Synthesis and conformational studies of α/β2,3-peptides derived from alternating β2,3-amino acids and l-Ala repeats†
Abstract
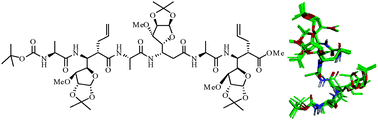
Cyclic β2,3-amino acids though have been extensively used in foldamer designs, their acyclic analogues have received less attention. In view of strong backbone constraints imparted by the substituents, β2,3-amino acids provide very attractive options for creating novel foldamers. In the present study, new C-linked carbo-β2,3-amino acids (β2,3-Caa) were prepared by the alkylation of Cα-carbon (C2) with allyl and propargyl halides, and used with L-Ala to design regular 1 : 1 hybrid α/β2,3-peptides. Extensive NMR and MD studies revealed the presence of right-handed 11/9-mixed helices in the peptides with the ‘α-β-α’ sequence at the C-terminus, while, induction of ‘turns’ in the peptides with the ‘β-α-β’ motif at the C-terminus. Despite the strong backbone constraints due to the substitution at both the β2 and β3-carbons, the folds of these α/β2,3-peptides are less robust compared to foldamers of the α/β3-family, reflecting the impact of Cα-substitution.
- This article is part of the themed collection: Foldamer Chemistry

 Please wait while we load your content...
Please wait while we load your content...