Rare-earth metal bis(alkyl) complexes bearing pyrrolidinyl-functionalized cyclopentadienyl, indenyl and fluorenyl ligands: synthesis, characterization and the ligand effect on isoprene polymerization†
Abstract
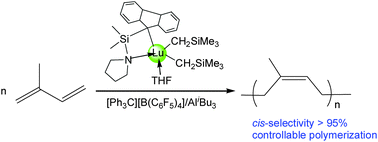
A series of rare-earth metal bis(alkyl) complexes bearing pyrrolidinyl-functionalized cyclopentadienyl, indenyl and fluorenyl ligands were prepared and characterized. Alkane elimination of Ln(CH2SiMe3)3(THF)2 with 1 equiv. of the corresponding ancillary ligand precursors in THF at room temperature afforded the rare-earth metal bis(alkyl) complexes LLn(CH2SiMe3)2(THF)2 (L = C5Me4SiMe2NC4H8, Ln = Y (1), Lu (2); L = C9H6SiMe2NC4H8, Lu (4)). Intramolecular C–H activation occurred when Sc(CH2SiMe3)3(THF)2 was treated with 1 equiv. of C5Me4HSiMe2NC4H8, leading to the generation of the scandium mono(alkyl) complex (C5Me4SiMe2NC4H7)Sc(CH2SiMe3)(THF) (3). One-pot salt metathesis reaction of LnCl3 with 1 equivalent of fluorenyl lithium C13H8SiMe2NC4H8Li, followed by the addition of 2 equivalents of LiCH2SiMe3 in THF at room temperature, gave the mono-fluorenyl-ligated rare-earth metal bis(alkyl) complexes (C13H8SiMe2NC4H8)Ln(CH2SiMe3)2(THF) (Ln = Y (5), Lu (6)). All these complexes were characterized by elemental analysis and NMR spectroscopy. 3, 5 and 6 were subjected to X-ray single crystal determination. Upon activation with one equivalent of [Ph3C][B(C6F5)4] and excess AliBu3, the rare-earth metal bis(alkyl) complexes 1, 2, 4–6 were active for cis-1,4-selective polymerization of isoprene in toluene at room temperature. The ligand effect on the polymerization activity was observed, with a sterically more bulky ligand providing higher activity (fluorenyl > indenyl > Cp). Moreover, employing 6/[Ph3C][B(C6F5)4]/AliBu3 as the catalyst system, polymerization proceeded in a controllable fashion.
- This article is part of the themed collection: Frontiers of Organo-f-Element Chemistry

 Please wait while we load your content...
Please wait while we load your content...