Perovskite chromate doped with titanium for direct carbon dioxide electrolysis†
Abstract
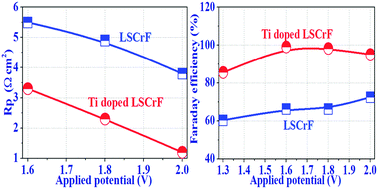
A composite cathode based on lanthanum chromate has uncovered tremendous potential for direct high temperature electrolysis. Unfortunately, a major setback of insufficient electrocatalytic activity limits the efficient carbon dioxide electrolysis. In this paper, catalytically active Ti is doped to partially replace Cr at the B-site in perovskite La0.75Sr0.25Cr0.5−xFe0.5TixO3−δ (LSCrFT, x = 0, 0.1, 0.2, 0.3, 0.4 and 0.5) to improve the polarization and electrocatalytic property of the cathode materials of the solid oxide electrolyzer. The transition of the dominant mechanism from p-type conduction to n-type conduction is observed for the LSCrFT cathode with the Ti doping level at x = 0.3. The doping of Ti in LSCrFT largely improves the electrode activity and therefore reduces electrode polarization resistance. A high-temperature CO2 electrolysis test demonstrates that the Faraday efficiencies of LSCrFT (x = 0.1) cathodes are enhanced by approximately 30% in contrast to bare LSCrF cathodes at 800 °C.

 Please wait while we load your content...
Please wait while we load your content...