Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
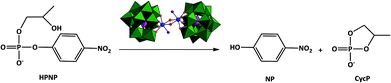
Hydrolysis of the RNA model substrate catalyzed by a binuclear ZrIV-substituted Keggin polyoxometalate†
Thi Kim Nga
Luong
a,
Gregory
Absillis
a,
Pavletta
Shestakova
ab and
Tatjana N.
Parac-Vogt
*a
aDepartment of Chemistry, KU Leuven, Celestijnenlaan 200F, 3001 Leuven, Belgium. E-mail: Tatjana.Vogt@chem.kuleuven.be; Web: http://www.chem.kuleuven.be/lbc/
bNMR Laboratory, Institute of Organic Chemistry with Centre of Phytochemistry, Bulgarian Academy of Sciences, Acad. G. Bontchev Str., B1.9, 1113 Sofia, Bulgaria
First published on 10th August 2015
Abstract
The reactivity and solution behaviour of the binuclear ZrIV-substituted Keggin polyoxometalate (Et2NH2)8[{α-PW11O39Zr(μ-OH)(H2O)}2]·7H2O (ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) towards phosphoester bond hydrolysis of the RNA model substrate 2-hydroxypropyl-4-nitrophenyl phosphate (HPNP) was investigated at different reaction conditions (pD, temperature, concentration, and ionic strength). The hydrolysis of the phosphoester bond of HPNP, followed by means of 1H NMR spectroscopy, proceeded with an observed rate constant, kobs = 11.5(±0.42) × 10−5 s−1 at pD 6.4 and 50 °C, representing a 530-fold rate enhancement in comparison with the spontaneous hydrolysis of HPNP. 1H and 31P NMR spectra indicate that at these reaction conditions the only products of hydrolysis are p-nitrophenol and the corresponding cyclic phosphate ester. The pD dependence of kobs exhibits a bell-shaped profile, with the fastest rate observed at pD 6.4. The formation constant (Kf = 455 M−1) and catalytic rate constant (kc = 42 × 10−5 s−1) for the HPNP–ZrK 2
2) towards phosphoester bond hydrolysis of the RNA model substrate 2-hydroxypropyl-4-nitrophenyl phosphate (HPNP) was investigated at different reaction conditions (pD, temperature, concentration, and ionic strength). The hydrolysis of the phosphoester bond of HPNP, followed by means of 1H NMR spectroscopy, proceeded with an observed rate constant, kobs = 11.5(±0.42) × 10−5 s−1 at pD 6.4 and 50 °C, representing a 530-fold rate enhancement in comparison with the spontaneous hydrolysis of HPNP. 1H and 31P NMR spectra indicate that at these reaction conditions the only products of hydrolysis are p-nitrophenol and the corresponding cyclic phosphate ester. The pD dependence of kobs exhibits a bell-shaped profile, with the fastest rate observed at pD 6.4. The formation constant (Kf = 455 M−1) and catalytic rate constant (kc = 42 × 10−5 s−1) for the HPNP–ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 complex, activation energy (Ea) of 63.35 ± 1.82 kJ mol−1, enthalpy of activation (ΔH‡) of 60.60 ± 2.09 kJ mol−1, entropy of activation (ΔS‡) of −133.70 ± 6.13 J mol−1 K−1, and Gibbs activation energy (ΔG‡) of 102.05 ± 0.13 kJ mol−1 at 37 °C were calculated from kinetic experiments. Binding between ZrK 2
2 complex, activation energy (Ea) of 63.35 ± 1.82 kJ mol−1, enthalpy of activation (ΔH‡) of 60.60 ± 2.09 kJ mol−1, entropy of activation (ΔS‡) of −133.70 ± 6.13 J mol−1 K−1, and Gibbs activation energy (ΔG‡) of 102.05 ± 0.13 kJ mol−1 at 37 °C were calculated from kinetic experiments. Binding between ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and the P–O bond of HPNP was evidenced by the change in the 31P chemical shift and signal line-broadening of the 31P atom in HPNP upon addition of ZrK 2
2 and the P–O bond of HPNP was evidenced by the change in the 31P chemical shift and signal line-broadening of the 31P atom in HPNP upon addition of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. Based on 31P NMR experiments and isotope effect studies, a mechanism for HPNP hydrolysis in the presence of ZrK 2
2. Based on 31P NMR experiments and isotope effect studies, a mechanism for HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was proposed.
2 was proposed.
Introduction
Polyoxometalates (POMs) are generally described as a large class of inorganic oxoclusters that contain early transition metals (V, Nb, Ta, Mo and W) in their highest oxidation state. POMs have applications in a broad range of research domains including material science,1,2 medicine3 and catalysis.4,5 Recently our group discovered that a series of ZrIV-substituted POM complexes could act as artificial phosphosesterases. ZrIV is ideally suited as an active center in artificial hydrolytic metalloenzymes because of its high Lewis-acidity and oxophilic properties, allowing both the coordination and activation of the substrate and nucleophile. Model DNA compounds such as 4-nitrophenyl phosphate (NPP) and bis-4-nitrophenyl phosphate (BNPP) are often used when testing the reactivity of newly developed artificial phosphatases and nucleases.6–12 The advantage of these model DNA substrates is the presence of good leaving groups in their structures, increasing their reactivity in comparison to for example DNA and RNA biomolecules. Phosphoester bond hydrolysis in RNA is an intramolecular transesterification, supported by the attack of the 2′-OH group to the phosphate backbone. As a result, RNA is hydrolyzed faster than DNA, which lacks such internal nucleophile. Because of the presence of an OH-group 2-hydroxypropyl-4-nitrophenylphosphate (HPNP, Scheme 1) is often used as a RNA model substrate to test the reactivity of new artificial nucleases.13,14A mononuclear ZrIV-substituted Wells–Dawson type POM K15H[Zr(α2-P2W17O61)2]·25H2O was demonstrated to be able to catalytically enhance the hydrolysis rate of the DNA model compound NPP by nearly two orders of magnitude.8 In a previous study we also found that the dinuclear ZrIV-substituted Keggin type POM (Et2NH2)8[{α-PW11O39Zr(μ-OH)(H2O)}2]·7H2O (ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, Fig. 1) efficiently promoted hydrolysis of the extremely stable phosphodiester BNPP, with a 320-fold rate enhancement in comparison with spontaneous hydrolysis.12 In addition, it was shown that the monomeric [α-PW11O39Zr(μ-OH)(H2O)]4− (ZrK 1
2, Fig. 1) efficiently promoted hydrolysis of the extremely stable phosphodiester BNPP, with a 320-fold rate enhancement in comparison with spontaneous hydrolysis.12 In addition, it was shown that the monomeric [α-PW11O39Zr(μ-OH)(H2O)]4− (ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) species is present in aqueous solution at near-neutral pD and that this species is responsible for the hydrolysis of the phosphoester bond in BNPP.6 ZrK 1
1) species is present in aqueous solution at near-neutral pD and that this species is responsible for the hydrolysis of the phosphoester bond in BNPP.6 ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is considered to be more catalytically active when compared to ZrK 2
1 is considered to be more catalytically active when compared to ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 as its Zr(IV) ion has more coordinated water molecules that can be replaced by the substrate or that can act as a nucleophile. Theoretical studies have shown that when ZrK 1
2 as its Zr(IV) ion has more coordinated water molecules that can be replaced by the substrate or that can act as a nucleophile. Theoretical studies have shown that when ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 interacts with NPP or BNPP, it prefers to form monodentate complexes which are more stable than the corresponding bidentate complexes.6
1 interacts with NPP or BNPP, it prefers to form monodentate complexes which are more stable than the corresponding bidentate complexes.6
Although natural nuclease enzymes play a critical role in various biotechnology applications, efficient artificial nucleases are still needed.15 For example, artificial restriction enzyme can be made sequence specific by encoding sequence specificity into the cleavage agent or by linking it to an appropriate DNA binding agent.15 Such enzymes are not only very useful for molecular biological field such as DNA cutting at a site not recognized by current restriction enzymes, but also for genomic analysis. In addition, artificial nucleases can be synthesized in large amounts16 and are often used to elucidate the precise role of active metal ions in natural nucleases.15 The metal ion accelerates the hydrolysis process by activation of the phosphodiester bond, activation and/or delivery of the nucleophile, and by stabilization of the pentacoordination transition state and the leaving group.17,18
In this study we explore the phosphoesterase activity of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 towards the RNA model system HPNP, a stable phosphodiester characterized by a half-life of hundreds of days at pH 8.0 and 30 °C.19 As a previous study showed that the solution speciation of this POM is highly dependent on several factors such as pD, temperature, initial concentration, ionic strength and substrate concentration,12 the speciation behavior of ZrK 2
2 towards the RNA model system HPNP, a stable phosphodiester characterized by a half-life of hundreds of days at pH 8.0 and 30 °C.19 As a previous study showed that the solution speciation of this POM is highly dependent on several factors such as pD, temperature, initial concentration, ionic strength and substrate concentration,12 the speciation behavior of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at the conditions used to study HPNP hydrolysis was studied in detail. These results are needed to understand the kinetic experiments and to propose a mechanism for HPNP hydrolysis in the presence of ZrK 2
2 at the conditions used to study HPNP hydrolysis was studied in detail. These results are needed to understand the kinetic experiments and to propose a mechanism for HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 POM.
2 POM.
Results and discussion
Aqueous study of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2
2
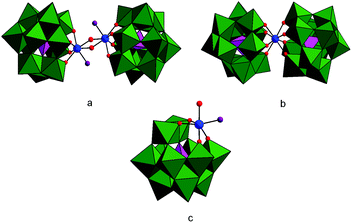
Depending on the pD, temperature, reaction time, starting concentration, and ionic strength, ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 having a single 31P resonance at −13.49 ppm20 (Fig. 1a) can convert into the catalytically less active ZrK 1
2 having a single 31P resonance at −13.49 ppm20 (Fig. 1a) can convert into the catalytically less active ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (Fig. 1b), characterized by two 31P resonances at −14.67 and −14.77 ppm.12,21,22 However, recent 31P DOSY study on a 3.0 mM aqueous solution of ZrK 2
2 (Fig. 1b), characterized by two 31P resonances at −14.67 and −14.77 ppm.12,21,22 However, recent 31P DOSY study on a 3.0 mM aqueous solution of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 also showed the presence of the 1
2 also showed the presence of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ZrIV-substituted Keggin type species (Fig. 1c) with a 31P resonance at −13.7 ppm. More importantly, the DFT calculations identified this species as the active species responsible for phosphoester bond hydrolysis in BNPP at pD 6.4 and 60 °C.6 Therefore, the aim of this aqueous solution study is to further optimize reaction conditions in such a way that the ZrK 1
1 ZrIV-substituted Keggin type species (Fig. 1c) with a 31P resonance at −13.7 ppm. More importantly, the DFT calculations identified this species as the active species responsible for phosphoester bond hydrolysis in BNPP at pD 6.4 and 60 °C.6 Therefore, the aim of this aqueous solution study is to further optimize reaction conditions in such a way that the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 concentration is increased and to apply these conditions to determine the kinetics of HPNP hydrolysis in the presence of ZrK 2
1 concentration is increased and to apply these conditions to determine the kinetics of HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. To the best of our knowledge the synthesis and isolation of pure ZrK 1
2. To the best of our knowledge the synthesis and isolation of pure ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at neutral pD conditions has not been reported. ZrK 1
1 at neutral pD conditions has not been reported. ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is only present as a single species in acidic solutions (pH ≤ 3.4),6 while the monolacunary Keggin species, a precursor of ZrK 1
1 is only present as a single species in acidic solutions (pH ≤ 3.4),6 while the monolacunary Keggin species, a precursor of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, only exists at near-neutral pH.23
1, only exists at near-neutral pH.23
The effect of pD on the solution behaviour of 1.0 mM ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was recently reported. It was concluded that ZrK 2
2 was recently reported. It was concluded that ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and ZrK 1
2 and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 are present in equilibrium in solutions with pD of 5.4 and 6.4. At pD values higher than 6.4, the dominant species is ZrK 2
1 are present in equilibrium in solutions with pD of 5.4 and 6.4. At pD values higher than 6.4, the dominant species is ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, while at pD values lower than 5.4, the dominant species is ZrK 1
2, while at pD values lower than 5.4, the dominant species is ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.6 In this study the effect of temperature on the speciation equilibria was determined at nearly neutral pD (pD 6.4).
1.6 In this study the effect of temperature on the speciation equilibria was determined at nearly neutral pD (pD 6.4).
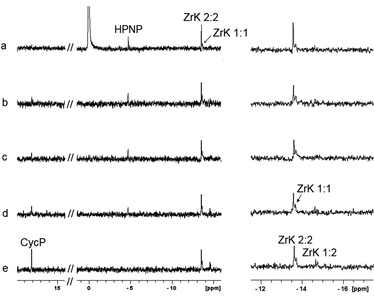
A ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 solution was kept at temperatures ranging from 37 °C to 80 °C, before assessing its species distribution by 31P NMR. Fig. S1† shows that both ZrK 2
2 solution was kept at temperatures ranging from 37 °C to 80 °C, before assessing its species distribution by 31P NMR. Fig. S1† shows that both ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (−13.49 ppm)12 and ZrK 1
2 (−13.49 ppm)12 and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (−13.7 ppm)6 are present after mixing for 1 h without heating. Unfortunately, partial overlap of the ZrK 1
1 (−13.7 ppm)6 are present after mixing for 1 h without heating. Unfortunately, partial overlap of the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and ZrK 1
2 and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 resonances makes integration and therefore quantification difficult. Temperature has no significant effect on the speciation of ZrK 2
1 resonances makes integration and therefore quantification difficult. Temperature has no significant effect on the speciation of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 after heating for 1 h up to 60 °C. From 70 °C upward only minor amounts (19%) of ZrK 1
2 after heating for 1 h up to 60 °C. From 70 °C upward only minor amounts (19%) of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 were detected, ZrK 1
2 were detected, ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is present up to 70 °C, and at 80 °C its intensity decreases significantly.
1 is present up to 70 °C, and at 80 °C its intensity decreases significantly.
To further observe the influence of concentration and incubation time on the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 species distribution, a 1.0 mM and 3.0 mM solution of ZrK 2
1 species distribution, a 1.0 mM and 3.0 mM solution of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at pD 6.4 were kept at 50 °C for a prolonged period of time. Fig. S2† indicates that for 1.0 mM of ZrK 2
2 at pD 6.4 were kept at 50 °C for a prolonged period of time. Fig. S2† indicates that for 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 both ZrK 2
2 both ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and ZrK 1
2 and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were detected after mixing and ZrK 1
1 were detected after mixing and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was still present in the solution after heating for 7 h. For a 3.0 mM ZrK 2
1 was still present in the solution after heating for 7 h. For a 3.0 mM ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 solution it can be seen from Fig. S3† that no ZrK 1
2 solution it can be seen from Fig. S3† that no ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was detected after mixing and after 2 h 25 min, a very small amount of ZrK 1
1 was detected after mixing and after 2 h 25 min, a very small amount of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was observed.
1 was observed.
Previous studies show that ionic strength has an effect on the POM solution behavior and/or the hydrolysis rate of different molecules such as NPP, BNPP and dipeptide.10,12,24,25 Therefore, the influence of ionic strength on the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 species distribution was also studied for a 1.0 mM sample of ZrK 2
1 species distribution was also studied for a 1.0 mM sample of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. 31P NMR spectra of a 1.0 mM solution of ZrK 2
2. 31P NMR spectra of a 1.0 mM solution of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 in the presence of increasing concentrations (0.25 M to 2.0 M) of NaClO4 at pD 6.4 were recorded after mixing. Fig. S4† shows that an addition of 0.25 M NaClO4 lead to the disappearance of ZrK 1
2 in the presence of increasing concentrations (0.25 M to 2.0 M) of NaClO4 at pD 6.4 were recorded after mixing. Fig. S4† shows that an addition of 0.25 M NaClO4 lead to the disappearance of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 signal while the signals of ZrK 2
1 signal while the signals of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and ZrK 1
2 and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 were clearly detected. This means that NaClO4 can promote the conversion of ZrK 1
2 were clearly detected. This means that NaClO4 can promote the conversion of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 into ZrK 2
1 into ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 or ZrK 1
2 or ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 212,26,27 and higher concentrations of NaClO4 favoured the conversion of ZrK 2
212,26,27 and higher concentrations of NaClO4 favoured the conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.
2.
In this study, the influence of the substrate HPNP and the inhibitor disodium diphenyl phosphate (DPP) on the equilibria between the different ZrIV-substituted Keggin type POMs was also examined. The concentration of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was kept constant (1.0 mM) and the concentration of HPNP was increased from 1.0 mM to 50.0 mM. As can be seen from Fig. S5,† an increase in HPNP concentration lead to the gradual disappearance of ZrK 1
2 was kept constant (1.0 mM) and the concentration of HPNP was increased from 1.0 mM to 50.0 mM. As can be seen from Fig. S5,† an increase in HPNP concentration lead to the gradual disappearance of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and promoted the conversion of ZrK 2
1 and promoted the conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The same trend was seen when using DPP in the concentration range 1.0 mM to 40.0 mM (Fig. S6†). These experiments suggest that the interaction of ZrK 2
2. The same trend was seen when using DPP in the concentration range 1.0 mM to 40.0 mM (Fig. S6†). These experiments suggest that the interaction of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 with HNPP and DPP ligands also plays an important role in the ZrK 1
2 with HNPP and DPP ligands also plays an important role in the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 species distribution.
1 species distribution.
The above results show that most ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was observed using a 1.0 mM solution of ZrK 2
1 was observed using a 1.0 mM solution of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 in the absence of salt at pD 6.4 and 50 °C. Therefore, these conditions will be used in the kinetic studies of HPNP hydrolysis.
2 in the absence of salt at pD 6.4 and 50 °C. Therefore, these conditions will be used in the kinetic studies of HPNP hydrolysis.
Hydrolysis of HPNP in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2
2
The hydrolysis of HPNP in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (Scheme 1) is conveniently followed by means of 1H NMR spectroscopy. P–O bond hydrolysis can be observed by the disappearance of the aromatic resonances of HPNP (8.28 ppm and 7.38 ppm) and the appearance of the aromatic p-nitrophenyl (NP) resonances at 8.19 ppm and 6.96 ppm. Fig. 2 shows an example of the 1H NMR spectra for HPNP hydrolysis in the presence of ZrK 2
2 (Scheme 1) is conveniently followed by means of 1H NMR spectroscopy. P–O bond hydrolysis can be observed by the disappearance of the aromatic resonances of HPNP (8.28 ppm and 7.38 ppm) and the appearance of the aromatic p-nitrophenyl (NP) resonances at 8.19 ppm and 6.96 ppm. Fig. 2 shows an example of the 1H NMR spectra for HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at different time intervals.
2 at different time intervals.
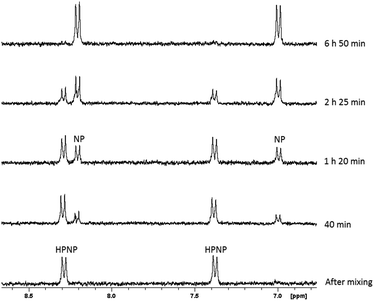 | ||
Fig. 2
1H NMR of the hydrolysis reaction of 1.0 mM of HPNP in the presence of 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at different time intervals at pD 6.4 and 50 °C. 2 at different time intervals at pD 6.4 and 50 °C. | ||
Based on the 1H NMR integration values from Fig. 2, the percentage of HPNP at different time increments was calculated in order to obtain the observed rate constant, and half-life. The percentage of HPNP and NP as a function of reaction time are shown in Fig. S7† and the natural logarithm of the concentration of NPP as a function of time (Fig. S8†) was fitted to a first-order linear decay function. At pD 6.4 and 50 °C an observed rate constant of 11.5(±0.42) × 10−5 s−1 and half-life of 1.7 h were calculated. The presence of equimolar amounts of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 resulted in a 530-fold rate acceleration in comparison with the spontaneous hydrolysis of HPNP (2.17(±0.11) × 10−7 s−1) under the same reaction conditions.
2 resulted in a 530-fold rate acceleration in comparison with the spontaneous hydrolysis of HPNP (2.17(±0.11) × 10−7 s−1) under the same reaction conditions.
31P NMR spectra (Fig. 3) recorded during the course of the reaction show that cyclic phosphate (CycP), with a 31P resonance at 18 ppm, is formed.19,28 At the end of hydrolysis, no HPNP resonances were detected in both 1H and 31P NMR spectra, indicating full conversion of HPNP into cyclic phosphate and NP.
Several control experiments were performed. At pD 6.4 and 50 °C, the reaction between HPNP and the monolacunary Keggin POM ([α-PW11O39]7−) did not show any change in observed rate constant when compared to spontaneous HPNP cleavage. This experiment indicates that the monolacunary POM does not promote HPNP hydrolysis and that the embedded Zr(IV) ions are responsible for the observed reactivity. Under identical conditions HPNP hydrolysis promoted by ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was about 12 times slower (kobs = 9.20(±0.56) × 10−6 s−1) in comparison to ZrK 2
2 was about 12 times slower (kobs = 9.20(±0.56) × 10−6 s−1) in comparison to ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 promoted hydrolysis.
2 promoted hydrolysis.
As ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 does not show any conversion into other POM species up to concentrations of 6.0 mM (pD 6.4 and 60 °C),12 the observed hydrolysis is exclusively the result of the presence of ZrK 1
2 does not show any conversion into other POM species up to concentrations of 6.0 mM (pD 6.4 and 60 °C),12 the observed hydrolysis is exclusively the result of the presence of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. This difference can be explained by the fact that in the case of ZrK 2
2. This difference can be explained by the fact that in the case of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, ZrK 2
2, ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 partially converted into a more active ZrK 1
2 partially converted into a more active ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 species and this species promoted the reaction resulting in the higher rate constant, while in the case of ZrK 1
1 species and this species promoted the reaction resulting in the higher rate constant, while in the case of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, no ZrK 1
2, no ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 species was detected. Under the same conditions, the reaction between the ZrCl2O·8H2O and HPNP was also examined. Under these conditions, the formation of insoluble Zr(IV) hydroxyl polymeric gels29,30 was observed, resulting in a rate constant of 2.83(±0.30) × 10−6 s−1. This rate constant represents a 40-fold decrease in comparison to HPNP hydrolysis in the presence of ZrK 2
1 species was detected. Under the same conditions, the reaction between the ZrCl2O·8H2O and HPNP was also examined. Under these conditions, the formation of insoluble Zr(IV) hydroxyl polymeric gels29,30 was observed, resulting in a rate constant of 2.83(±0.30) × 10−6 s−1. This rate constant represents a 40-fold decrease in comparison to HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.
2.
Effect of pD on HPNP hydrolysis
Since the reactivity and speciation of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is strongly affected by pD, the effect of pD on the rate constant of HPNP hydrolysis in the absence and in the presence of ZrK 2
2 is strongly affected by pD, the effect of pD on the rate constant of HPNP hydrolysis in the absence and in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was determined in the pD range 4.5 to 9.5. In the absence of ZrK 2
2 was determined in the pD range 4.5 to 9.5. In the absence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at pD 4.5 and 5.5, no hydrolysis of HPNP was observed after 3 months, while in the pD range 6.4 to 9.5, the rate constants are at least two orders of magnitude slower than that in the presence of ZrK 2
2 at pD 4.5 and 5.5, no hydrolysis of HPNP was observed after 3 months, while in the pD range 6.4 to 9.5, the rate constants are at least two orders of magnitude slower than that in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (Table S1†). The pD-rate constant profile for HPNP hydrolysis in the presence of ZrK 2
2 (Table S1†). The pD-rate constant profile for HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is bell-shaped, with fastest hydrolysis observed at pD 6.4 (Fig. 4).
2 is bell-shaped, with fastest hydrolysis observed at pD 6.4 (Fig. 4).
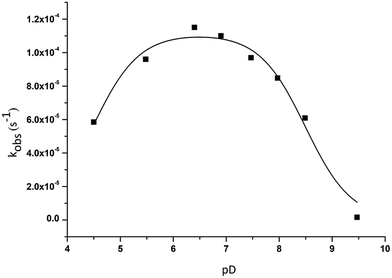 | ||
Fig. 4 pD dependence profile of kobs for the cleavage of 1.0 mM of HPNP in the presence of 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at 50 °C. 2 at 50 °C. | ||
This bell-shaped profile further supports a Lewis-acid catalyzed hydrolysis mechanism because if hydrolysis would be Brønsted acid mediated, the rate constants would increase at low pH values. The bell-shaped profile can be explained in the following way. HPNP hydrolysis in the presence of this POM requires OH− as an active nucleophile and this OH group can come from either the POM or solvent water molecules.6 The deprotonation of water molecules is hindered at pD values smaller than 6.4, resulting in a decrease of kobs at low pD values. The formation of OH− nucleophiles is facilitated by an increase of pD, leading to a gradual increase of kobs. After reaching a maximum at near neutral pD a decrease of kobs is usually observed due to the formation of a hydrolytically inactive HPNP coordination complex. 31P NMR shows that during the reaction performed at pD 4.5 (Fig. S9†) only ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was observed, suggesting that indeed lower reaction rates are not due to the conversion of catalytically more active ZrK 1
1 was observed, suggesting that indeed lower reaction rates are not due to the conversion of catalytically more active ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 into ZrK 2
1 into ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 or ZrK 1
2 or ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, but rather due to the low concentration of OH− nucleophiles present at low pD. The deprotonation of water nucleophiles as well as the promotion of the conversion of ZrK 2
2, but rather due to the low concentration of OH− nucleophiles present at low pD. The deprotonation of water nucleophiles as well as the promotion of the conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into less active ZrK 1
2 into less active ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 become more evident at pD values higher than 6.4 when kobs of HPNP hydrolysis starts to decrease.
2 become more evident at pD values higher than 6.4 when kobs of HPNP hydrolysis starts to decrease.
The conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 during the course of reaction was evidenced in the reaction mixtures studied at different pD values and 50 °C. Fig. 3 shows that only 11% of ZrK 1
2 during the course of reaction was evidenced in the reaction mixtures studied at different pD values and 50 °C. Fig. 3 shows that only 11% of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was observed after 2 h 25 min at pD 6.4, while in highly alkaline solutions (pD 9.5) further decomposition of ZrK 2
2 was observed after 2 h 25 min at pD 6.4, while in highly alkaline solutions (pD 9.5) further decomposition of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into the hydrolytically inactive monolacunary species was observed immediately after mixing (Fig. S10†), resulting in a complete loss of catalytic activity.
2 into the hydrolytically inactive monolacunary species was observed immediately after mixing (Fig. S10†), resulting in a complete loss of catalytic activity.
Effect of temperature on HPNP hydrolysis
As the temperature also influences the ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 solution equilibria, the effect of temperature on the hydrolysis reaction rate was examined on a solution containing equimolar amounts of HPNP and ZrK 2
2 solution equilibria, the effect of temperature on the hydrolysis reaction rate was examined on a solution containing equimolar amounts of HPNP and ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (1.0 mM) at pD 6.4 in the temperature range from 37 °C to 80 °C. From the data shown in Fig. S11a,† the activation energy (63.35 ± 1.82 kJ mol−1) was calculated from the Arrhenius equation, which is significantly lower than the one in the absence of ZrK 2
2 (1.0 mM) at pD 6.4 in the temperature range from 37 °C to 80 °C. From the data shown in Fig. S11a,† the activation energy (63.35 ± 1.82 kJ mol−1) was calculated from the Arrhenius equation, which is significantly lower than the one in the absence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 under identical reaction conditions (131.64 ± 2.73 kJ mol−1).
2 under identical reaction conditions (131.64 ± 2.73 kJ mol−1).
Based on the Eyring equation (Fig. S11b†), the enthalpy of activation (ΔH‡ = 60.60 ± 2.09 kJ mol−1) and entropy of activation (ΔS‡ = −133.70 ± 6.13 J mol−1 K) were calculated. The negative entropy of activation results from the coordination of HPNP to the ZrK POM catalyst. Out of these data, the Gibbs activation energy (ΔG‡) was calculated to be 102.05 ± 0.13 kJ mol−1 at 37 °C. This ΔG‡ value is similar to that of BNPP hydrolysis (111.12 kJ mol−1 at 37 °C) by ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 212 and of NPP hydrolysis (96.94 kJ mol−1 at 37 °C) by the Wells–Dawson type K15H[Zr(α2-P2W17O61)2]·25H2O POM.8
212 and of NPP hydrolysis (96.94 kJ mol−1 at 37 °C) by the Wells–Dawson type K15H[Zr(α2-P2W17O61)2]·25H2O POM.8
Although ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 speciation studies have shown that high temperatures favor conversion of ZrK 2
2 speciation studies have shown that high temperatures favor conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and/or ZrK 1
1 and/or ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, the rate of HPNP hydrolysis did increase when the temperature was increased. This result might be explained by the fact that conversion of ZrK 2
2, the rate of HPNP hydrolysis did increase when the temperature was increased. This result might be explained by the fact that conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is rather slow and ZrK 1
2 is rather slow and ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is still present during the course of hydrolysis.
1 is still present during the course of hydrolysis.
Effect of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2 concentration on HPNP hydrolysis
2 concentration on HPNP hydrolysis
In order to observe the influence of catalyst concentration on the reaction rate, reaction mixtures containing 1.0 mM HPNP and increasing amounts of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, ranging from 0.25 mM to 3.0 mM were studied at pD 6.4 and 50 °C. The general catalytic scheme for the hydrolysis of HPNP in the presence of ZrK 2
2, ranging from 0.25 mM to 3.0 mM were studied at pD 6.4 and 50 °C. The general catalytic scheme for the hydrolysis of HPNP in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is shown in Scheme 2.
2 is shown in Scheme 2.
Assuming that the formation of the products (kc) is much slower than reaching the equilibrium between HPNP and ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, we have k1 + k−1 ≫ kc. Because the formation of nitrophenol (NP) is a first-order reaction, kobs can be written as in eqn (1).8,10,11,31
2, we have k1 + k−1 ≫ kc. Because the formation of nitrophenol (NP) is a first-order reaction, kobs can be written as in eqn (1).8,10,11,31
By fitting the data to eqn (1), the binding constant (Kf = k1/k−1 = 455 M−1) and the catalytic rate constant (kc = 42 × 10−5 s−1) were obtained (Fig. 5). From this figure it is evident that the POM can hydrolyse an excess of HPNP. Complete hydrolysis of HPNP was still observed when 1.0 mM of HPNP and 0.25 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 were used. This suggests that one equivalent of ZrK 2
2 were used. This suggests that one equivalent of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 hydrolyses at least 4 equivalents of HPNP, demonstrating its catalytic activity.
2 hydrolyses at least 4 equivalents of HPNP, demonstrating its catalytic activity.
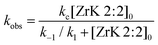 | (1) |
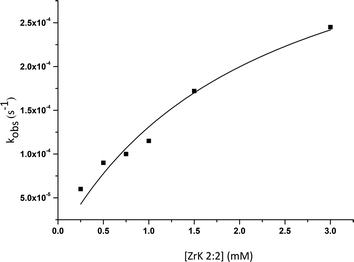 | ||
Fig. 5 Dependence of the observed rate constant on the concentration of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 for the hydrolysis of 1.0 mM of HPNP at pD 6.4 and 50 °C. 2 for the hydrolysis of 1.0 mM of HPNP at pD 6.4 and 50 °C. | ||
Fig. 5 shows that an increase in ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 concentration leads to an increase in reaction rate. Despite the fact that higher initial concentrations of ZrK 2
2 concentration leads to an increase in reaction rate. Despite the fact that higher initial concentrations of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 result in a more favorable conversion to ZrK 1
2 result in a more favorable conversion to ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2,12 the total molar amount of ZrK 2
2,12 the total molar amount of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 in solution is still much higher, resulting in faster hydrolysis. For example, for a 0.5 mL mixture containing 1.0 mM of HPNP and 0.25 mM of ZrK 2
2 in solution is still much higher, resulting in faster hydrolysis. For example, for a 0.5 mL mixture containing 1.0 mM of HPNP and 0.25 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (0.13 × 10−6 mole), no signal of ZrK 1
2 (0.13 × 10−6 mole), no signal of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was detected during the course of reaction (Fig. S12†). Fig. 3 shows that a 0.5 mL mixture of 1.0 mM of HPNP and 1.0 mM of ZrK 2
2 was detected during the course of reaction (Fig. S12†). Fig. 3 shows that a 0.5 mL mixture of 1.0 mM of HPNP and 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 contains 81% of ZrK 2
2 contains 81% of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 after 2 h 25 min, resulting in 0.43 × 10−6 mole of ZrK 2
2 after 2 h 25 min, resulting in 0.43 × 10−6 mole of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. Similarly, when 3.0 mM of ZrK 2
2. Similarly, when 3.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 were used the conversion to ZrK 1
2 were used the conversion to ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was larger and 71% of ZrK 2
2 was larger and 71% of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was present after 2 h 25 min (Fig. S13†), leading to 1.06 × 10−6 mole of ZrK 2
2 was present after 2 h 25 min (Fig. S13†), leading to 1.06 × 10−6 mole of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 present in solution.
2 present in solution.
Effect of ionic strength on HPNP hydrolysis
Since both ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and HPNP are negatively charged at pD 6.4, the influence of ionic strength on HPNP hydrolysis was examined by adding stepwise NaClO4 to a mixture containing 1.0 mM of HPNP and 1.0 mM of ZrK 2
2 and HPNP are negatively charged at pD 6.4, the influence of ionic strength on HPNP hydrolysis was examined by adding stepwise NaClO4 to a mixture containing 1.0 mM of HPNP and 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. From Fig. S14,† a significant decrease of the rate constant was observed upon adding increasing amounts of NaClO4. As above control experiments with the monolacunary Keggin POM ([α-PW11O39]7−) showed that ZrIV is required factor for the catalytic activity, it is very likely that the binding between HPNP and POM occurs via coordination between the negatively charged oxygen of phosphate group of HPNP and the positively charged ZrIV of POM. Addition of salt may hinder this interaction, leading lower reaction rates. However, NaClO4 also promotes conversion of ZrK 2
2. From Fig. S14,† a significant decrease of the rate constant was observed upon adding increasing amounts of NaClO4. As above control experiments with the monolacunary Keggin POM ([α-PW11O39]7−) showed that ZrIV is required factor for the catalytic activity, it is very likely that the binding between HPNP and POM occurs via coordination between the negatively charged oxygen of phosphate group of HPNP and the positively charged ZrIV of POM. Addition of salt may hinder this interaction, leading lower reaction rates. However, NaClO4 also promotes conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 as can be seen from Fig. S15, S16 and S17.† Fig. S15† shows that at pD 6.4, 26% of ZrK 1
2 as can be seen from Fig. S15, S16 and S17.† Fig. S15† shows that at pD 6.4, 26% of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is detected in a reaction mixture containing 0.25 M NaClO4, while in the presence of 1.0 M NaClO4 (Fig. S16†) 70% of ZrK 1
2 is detected in a reaction mixture containing 0.25 M NaClO4, while in the presence of 1.0 M NaClO4 (Fig. S16†) 70% of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was observed. When the concentration of NaClO4 was increased up to 2.0 M (Fig. S17†), full conversion of ZrK 2
2 was observed. When the concentration of NaClO4 was increased up to 2.0 M (Fig. S17†), full conversion of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 into ZrK 1
2 into ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was observed immediately after mixing. From these experiments, it is concluded that NaClO4 not only plays a role in the binding between HPNP and ZrK 2
2 was observed immediately after mixing. From these experiments, it is concluded that NaClO4 not only plays a role in the binding between HPNP and ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, but also in the speciation of this POM in the reaction mixture.
2, but also in the speciation of this POM in the reaction mixture.
Inhibition study
As DPP is not hydrolyzed after 4 months both in the absence and in the presence of 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at pD 6.4 and 60 °C,12 it is a good candidate for inhibitor studies. The hydrolysis of 1.0 mM of HPNP was followed in the presence of 1.0 mM of ZrK 2
2 at pD 6.4 and 60 °C,12 it is a good candidate for inhibitor studies. The hydrolysis of 1.0 mM of HPNP was followed in the presence of 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and increasing amounts from 1.0 mM to 40 mM of DPP. As it can be seen from Fig. S18,† the rate constant of HPNP hydrolysis significantly decreased upon adding increasing amounts of DPP. DPP has a negative effect, as on the one hand it competes with HPNP for the binding to ZrIV and on the other hand it largely shifts equilibrium toward the formation of ZrK 1
2 and increasing amounts from 1.0 mM to 40 mM of DPP. As it can be seen from Fig. S18,† the rate constant of HPNP hydrolysis significantly decreased upon adding increasing amounts of DPP. DPP has a negative effect, as on the one hand it competes with HPNP for the binding to ZrIV and on the other hand it largely shifts equilibrium toward the formation of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, resulting in reaction rate decrease.
2, resulting in reaction rate decrease.
Binding between HPNP and ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2 by 31P NMR
2 by 31P NMR
The interaction between HPNP and ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was studied by 31P NMR. At pD 6.4 (Fig. S19†) ZrK 2
2 was studied by 31P NMR. At pD 6.4 (Fig. S19†) ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is characterized by a peak at −13.49 ppm, while the HPNP signal can be found at −4.61 ppm (half-width 2.30 Hz). In the presence of 1.0 mM of ZrK 2
2 is characterized by a peak at −13.49 ppm, while the HPNP signal can be found at −4.61 ppm (half-width 2.30 Hz). In the presence of 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 the peak of HPNP is shifted by 0.14 ppm and the half-width increased to 5.14 Hz (Fig. 6). Both the change in chemical shift and the line-broadening support that interaction between HPNP and ZrK 2
2 the peak of HPNP is shifted by 0.14 ppm and the half-width increased to 5.14 Hz (Fig. 6). Both the change in chemical shift and the line-broadening support that interaction between HPNP and ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 takes place.
2 takes place.
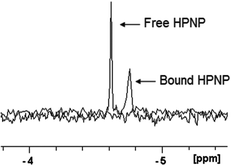 | ||
Fig. 6
31P NMR spectra of 1.0 mM of HPNP and the mixture of 1.0 mM of HPNP and 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 after adjusting the pD to 6.4 (400 MHz, D2O, 293 K, NS = 1024, 25% H3PO4). 2 after adjusting the pD to 6.4 (400 MHz, D2O, 293 K, NS = 1024, 25% H3PO4). | ||
Kinetic isotope effect on HPNP hydrolysis and proposed mechanism
The kinetic solvent deuterium isotope effect is a powerful tool for mechanistic studies and gives information on the proton transfer processes in or before the rate-limiting step of the reaction.32 RNA model phosphate diesters can be hydrolysed by two possible mechanisms that can be distinguished by examining the influence of H2O or D2O as a solvent on the kinetics of hydrolysis. When the solvent deuterium kinetic isotope effect (skie), characterized by the kH2O/kD2O ratio, is lower than 1.5 a nucleophilic mechanism is expected. At skie ratios higher than 2 the reaction preceeds via a general base mechanism.28,33In order to propose a mechanism for HPNP hydrolysis, the skie was calculated. The reaction rates for 1.0 mM HPNP hydrolysis in the presence of 1.0 mM ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 in H2O or D2O were calculated at identical conditions (pD 6.4 and 50 °C). The hydrolysis in H2O proceeded with the rate constant 13.80(±0.83) × 10−5 s−1, while for the reaction in D2O a rate constant of 11.50(±0.42) × 10−5 s−1 was observed. From these results, a skie effect of 1.2 was calculated and this value of skie effect supports the nucleophilic mechanism.28,331H spectra (Fig. 2) show that one of the products of HPNP hydrolysis is nitrophenol and 31P spectra (Fig. 3) show that another product of HPNP hydrolysis is cyclic phosphate with a 31P chemical shift at 18 ppm.19,28 From 31P NMR measurements, kinetic experiments and isotope effect study, the mechanism of HPNP hydrolysis in the presence of ZrK 2
2 in H2O or D2O were calculated at identical conditions (pD 6.4 and 50 °C). The hydrolysis in H2O proceeded with the rate constant 13.80(±0.83) × 10−5 s−1, while for the reaction in D2O a rate constant of 11.50(±0.42) × 10−5 s−1 was observed. From these results, a skie effect of 1.2 was calculated and this value of skie effect supports the nucleophilic mechanism.28,331H spectra (Fig. 2) show that one of the products of HPNP hydrolysis is nitrophenol and 31P spectra (Fig. 3) show that another product of HPNP hydrolysis is cyclic phosphate with a 31P chemical shift at 18 ppm.19,28 From 31P NMR measurements, kinetic experiments and isotope effect study, the mechanism of HPNP hydrolysis in the presence of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was proposed as following. As already shown in our previous study, under the reaction conditions there is an interconversion between ZrK 2
2 was proposed as following. As already shown in our previous study, under the reaction conditions there is an interconversion between ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, ZrK 1
2, ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and/or ZrK 1
1 and/or ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.6 The Zr atom in the ZrK 1
2.6 The Zr atom in the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 species is coordinatively saturated and hardly accessible for the bulky HPNP ligand, as it is sandwiched by the two Keggin moieties. Therefore, HPNP coordination to ZrK 1
2 species is coordinatively saturated and hardly accessible for the bulky HPNP ligand, as it is sandwiched by the two Keggin moieties. Therefore, HPNP coordination to ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 is not expected.6 ZrK 1
2 is not expected.6 ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was found to be more catalytically active when compared to ZrK 2
1 was found to be more catalytically active when compared to ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 as its Zr(IV) ion has more coordinated water molecules that can be replaced by the substrate or that can act as a nucleophile.6 We suggest that ZrK 1
2 as its Zr(IV) ion has more coordinated water molecules that can be replaced by the substrate or that can act as a nucleophile.6 We suggest that ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 can act as an active species in this HPNP hydrolytic reaction by coordination to the oxygen atom of the phosphate group of HPNP. This suggestion is experimentally supported by the observed shift and line-broadening of the HPNP signal in the 31P spectrum (Fig. 6). This binding would result in a more positive charge on the phosphorous atom of HPNP, which facilitates the nucleophilic attack of the OH group of ZrK 1
1 can act as an active species in this HPNP hydrolytic reaction by coordination to the oxygen atom of the phosphate group of HPNP. This suggestion is experimentally supported by the observed shift and line-broadening of the HPNP signal in the 31P spectrum (Fig. 6). This binding would result in a more positive charge on the phosphorous atom of HPNP, which facilitates the nucleophilic attack of the OH group of ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 or from water molecule of the solvent, thus leading to cleavage of the P–O bond and the formation of nitrophenol and cyclic phosphate.
1 or from water molecule of the solvent, thus leading to cleavage of the P–O bond and the formation of nitrophenol and cyclic phosphate.
Conclusions
In summary, we report the first example of the hydrolysis of HPNP promoted by a binuclear Zr(IV)-substituted Keggin type polyoxometalate. 31P NMR measurements offer the evidence that the ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 species, which is proposed as the active species, is the major and stable species under the reaction conditions. A 530-fold rate increase in comparison to the blank reaction was observed for HPNP hydrolysis. The present study demonstrates the potential of Zr(IV)-substituted POMs as artificial phosphatase and contributes to the further development of POMs as Lewis acid catalysts for the hydrolysis of biomolecules.38–40 The results from this study encourage us to further exploit the hydrolytic activity of this POM towards other more complex substrates such as adenosine triphosphate (ATP), DNA/RNA fragments, pesticides such as paraoxon and parathion, and nerve agents such as soman and sarin.
1 species, which is proposed as the active species, is the major and stable species under the reaction conditions. A 530-fold rate increase in comparison to the blank reaction was observed for HPNP hydrolysis. The present study demonstrates the potential of Zr(IV)-substituted POMs as artificial phosphatase and contributes to the further development of POMs as Lewis acid catalysts for the hydrolysis of biomolecules.38–40 The results from this study encourage us to further exploit the hydrolytic activity of this POM towards other more complex substrates such as adenosine triphosphate (ATP), DNA/RNA fragments, pesticides such as paraoxon and parathion, and nerve agents such as soman and sarin.
Experimental
Materials
The Zr-substituted Keggin POMs (Et2NH2)8[{α-PW11O39Zr(μ-OH)(H2O)}2]·7H2O (δ = −13.49 ppm)20,34 (ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and (Et2NH2)10[Zr(PW11O39)2]·7H2O (δ = −14.67 and −14.77 ppm)21 (ZrK 1
2), and (Et2NH2)10[Zr(PW11O39)2]·7H2O (δ = −14.67 and −14.77 ppm)21 (ZrK 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as well as 2-hydroxypropyl-4-nitrophenyl phosphate (δ = −4.64 ppm)35,36 (HPNP) were synthesized and characterized according to literature. Disodium diphenyl phosphate (DPP, C6H5PO4Na2·2H2O), DCl, and NaOD were purchased from Acros and used without any further purification.
2) as well as 2-hydroxypropyl-4-nitrophenyl phosphate (δ = −4.64 ppm)35,36 (HPNP) were synthesized and characterized according to literature. Disodium diphenyl phosphate (DPP, C6H5PO4Na2·2H2O), DCl, and NaOD were purchased from Acros and used without any further purification.
Kinetic measurements
Solutions containing 1.0 mM of HPNP and 1.0 mM of ZrK 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 were prepared in D2O. The final pD was adjusted with minor amounts of 10% DCl and 15% NaOD solutions in D2O. The pH-meter value was corrected by using the equation: pD = pH meter reading + 0.41.37 The pD of the samples was measured at the beginning and at the end of hydrolysis, and the difference was typically less than 0.5 units. The reaction mixture was kept at constant temperature and 1H NMR spectra were measured at certain time intervals during the hydrolytic reaction to calculate the observed rate constant (kobs) by the integral method.
2 were prepared in D2O. The final pD was adjusted with minor amounts of 10% DCl and 15% NaOD solutions in D2O. The pH-meter value was corrected by using the equation: pD = pH meter reading + 0.41.37 The pD of the samples was measured at the beginning and at the end of hydrolysis, and the difference was typically less than 0.5 units. The reaction mixture was kept at constant temperature and 1H NMR spectra were measured at certain time intervals during the hydrolytic reaction to calculate the observed rate constant (kobs) by the integral method.
NMR spectroscopy
1H NMR spectra were recorded on a Bruker Avance 400 and 0.5 mM of 3-(trimethylsilyl)propionic-2,2,3,3-d4 acid (TMSP) was used as an internal standard. 31P NMR spectra were recorded on a Bruker Avance 400 and Bruker Avance II+ 600 NMR spectrometer using power-gated 1H decoupling with 30 degree flip angle (zgpg30 pulse programme) and 25% phosphoric acid was used as a 31P external reference.Acknowledgements
T.N.P-V. and P.S. (BOF + fellowship) thank KU Leuven for the financial support and F. W. O. Flanders for a bilateral project. T.K.N.L. thanks the Vietnamese Government and KU Leuven for a doctoral fellowship. G.A. thanks F. W. O. Flanders for a post-doctoral fellowship. The authors acknowledge the CMST COST Action CM1203 (Polyoxometalate Chemistry for Molecular Nanoscience) for the financial support in terms of STSM applications.Notes and references
- M. Carraro and S. Gross, Materials, 2014, 7, 3956–3989 CrossRef CAS PubMed.
- A. Proust, B. Matt, R. Villanneau, G. Guillemot, P. Gouzerh and G. Izzet, Chem. Soc. Rev., 2012, 41, 7605–7622 RSC.
- H. Stephan, M. Kubeil, F. Emmerling and C. E. Müller, Eur. J. Inorg. Chem., 2013, 2013, 1585–1594 CrossRef CAS PubMed.
- N. V. Izarova, M. T. Pope and U. Kortz, Angew. Chem., Int. Ed., 2012, 51, 9492–9510 CrossRef CAS PubMed.
- A. Sartorel, M. Bonchio, S. Campagna and F. Scandola, Chem. Soc. Rev., 2013, 42, 2262–2280 RSC.
- T. K. N. Luong, P. Shestakova, T. T. Mihaylov, G. Absillis, K. Pierloot and T. N. Parac-Vogt, Chem. – Eur. J., 2015, 21, 4428–4439 CrossRef CAS PubMed.
- P. Nunes, A. C. Gomes, M. Pillinger, I. S. Gonçalves and M. Abrantes, Microporous Mesoporous Mater., 2015, 208, 21–29 CrossRef CAS PubMed.
- S. Vanhaecht, G. Absillis and T. N. Parac-Vogt, Dalton Trans., 2012, 41, 10028–10034 RSC.
- N. Steens, A. M. Ramadan, G. Absillis and T. N. Parac-Vogt, Dalton Trans., 2010, 39, 585–592 RSC.
- G. Absillis, R. van Deun and T. N. Parac-Vogt, Inorg. Chem., 2011, 50, 11552–11560 CrossRef CAS PubMed.
- E. Cartuyvels, G. Absillis and T. N. Parac-Vogt, Chem. Commun., 2008, 85–87 RSC.
- T. K. N. Luong, G. Absillis, P. Shestakova and T. N. Parac-Vogt, Eur. J. Inorg. Chem., 2014, 2014, 5276–5284 CrossRef CAS PubMed.
- D. M. Perreault and E. V. Anslyn, Angew. Chem., Int. Ed. Engl., 1997, 36, 432–450 CrossRef PubMed.
- M. Oivanen, S. Kuusela and H. Lönnberg, Chem. Rev., 1998, 98, 961–990 CrossRef CAS PubMed.
- E. L. Hegg and J. N. Burstyn, Coord. Chem. Rev., 1998, 173, 133–165 CrossRef CAS.
- D. T. Thomas, in Metal-DNA Chemistry, American Chemical Society, 1989, ch. 1, vol. 402, pp. 1–23 Search PubMed.
- D. E. Wilcox, Chem. Rev., 1996, 96, 2435–2458 CrossRef CAS PubMed.
- N. Sträter, W. N. Lipscomb, T. Klabunde and B. Krebs, Angew. Chem., Int. Ed. Engl., 1996, 35, 2024–2055 CrossRef PubMed.
- H. Arora, S. K. Barman, F. Lloret and R. Mukherjee, Inorg. Chem., 2012, 51, 5539–5553 CrossRef CAS PubMed.
- K. Nomiya, Y. Saku, S. Yamada, W. Takahashi, H. Sekiya, A. Shinohara, M. Ishimaru and Y. Sakai, Dalton Trans., 2009, 5504–5511 RSC.
- C. N. Kato, A. Shinohara, K. Hayashi and K. Nomiya, Inorg. Chem., 2006, 45, 8108–8119 CrossRef CAS PubMed.
- L. Cai, Y. Li, C. Yu, H. Ji, Y. Liu and S. Liu, Inorg. Chim. Acta, 2009, 362, 2895–2899 CrossRef CAS PubMed.
- S. Himeno, M. Takamoto and T. Ueda, Bull. Chem. Soc. Jpn., 2005, 78, 1463–1468 CrossRef CAS.
- P. H. Ho, E. Breynaert, C. E. A. Kirschhock and T. N. Parac-Vogt, Dalton Trans., 2011, 40, 295–300 RSC.
- P. H. Ho, T. Mihaylov, K. Pierloot and T. N. Parac-Vogt, Inorg. Chem., 2012, 51, 8848–8859 CrossRef CAS PubMed.
- C. Zhang, R. C. Howell, Q.-H. Luo, H. L. Fieselmann, L. J. Todaro and L. C. Francesconi, Inorg. Chem., 2005, 44, 3569–3578 CrossRef CAS PubMed.
- C. Zhang, L. Bensaid, D. McGregor, X. F. Fang, R. C. Howell, B. Burton-Pye, Q. H. Luo, L. Todaro and L. C. Francesconi, J. Cluster Sci., 2006, 17, 389–425 CrossRef CAS.
- L. Bonfá, M. Gatos, F. Mancin, P. Tecilla and U. Tonellato, Inorg. Chem., 2003, 42, 3943–3949 CrossRef PubMed.
- A. Singhal, L. M. Toth, J. S. Lin and K. Affholter, J. Am. Chem. Soc., 1996, 118, 11529–11534 CrossRef CAS.
- R. A. Moss, J. Zhang and K. G. Ragunathan, Tetrahedron Lett., 1998, 39, 1529–1532 CrossRef CAS.
- J. H. Espenson, Chemical kinetics and reaction mechanisms, Mc Graw-Hill, New York, 1994 Search PubMed.
- N. Virtanen, L. Polari, M. Välilä and S. Mikkola, J. Phys. Org. Chem., 2005, 18, 385–397 CrossRef CAS PubMed.
- C. Maxwell, A. A. Neverov and R. S. Brown, Org. Biomol. Chem., 2005, 3, 4329–4336 CAS.
- H. G. T. Ly, G. Absillis and T. N. Parac-Vogt, Dalton Trans., 2013, 42, 10929–10938 RSC.
- D. M. Brown and D. A. Usher, J. Chem. Soc., 1965, 6558–6564 RSC.
- J. S. W. Tsang, A. A. Neverov and R. S. Brown, J. Am. Chem. Soc., 2003, 125, 1559–1566 CrossRef CAS PubMed.
- P. K. Glasoe and F. A. Long, J. Phys. Chem., 1960, 64, 188–190 CrossRef CAS.
- K. Stroobants, G. Absillis, E. Moelants, P. Proost and T. N. Parac-Vogt, Chem. – Eur. J., 2014, 20, 3894–3897 CrossRef CAS PubMed.
- K. Stroobants, V. Goovaerts, G. Absillis, G. Bruylants, E. Moelants, P. Proost and T. N. Parac-Vogt, Chem. – Eur. J., 2014, 20, 9567–9577 CrossRef CAS PubMed.
- H. G. T. Ly, G. Absillis, R. Janssens, P. Proost and T. N. Parac-Vogt, Angew. Chem., 2015, 127, 7499 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Data on the percentage of HPNP as a function of the reaction time, 31P NMR spectrum, kinetic data on the influence of pD, temperature. See DOI: 10.1039/c5dt02077h |
| This journal is © The Royal Society of Chemistry 2015 |