Blue phosphorescent N-heterocyclic carbene chelated Pt(ii) complexes with an α-duryl-β-diketonato ancillary ligand†
Abstract
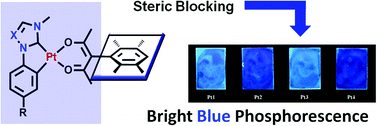
C^C* chelated Pt(II) compounds that contain an N-heterocyclic carbene (NHC) donor have recently attracted great interest as blue or white phosphorescent emitters for organic light-emitting devices (OLEDs). To overcome the tendency for excimer formation in Pt(II) compounds, an α-duryl-β-diketonato ligand was selected as the ancillary ligand for blue phosphorescent C^C* chelated Pt(II) compounds. Using this approach, a series of NHC-based C^C* chelated Pt(II) compounds has been designed and synthesized. The chelate ligands used in the new C^C* chelated Pt(II) compounds include 1-phenylimidazol-2-ylidene in Pt1, 1-phenyl-1,2,4-triazol-5-ylidene in Pt2, p-TMS-1-phenylimidazol-2-ylidene in Pt3, and p-TMS-1-phenyl-1,2,4-triazol-5-ylidene in Pt4. A single-crystal X-ray diffraction analysis revealed that the presence of the α-duryl-β-diketonato ligand in the Pt(II) compounds effectively suppresses the dimer formation in the crystal lattice. Pt1, Pt2, Pt3, and Pt4 display blue phosphorescence at room temperature. The p-TMS substituted complex Pt3 was found to display the most efficient blue phosphorescence at λem = 468 nm with a ΦPL of 0.68. Spectroscopic and computational studies established that the blue phosphorescence in the phenyl-imidazolylidene chelated complexes originates mainly from a 3MLCT state while that in the phenyl-triazolylidene chelated compounds arises from a 3ILCT state. Electroluminescent devices using Pt1 and Pt3 as the dopant were fabricated which display both monomer and exciplex emission, leading to pure white light electroluminescence with CIE coordinates of (0.32, 0.31).
- This article is part of the themed collection: Luminescent Complexes and Materials for Light-Emitting Devices

 Please wait while we load your content...
Please wait while we load your content...