58%) as a white solid. m.p. 159–161 °C.1H (CDCl3, 400 MHz) δ: 7.25 (2H, d, JHH = 8.6 Hz), 6.94 (2H, d, JHH = 8.6 Hz), 5.83 (1H, s), 4.94 (1H, dt, JHF = 47.1 Hz, JHH = 4.6 Hz), 4.56–4.51 (1H, m), 3.84 (3H, s), 2.34–2.24 (1H, m), 2.13–1.96 (3H, m). 19F (CDCl3, 470 MHz) δ: −184.8. 13C (CDCl3, 125 MHz) δ: 167.2 (d, JCF = 22.1 Hz), 159.6, 133.4, 127.3 (2C), 114.4 (2C), 85.7 (d, JCF = 176.1 Hz), 57.0, 55.4, 27.4 (d, JCF = 4.6 Hz), 26.4 (d, JCF = 21.2 Hz). m/z [EI (+ve)] 223.1 [M]+. HRMS found [M]+ 223.0999, C12H14FNO2 requires 223.1009. IR (thin film) νmax = 2930, 1695, 1510, 1230 cm−1.
3-Fluoro-6-(4′-trifluoromethanephenyl)-piperidin-2-one, 11c
Dihydropyridone 10c (29 mg, 0.11 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–40% EtOAc in petroleum ether) to yield the desired δ-lactam 11c (29 mg, 0.11 mmol, quantitative yield) as a white solid. m.p. 122–124 °C.1H (CDCl3, 400 MHz) δ: 7.60 (2H, d, JHH = 8.2 Hz), 7.38 (2H, d, JHH = 8.2 Hz), 5.93 (1H, s), 4.86 (1H, dt, JHF = 47.1 Hz, JHH = 5.0 Hz), 4.59 (1H, br t, JHH = 5.7 Hz), 2.22–2.12 (1H, m), 2.11–1.89 (3H, m). 19F (CDCl3, 470 MHz) δ: −62.4, −185.0. 13C (CDCl3, 125 MHz) δ: 167.4 (d, JCF = 22.3 Hz), 145.4, 130.9, 130.6, 126.6 (2C), 126.1 (2C), 85.5 (d, JCF = 176.5 Hz), 57.0, 27.2 (d, JCF = 4.2 Hz), 26.1 (d, JCF = 21.0 Hz). m/z [EI (+ve)] 261.1 [M]+. HRMS found [M]+ 261.0773, C12H11F4NO requires 261.0777. IR (thin film) νmax = 3005, 2970, 1675, 1430 cm−1.
3-Fluoro-6-(naphthalen-1′-yl)-piperidin-2-one, 11d
Dihydropyridone 10d (38 mg, 0.15 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–40% EtOAc in petroleum ether) to yield the desired δ-lactam 11d (29 mg, 0.12 mmol, 81%) as a white solid. m.p. 144–147 °C.1H (CDCl3, 400 MHz) δ: 7.99–7.93 (2H, m), 7.87 (1H, d, JHH = 8.0 Hz), 7.62–7.51 (4H, m), 6.02 (1H, s), 5.48–5.46 (1H, m), 5.02 (1H, dt, JHF = 46.9 Hz, JHH = 5.7 Hz), 2.43–2.33 (1H, m), 2.28–2.23 (3H, m). 19F (CDCl3, 377 MHz) δ: −186.1. 13C (CDCl3, 100 MHz) δ: 168.3 (d, JCF = 19.9 Hz), 136.6, 134.0, 129.8, 129.4, 128.9, 126.8, 126.0, 125.4, 123.7, 121.9, 85.8 (d, JCF = 177.0 Hz), 53.3, 25.9 (d, JCF = 6.0 Hz), 25.7 (d, JCF = 20.0 Hz). m/z [CI (+ve)] 244.0 [M + H]+, HRMS found [M + H]+ 244.1137, C15H15FNO requires 244.1138. IR (thin film) νmax = 3240, 2900, 1650, 1110 cm−1.
3-Fluoro-6-isobutyl-piperidin-2-one, 11f
Dihydropyridone 10f (39 mg, 0.23 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–20% EtOAc in petroleum ether) to yield the desired δ-lactam 11f (40 mg, 0.23 mmol, quantitative yield) as a white solid. m.p. 78–81 °C.1H (CDCl3, 400 MHz) δ: 6.00 (1H, s), 4.80 (1H, dt, JHF = 48.6 Hz, JHH = 4.5 Hz), 3.40–3.37 (1H, m), 2.16–2.13 (1H, m), 1.96–1.89 (1H, m), 1.81–1.77 (1H, m), 1.64–1.58 (2H, m), 1.40–1.31 (2H, m), 0.87 (3H, d, JHH = 6.6 Hz), 0.85 (3H, d, JHH = 6.6 Hz). 19F (CDCl3, 470 MHz) δ: −185.0. 13C (CDCl3, 125 MHz) δ: 167.4 (d, JCF = 20.0 Hz), 86.3 (d, JCF = 173.8 Hz), 50.8, 45.6, 26.5 (d, JCF = 21.3 Hz), 24.3, 24.0 (d, JCF = 3.7 Hz), 22.6, 22.3. m/z [CI (+ve)] 174.1 [M + H]+, HRMS found [M + H]+ 174.1300, C9H17FNO requires 174.1294. IR (thin film) νmax = 2950, 2935, 1630 cm−1.
3-Fluoro-6-cyclohexane-piperidin-2-one, 11g
Dihydropyridone 10g (80 mg, 0.40 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–20% EtOAc in petroleum ether) to yield the desired δ-lactam 11g (81 mg, 0.40 mmol, quantitative yield) as a white solid. m.p. 148–159 °C.1H (CDCl3, 500 MHz) δ: 5.93 (1H, s), 4.74 (1H, dt, JHF = 47.2 Hz, JHH = 4.4 Hz), 3.13–3.09 (1H, m), 2.21–2.13 (1H, m), 1.90–1.79 (1H, m), 1.74–1.62 (7H, m), 1.35–1.30 (1H, m), 1.22–1.14 (2H, m), 1.11–1.03 (1H, m), 0.99–0.89 (2H, m). 19F (CDCl3, 470 MHz) δ: −184.6. 13C (CDCl3, 125 MHz) δ: 167.4 (d, JCF = 19.9 Hz), 86.0 (d, JCF = 173.7 Hz), 57.8, 42.6, 28.6, 28.4, 26.9 (d, JCF = 21.3 Hz), 26.2, 26.0, 25.9, 20.2 (d, JCF = 3.4 Hz). m/z [EI (+ve)] 199.1 [M]+. HRMS found [M]+ 199.1376, C11H18FNO requires 199.1372. IR (thin film) νmax = 2926, 2870, 1664, 1410 cm−1.
3-Fluoro-6-(4′-phenylphenyl)-5,6-dihydro-1H-pyridin-2-one, 12
Dihydropyridone 10e (98 mg, 0.36 mmol) was dissolved in a mixture of toluene (12 mL) and H2O (2 mL). K2CO3 (0.11 g, 0.79 mmol), PhB(OH)2 (88 mg, 0.72 mmol) and Pd(PPh3)4 (84 mg, 20 mol%) were then sequentially added and the resulting solution was heated at 90 °C for 16 h. The reaction was cooled down to room temperature and was filtered through celite and the celite was washed with EtOAc (30 mL). The organic layer was washed with H2O (1 × 10 mL) and brine (1 × 10 mL) and then dried (Na2SO4) and evaporated in vacuo. The crude residue was purified by flash column chromatography (0–20% EtOAc in petroleum ether) to yield the desired cross-coupled product 12 (87 mg, 0.33 mmol, 90%) as a white solid. m.p. 199–201 °C.1H NMR (CDCl3, 400 MHz) δ: 7.58 (2H, d, JHH = 8.0 Hz), 7.52 (2H, d, JHH = 8.0 Hz), 7.40–7.31 (5H, m), 6.02 (1H, ddd, JHH = 11.0, 5.8, 3.3 Hz), 5.59 (1H, s), 4.77 (1H, dd, JHH = 11.3, 6.0 Hz), 2.70–2.55 (2H, m). 19F (CDCl3, 377 MHz) δ: −129.8. 13C (CDCl3, 100 MHz) δ: 161.0 (d, JCF = 31.1 Hz), 149.9 (d, JCF = 253.6 Hz), 141.8, 140.2, 138.9, 128.9 (2C), 127.8 (2C), 127.7, 127.1 (2C), 126.8 (2C), 113.5 (d, JCF = 14.0 Hz), 55.8, 31.1 (d, JCF = 5.7 Hz). m/z [EI (+ve)] 267.1 [M]+, HRMS found [M]+ 267.1058, C17H14FNO requires 267.1059. IR (thin film) νmax = 2358, 1693, 1658, 1258, 1198 cm−1.
3-Fluoro-6-(4′-phenylphenyl)-piperidin-2-one, 13
Dihydropyridone 12 (34 mg, 0.13 mmol) was subjected to general procedure E. The crude product was purified by flash column chromatography (0–40% EtOAc in petroleum ether) to yield the desired δ-lactam 13 (34 mg, 0.13 mmol, quantitative) as a white solid. m.p. 171–173 °C.1H (CDCl3, 400 MHz) δ: 7.64 (2H, d, JHH = 7.5 Hz), 7.61 (2H, d, JHH = 7.5 Hz), 7.50–7.38 (5H, m), 5.91 (1H, s), 4.97 (1H, dt, JHF = 43.7 Hz, JHH = 4.5 Hz), 4.67–4.63 (1H, m), 2.36–2.27 (1H, m), 2.20–2.04 (3H, m). 19F (CDCl3, 377 MHz) δ: −185.1. 13C (CDCl3, 100 MHz) δ: 167.5 (d, JCF = 19.8 Hz), 141.4, 140.3, 140.2, 128.9 (2C), 127.7 (2C), 127.6, 127.1 (2C), 126.6 (2C), 85.7 (d, JCF = 176.0 Hz), 57.2, 27.3 (d, JCF = 4.0 Hz), 26.3 (d, JCF = 21.0 Hz). m/z [EI (+ve)] 269.0 [M]+, HRMS found [M]+ 269.1214, C17H16FNO requires 269.1216. IR (thin film) νmax = 3239, 2949, 2356, 1676, 1486 cm−1.
Crystallographic data collection and refinement details
X-ray diffraction data of crystals of 11a were collected at 150 K on an Oxford Diffraction Gemini CCD diffractometer equipped with an Oxford Cryosystems Cryostream low-temperature device and using graphite monochromated Cu Kα radiation (λ = 1.54184 Å). Data reduction was carried out and an analytical numeric absorption correction was applied [based on expressions derived in R. C. Clark and J. S. Reid, Acta Crystallogr., Sect. A: Fundam. Crystallogr., 1995, 51, 887–897] using the CrysAlisPro [Oxford Diffraction Limited., Version 1.171.33.55, Oxfordshire, UK]. The structures were all solved by direct methods using the program SHELXS97 [SHELX, G. M. Sheldrick, Acta Crystallogr., Sect. A: Fundam. Crystallogr., 2008, 64, 112–122] and refined using full-matrix least-squares refinement on F2 using SHELXL97 [SHELX, G. M. Sheldrick, Acta Crystallogr., Sect. A: Fundam. Crystallogr., 2008, 64, 112–122] within the WinGX program suite [L. J. Farrugia, J. Appl. Crystallogr., 1999, 32, 837–838]. The Flack x parameter [H. D. Flack, Acta Crystallogr., Sect. A: Fundam. Crystallogr., 1983, 39, 876.] was determined for 11a.Acknowledgements
We would like to thank the EPSRC and AstraZeneca for postgraduate support (T.C.). The authors also thank Dr Ian Sword, the EPSRC (grant EP/H005692/1) and AstraZeneca for funding.Notes and references
- (a) S. S. Salim, R. K. Bellingham, V. Satcharoen and R. C. D. Brown, Org. Lett., 2003, 5, 3403–3406 CrossRef CAS PubMed; (b) C. Isanbor and D. O'Hagan, J. Fluorine Chem., 2006, 127, 303–319 CrossRef CAS PubMed; (c) V. Dinoiu, Rev. Roum. Chim., 2006, 51, 1141–1152 CAS; (d) S. Purser, P. R. Moore, S. Swallow and V. Governeur, Chem. Soc. Rev., 2008, 37, 320–330 RSC; (e) D. O'Hagan, Chem. Soc. Rev., 2008, 37, 308–319 RSC; (f) W. K. Hagmann, J. Med. Chem., 2008, 51, 4359–4369 CrossRef CAS PubMed; (g) R. Filler and R. Saha, Future Med. Chem., 2009, 1, 777–791 CrossRef CAS PubMed.
- (a) X.-G. Hu and L. Hunter, Beilstein J. Org. Chem., 2013, 9, 2696–2708 CrossRef PubMed; (b) R. Surmont, G. Verniest, J. W. Thuring, G. Macdonald, F. Deroose and N. De Kimpe, J. Org. Chem., 2010, 75, 929–932 CrossRef CAS PubMed.
- (a) T. Hirose, Y. Uwabodo, S. Yamada, T. Kikuchi, H. Kitagawa, K. D. Burris, C. A. Altar and T. Tabeshima, J. Psychopharmacol., 2004, 18, 375–383 CrossRef CAS PubMed; (b) S. Jafari, F. Fernandez-Enright and X. Huang, J. Neurochem., 2012, 120, 371–384 CrossRef CAS PubMed.
- M. Marhold, A. Buer, H. Hiemstra, J. H. Van Maarseveen and G. Haufe, Tetrahedron Lett., 2004, 45, 57–60 CrossRef CAS PubMed.
- (a) V. De Matteis, F. L. van Delft, R. Gelder, J. Tiebes and F. P. J. T. Rutjes, Tetrahedron Lett., 2004, 45, 959–963 CrossRef CAS PubMed; (b) V. De Matteis, F. L. van Delft, J. Tiebes and F. P. J. T. Rutjes, Eur. J. Org. Chem., 2006, 1166–1176 CrossRef CAS; (c) V. De Matteis, O. Dufay, D. C. J. Waalboer, F. L. van Delft, J. Tiebes and F. P. J. T. Rutjes, Eur. J. Org. Chem., 2007, 2667–2675 CrossRef CAS.
- S. Arimitsu and G. B. Hammond, Beilstein J. Org. Chem., 2010, 6(48) CAS.
- M. Sugiura, K. Hirano and S. Kobayashi, J. Am. Chem. Soc., 2004, 126, 7182–7183 CrossRef CAS PubMed.
- P. Formentin, N. Gimeno, J. H. G. Steinke and R. Vilar, J. Org. Chem., 2005, 70, 8235–8238 CrossRef CAS PubMed.
- V. Jurčík, A. M. Z. Slawin and D. O'Hagan, Beilstein J. Org. Chem., 2011, 7, 759–766 CrossRef PubMed.
- The atomic coordinates for 11a (CCDC deposition number CCDC1006297) are available upon request from the Cambridge Crystallographic Centre, University Chemical Laboratory, Lensfield Road, Cambridge, CB2 1EW, United Kingdom. The crystallographic numbering system differs from that used in the text; therefore, any request should be accompanied by the full literature citation of this paper.
- N. Miyaura, T. Yanagi and A. Suzuki, Synth. Commun., 1981, 11, 513–519 CrossRef CAS.
- B. Hatano, T. Kijima, M. Kusakari, S. Matsuba, S. Murakami, T. Obu and X. Sun, Heterocycles, 2013, 87, 2395–2402 CrossRef.
- C. Botteghi, S. Paganelli, B. Vicentini and C. Zarantonello, J. Fluorine Chem., 2001, 107, 113–116 CrossRef CAS.
- H.-J. Kim, D. K. Dogutan, M. Ptaszek and J. S. Lindsey, Tetrahedron, 2007, 63, 37–55 CrossRef CAS PubMed.
- C. L. Man, H. C. Tak, W. C. Tin and K. Wong, J. Org. Chem., 2007, 72, 923–929 CrossRef PubMed.
- D. A. Chaudhari and R. A. Fernandes, Eur. J. Org. Chem., 2012, 1945–1952 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Full refinement details and crystallographic data (excluding structural factors). CCDC 1006297. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ob01547a |
| ‡ The author to whom crystallographic data questions should be addressed. |
| § Ian Sword Reader of Organic Chemistry. |
| This journal is © The Royal Society of Chemistry 2015 |