Unravelling the anomalous dielectric permittivity of nanoconfined electrolyte solutions
Abstract
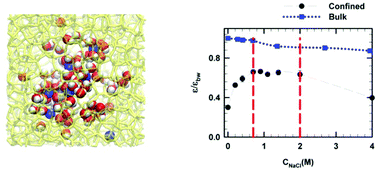
The dielectric properties of sodium chloride solutions confined in a hydrophilic nanocavity were investigated by means of molecular dynamics simulations. Unlike what is observed in the bulk phase, three dielectric regimes were evidenced, namely an anomalous increase in the dielectric permittivity at low concentrations (with respect to confined pure water), a dielectric plateau at intermediate concentrations and finally a bulk-like behavior for salt concentrations higher than a critical value. It was shown that this peculiar behavior results from the competition between dielectric saturation due to the electric field generated by ions (which tends to lower the dielectric permittivity) and the ion-induced perturbation of pre-oriented water molecules inside the nanocavity which gain some rotational degrees of freedom (entropic contribution) leading to an increase in dipolar fluctuations responsible for the increase in the dielectric permittivity.

 Please wait while we load your content...
Please wait while we load your content...