Research highlights: elucidating the role of complex interactions affecting nanoparticle deposition
Stacey M.
Louie
* and
John M.
Pettibone
Materials Measurement Science Division, National Institute of Standards and Technology, Gaithersburg, MD 20899, USA. E-mail: stacey.louie@nist.gov
First published on 19th May 2015
Abstract
In this issue, we highlight recent works that advance our understanding of physicochemical mechanisms governing nanoparticle (NP) deposition in the environment. In one study, the transport of citrate-stabilized silver NPs in an unsaturated porous medium was assessed experimentally, which provided data to assess the contributions of NP attachment at the air–water interface with a modified transport model. Another study applied cryogenic transmission electron microscopy and small angle X-ray scattering to examine the effect that ferrihydrite NP aggregation state (density and structure) has on deposition behavior. Finally, we highlight a study that investigated the influence of excess polymeric constituents on the deposition of polymer-coated quantum dots and incorporated this effect in a multi-component site-blocking model. Overall, these studies highlight three distinct mechanisms of NP attachment beyond simple scenarios of saturated conditions, stable NPs, and single-constituent systems. These studies also point to new directions for improving the predictive capability of transport models.
Introduction
The attachment behavior of nanoparticles (NPs) (i.e., homoaggregation, heteroaggregation, and sorption or deposition) is a key component in assessments of the transport, and hence exposure risk, of NPs in the natural environment. Recent debates over the formulation and parameterization of fate and transport models1–6 and experimental approaches to assess attachment behavior1,4 reflect ongoing challenges in explaining and predicting NP transport. Here, we focus on the fundamental deposition behavior of NPs in porous media and the challenge in accounting for a potentially wide and complex range of NP–interface interactions in the environment, including contributions from dissolved constituents. Progress in elucidating the mechanisms of NP attachment in these complex systems has been driven by more thorough characterization of the NP and system properties and improved experimental design. These advancements in understanding competing complex interactions should ultimately lead to more predictive transport models. We highlight three recent articles that contribute to this theme by evaluating NP deposition at air–water interfaces, as a function of NP aggregate structure, and the effects of dissolved constituents.Assessing NP attachment at the air–water interface in unsaturated porous media
NPs applied to surface soils, e.g. during application of wastewater sludge for crop fertilization, will interact with not only solid–water interfaces (SWI) but also air–water interfaces (AWI). Deposition studies performed in unsaturated media are necessary to determine the relative contributions in each region governing overall NP transport in surface soils. However, the majority of NP deposition studies assess the saturated condition, in part because of the difficulty in establishing a well-defined unsaturated system, and thus result in the development of less refined models.Recently, Kumahor et al. developed a multistep-transport approach to establish and characterize a well-defined unsaturated sand column, in which uniform pressure head and water content are achieved throughout the column.7 Different pore water velocities and degrees of saturation are obtained by varying the input flowrate under gravity-driven flow. The area of the AWI was estimated by analysis of X-ray microtomography images. Here, we highlight their application of this method to assess the deposition of citrate-reduced silver nanoparticles (cit-AgNPs) in an unsaturated packed sand bed.8
Both the breakthrough curves of cit-AgNPs eluting from the column and retention profiles of the deposited NPs (by column sectioning) were measured. The breakthrough curves showed significant retardation of the NPs observed compared to a tracer (Fig. 1a), and the retention profiles showed decreasing deposited mass with distance from the column inlet. The observation of NP retardation is inconsistent with deposition models considering only kinetically controlled attachment/detachment, prompting further theoretical analysis of the attachment at the AWI. Kumahor et al. found that classical DLVO theory, in which only van der Waals and electrostatic forces are considered, predicted a primary energy minimum and high repulsive energy barrier for both the SWI and AWI. On the contrary, extended DLVO theory including a hydrophobic interaction predicted a secondary minimum for the AWI, suggesting an equilibrium partitioning process that could explain the observed NP retardation (Fig. 1b).
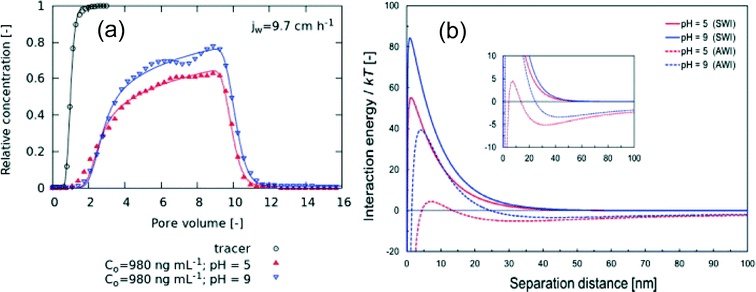 | ||
| Fig. 1 Experimental breakthrough curves (a) show the eluting nanoparticle concentration (normalized by the influent concentration, C0) versus pore volume (i.e., eluted volume of suspension normalized by the total liquid-containing pore volume of the column). The breakthrough curves showed significant retardation of cit-AgNPs compared to a KBr tracer, which could be modeled by including a retardation term representing equilibrium attachment at the AWI. This mechanism is supported by extended DLVO models including hydrophobic interaction, which indicate a secondary minimum for cit-AgNP attachment at the AWI but not the SWI (b). Figures reprinted with permission from Kumahor et al.8 Copyright 2015 Elsevier. | ||
Because the data and theory supported the presence of distinct processes occurring at different interfaces, a one-dimensional NP transport model incorporating attachment/detachment rates at the SWI and an equilibrium retardation factor for the AWI, along with a particle straining term that accounts for geometries of the pores, flow fields, and NPs and the pore-water chemistry, was developed to more accurately model the system. Good fits to the breakthrough data were achieved with the incorporation of these factors (Fig. 1a).
In summary, Kumahor et al. determined that equilibrium sorption attributable to hydrophobic interactions at the air–water interface was an important mechanism affecting the transport of cit-AgNPs in unsaturated porous media. Their implementation of a well-controlled and well-characterized unsaturated column allowed for more confident modeling of the cit-AgNP deposition behavior. Application of this experimental design to assess a broader range of NP types should result in data sets for a range of NPs to assess distinct property contributions of attachment at the AWI that could be used to better predict NP transport in surface soils. These models could ultimately improve assessment of NP exposure risks in terrestrial environments and runoff into surface waters.
Evaluating effects of aggregate structure on NP deposition
The aggregation state of NPs is expected to affect their deposition behavior9,10 through the effects on the hydrodynamics and, debatably,11 the interaction forces of the NP aggregates. Although measurement of the hydrodynamic size of the aggregates is straightforward using dynamic light scattering for uniform size distributions or flow field flow fractionation for polydisperse populations, characterization of the aggregate structure (i.e., fractal dimension) requires advanced characterization methods and is therefore not often performed, precluding definitive assessment of the effects of NP aggregate density and morphology on transport behavior.Legg et al. recently tackled this problem in their study of the deposition of ferrihydrite NP aggregates, which allowed distinct structural changes over an ionic strength range of 0 to 20 mM (NaNO3).12 In deionized water, low density, linear NP structures were observed (Fig. 2a), which collapse into denser structures with increasing ionic strength as observed with cryogenic transmission electron microscopy (cryo-TEM) (Fig. 2b). Apparent fractal dimensions were determined from small-angle X-ray scattering (SAXS) data. The authors reported that the fractal dimensions determined by SAXS did not correspond exactly to the true fractal dimension because of the finite aggregate size (violating assumptions of infinite aggregate size) as well as polydispersity effects, as they discussed in detail previously,13 demonstrating the importance of applying a complementary method such as cryo-TEM. However, the SAXS results generally concurred with the trend of increasing fractal dimension with ionic strength, corresponding to denser aggregate structures.
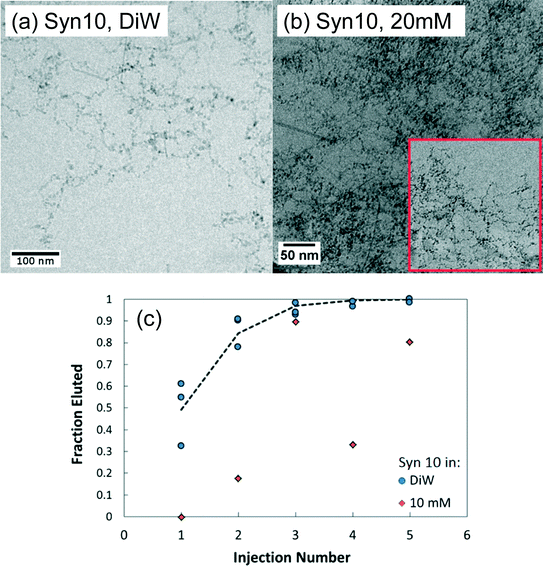 | ||
| Fig. 2 Ferrihydrite NPs formed low density, linear aggregate structures in deionized water (a) and collapsed to higher density structures at higher ionic strength (b). Transport models including a filter coefficient with site blocking could only explain aggregate deposition at low ionic strength, whereas increased deposition with sporadic releases of large quantities of NP was observed for the denser aggregate structures (c). Figures reprinted with permission from Legg et al.12 Copyright 2014 American Chemical Society. | ||
Column transport experiments were performed in a saturated packed sand column at ionic strengths ranging from 0 to 10 mM to assess the deposition of different aggregate structures. The experiments were performed at pH 5.5, between the isoelectric point of the NPs and the sand, such that favorable deposition conditions and hence lesser impact of charge screening could be expected. Breakthrough curves were measured for multiple sequential pulse injections, and inverse modeling using a deep-bed filtration model was applied to determine the maximum specific particle loading and filter coefficient. Legg et al. determined that the breakthrough curves for the ferrihydrite aggregates could only be successfully modeled at low ionic strength (<2 mM NaNO3). At higher ionic strengths, the maximum particle loading increased significantly and erratic elution of large quantities of NPs were observed with later pulse injections (Fig. 2c). Furthermore, higher deposition was visually observed toward the column outlet, contrary to expected deposition behavior for the applied filtration model.
The authors proposed that the denser aggregates at higher ionic strength produced denser and thicker deposits on the sand (thus allowing for higher specific loading). Hydrodynamic instability of the growing deposits was suggested to explain the large releases of NPs. The authors suggest that aggregate restructuring during transport may contribute to the retention profiles but acknowledge that further characterization of the mechanical properties of the aggregates is needed. Overall, Legg et al. concluded that NP deposition behavior depended significantly on aggregate structure and the mechanisms involved in deposition of large, dense ferrihydrite aggregates could not be represented using simple deposition models. While in situ characterization would be useful to evaluate their proposed mechanism of aggregate deposition, scanning electron microscopy imaging of the aggregate deposits performed by the authors was inconclusive because of the roughness of the sand substrate.
This study suggests that more thorough characterization of aggregate structure may be required for systems in which NP aggregation occurs. The large, sporadic releases observed as the NP deposits grew at higher ionic strength may also have important implications for the long-term risk of releasing NP-containing deposits in the environment. Little information is currently available in this area, and warrants further investigation of deposition behavior in other systems.
Incorporating blocking effects of excess polymer in NP transport models
The deposition of NPs can be strongly affected by the presence of macromolecules in the system, either dissolved or pre-adsorbed onto collector surfaces.14,15 Models of the interaction energy of nanoparticles coated with neutral polymers have suggested that coating of the collector surface may in fact be necessary to prevent attachment.16 These scenarios are highly relevant to natural aqueous environments, where natural organic matter is ubiquitous. Excess synthetic polymers may also be present in applications of nanomaterials, e.g., for subsurface remediation. Even in relatively clean laboratory systems, free macromolecules may be present in coated nanoparticle samples because of desorption or incomplete washing. Despite the importance of these scenarios, analysis of the observed effect of excess macromolecules is not typically extended to model their effect on NP deposition.We highlight a recent study by Becker et al. that incorporated the effect of excess polymer in a NP transport model,17 thus, advancing the ability to quantify and predict these effects. This study builds upon a previous study by their group in which atypical deposition behavior, i.e., lower deposition near the inlet of a packed sand column, was observed for CdSe/ZnSe quantum dots (QD) coated with polyacrylic acid–octylamine, PAA–OA.18 This behavior was attributed to the competitive adsorption and site blocking effect of residual polymer onto the sand. In the highlighted article, additional transport experiments were performed on this system. A multi-constituent site blocking (MCB) model was proposed to account for the effect of the excess polymer, and breakthrough curves and retention profiles were fitted to the model. The MCB model included attachment/detachment rates for both the QDs and free PAA–OA, as well as a site blocking function dependent on the area of sorption sites occupied by adsorbed constituents. Taking detachment to be insignificant based on the breakthrough data, the attachment rates for the QDs (kQDa) and PAA–OA (kPAAa) and the maximum retention capacity for the QDs (SQDmax) were estimated by inverse modeling.
Becker et al. found that the breakthrough curves of eluting QDs could be explained using either the MCB or a single-constituent site blocking (SCB) model (Fig. 3a). However, retention profiles showing reduced QD deposition near the column inlet could only be explained using the MCB model (Fig. 3b). This result emphasizes the need for assessment of both breakthrough and retention profiles in order to fully assess NP deposition behavior. Comparing the parameters obtained for the MCB and SCB models, Becker et al. found that the fitted kQDa were similar for both models, whereas the fitted SQDmax was higher for the MCB model, suggesting that PAA–OA acted to block deposition sites rather than change the deposition rate of the QDs.
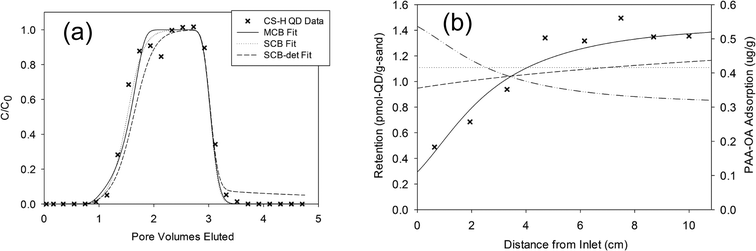 | ||
| Fig. 3 Breakthrough curves for QDs with residual PAA–OA (a), shown here for coarse sand at high flowrate (CS-H), could be successfully fitted with either the MCB model or single-constituent site-blocking models excluding or including a QD detachment parameter (SCB or SCB-det, respectively). On the contrary, lower observed retention of QDs near the column inlet (b) could not be explained with either the SCB or SCB-det model, whereas the MCB model better fitted the data by accounting for site-blocking attributable to adsorption of the residual PAA–OA (dash-dotted line). C/C0 is the eluting QD concentration normalized by the influent concentration, and “pore volumes eluted” is the eluted volume normalized by the total pore volume of the column. Figure reprinted with permission from Becker et al.17 Copyright 2015 Royal Society of Chemistry. | ||
Becker et al. performed a sensitivity analysis on their model to further assess the role of excess PAA–OA on NP deposition. The PAA–OA adsorption rate to sand primarily affected the NP retention near the column inlet, with highest model sensitivity in the regime of slow polymer adsorption rates comparable to that of the NP. For rapid polymer adsorption, NP breakthrough and retention behavior became relatively insensitive to the value of kPAAa. The modeled NP retention decreased with increasing PAA–OA concentration, following expectations for the site-blocking conceptual model.
In conclusion, Becker et al. successfully formulated a multi-constituent site-blocking model to better quantify and predict the effect of polymer adsorption on both the elution and retention behavior of the NPs. Their model comparisons showed that fundamental transport parameters, such as the maximum NP retention capacity, estimated by fitting experimental data can depend greatly on whether the effects of residual polymer are included in the model. Further assessment of the applicability of their model for other systems, such as those containing natural organic matter, would be of high interest, because the authors demonstrated that multi-constituent models will likely be necessary for more accurate prediction of nanoparticle transport in the environment. The authors also recognize that extrapolation of laboratory scale studies should be performed judiciously because residual polymer initially present in samples may be depleted quickly by adsorption to substrates in the environment, resulting in significant estimating errors for NP retention if the effect of the polymer is ignored.
References
- G. Cornelis, Environ. Sci.: Nano, 2015, 2, 19–26 RSC.
- A. L. Dale, G. V. Lowry and E. A. Casman, Environ. Sci.: Nano, 2015, 2, 27–32 RSC.
- A. Praetorius, N. Tufenkji, K. U. Goss, M. Scheringer, F. von der Kammer and M. Elimelech, Environ. Sci.: Nano, 2014, 1, 317–323 RSC.
- P. Westerhoff and B. Nowack, Acc. Chem. Res., 2013, 46, 844–853 CrossRef CAS PubMed.
- A. L. Dale, E. A. Casman, G. V. Lowry, J. R. Lead, E. Viparelli and M. Baalousha, Environ. Sci. Technol., 2015, 49, 2587–2593 CrossRef CAS PubMed.
- E. Goldberg, M. Scheringer, T. D. Bucheli and K. Hungerbuhler, Environ. Sci. Technol., 2014, 48, 12732–12741 CrossRef CAS PubMed.
- S. K. Kumahor, G. H. de Rooij, S. Schlüter and H.-J. Vogel, Vadose Zone J., 2015, 14 DOI:10.2136/vzj2014.08.0105.
- S. K. Kumahor, P. Hron, G. Metreveli, G. E. Schaumann and H.-J. Vogel, Sci. Total Environ. DOI:10.1016/j.scitotenv.2015.03.023 , in press.
- E. M. Hotze, T. Phenrat and G. V. Lowry, J. Environ. Qual., 2010, 39, 1909–1924 CrossRef CAS.
- I. Chowdhury, Y. Hong, R. J. Honda and S. L. Walker, J. Colloid Interface Sci., 2011, 360, 548–555 CrossRef CAS PubMed.
- S. H. Lin and M. R. Wiesner, Environ. Sci. Technol., 2012, 46, 13270–13277 CrossRef CAS PubMed.
- B. A. Legg, M. Q. Zhu, L. R. Comolli, B. Gilbert and J. F. Banfield, Environ. Sci. Technol., 2014, 48, 13703–13710 CrossRef CAS PubMed.
- B. A. Legg, M. Q. Zhu, L. R. Comolli, B. Gilbert and J. F. Banfield, Langmuir, 2014, 30, 9931–9940 CrossRef CAS PubMed.
- K. L. Chen and M. Elimelech, Environ. Sci. Technol., 2008, 42, 7607–7614 CrossRef CAS.
- O. Furman, S. Usenko and B. L. T. Lau, Environ. Sci. Technol., 2013, 47, 1349–1356 CAS.
- S. H. Lin, Y. W. Cheng, J. Liu and M. R. Wiesner, Langmuir, 2012, 28, 4178–4186 CrossRef CAS PubMed.
- M. D. Becker, Y. Wang, K. D. Pennell and L. M. Abriola, Environ. Sci.: Nano, 2015, 2, 155–166 RSC.
- Y. G. Wang, M. D. Becker, V. L. Colvin, L. M. Abriola and K. D. Pennell, Environ. Sci. Technol., 2014, 48, 10664–10671 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |
