Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Metal complexes of curcumin – synthetic strategies, structures and medicinal applications†
Simon
Wanninger
a,
Volker
Lorenz
a,
Abdus
Subhan
b and
Frank T.
Edelmann
*a
aChemisches Institut der Otto-von-Guericke-Universität Magdeburg, Universitätsplatz 2, 39106 Magdeburg, Germany. E-mail: frank.edelmann@ovgu.de; Fax: +49 391 6712933; Tel: +49 391 6758327
bDepartment of Chemistry, Shah Jalal University of Science and Technology, Sylhet, Bangladesh. E-mail: subhan-che@sust.edu
First published on 12th May 2015
Abstract
This Tutorial Review presents an overview on the synthesis, characterization and applications of metal complexes containing curcumin (=1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) and its derivatives as ligands. Innovative synthetic strategies leading to soluble and crystallizable metal curcumin complexes are outlined in detail. Special emphasis is placed on the highly promising and exciting medicinal applications of metal curcumin complexes, with the three most important areas being anticancer activity and selective cytotoxicity, anti-Alzheimer's disease activity, and antioxidative/neuroprotective effects. Overall, this Tutorial Review provides the first general overview of this emerging and rapidly expanding field of interdisciplinary research.
Key learning points1. Curcumin and its derivatives.2. Synthesis and characterization of metal curcumin complexes. 3. Non-medicinal applications of metal curcumin complexes. 4. Medicinal applications of metal curcumin complexes. 5. Anti-cancer activity of metal curcumin complexes. |
1. Introduction
“From kitchen to clinic” or “Curry against Alzheimer” – these are only two of the recent sensational headlines associated with the various health benefits of curcumin. Curcumin1 (=1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) is a component of the Indian spice turmeric, manufactured from the rhizome of the perennial herb Curcuma longa that is widely cultivated in tropical countries in South and South East Asia, especially in China and India. Curcuma longa belongs to the Zingiberaceae (ginger) family. Curcumin (=CurcH) is the major component of three curcuminoids that give turmeric its characteristic yellow color and is used as a food colorant, flavoring and additive (E number E100). The minor curcuminoid components are demethoxycurcumin (=DMCurcH) and bis-demethoxycurcumin (=BDMCurcH) in which one or both –OMe functionalities at the outer phenol rings are removed. Very recently, an improved separation of the three curcuminoids was realized using a combination of normal-phase column and phosphate-impregnated preparative-thin layer chromatographies.2 Besides its widespread use as food flavor and colorant, turmeric has been used in traditional Chinese and Ayurvedic medicine for around 4000 years. Thus the headline “Curry against Alzheimer”, although sounding modern and sensational, has its roots in this traditional knowledge. It has been demonstrated that the incidents of Alzheimer's disease among elderly people of age 70–79 in rural India, who eat curry dishes on a daily basis, is about 4.4 times lower than that of Americans of the same age.3Over the past few decades, numerous studies have explored the medicinal properties of turmeric and curcumin, including antitumoral, antimicrobial, anti-inflammatory, antioxidant, antihepatotoxic, antihyperlipidemic, antiviral, and anti-Alzheimer's disease effects. In fact, turmeric has even been termed the “multi-anti spice” in herbal medicine, and curcumin has been referred to as “curecumin”.4,5 In more detail, the following diseases and disorders may be treated successfully with curcumin: liver and biliary diseases, wounds and ulcers caused by injuries and diabetes, psoriasis, arthritis and rheumatism, sinusitis, heart diseases and high blood cholesterol, diabetes, amyloidosis as well as cervical, colon and pancreatic cancer. The anticancer properties include suppression of cellular transformation, prevention of cancer cell proliferation, and suppression of carcinogenic effects.6 Curcumin compounds alone or in combination with other anticancer drugs have been reported to inhibit the clonogenicity of cancer cells and induce anti-proliferative and apoptotic effects on drug-resistant and sphere-forming cancer cells expressing stem cell-like markers as well as reverse the chemoresistance. Thereby they improve the cytotoxic effects induced by diverse chemotherapeutic drugs on these immature cancer cells. These beneficial health effects of curcumin are all well documented in the current literature. For example, in 2013 the journal “Current Pharmaceutical Design” published a special issue entitled “Recent Progress and Novel Insights in Curcumin Research – From Chemistry to Clinical Use”. Clinical studies in humans showed that curcumin is generally safe even at high daily doses of up to 12 grams with only few side-effects.5 A severe problem encountered in clinical trials involving curcumin is its poor bioavailability, leading to low levels in plasma and tissues. The bioavailability of curcumin in the form of food colorants or additives (spices) may be higher due to the cooking process or the dissolution in oils. The insolubility of curcumin in water, poor absorption, rapid metabolism and systemic elimination have been shown to be the main factors limiting its bioavailability. As a result, numerous studies have been directed to increasing curcumin bioavailability, including the use of absorption factors (e.g. piperidine/piperine), the encapsulation of curcumin in the cavities of cyclodextrins, or the use of nanoparticles6 and ceramic particles.7
A highly promising and innovative approach to deal with the bioavailability issue and to achieve even more diverse potential health benefits is the use of metal curcumin complexes. Curcumin and the curcuminoids are rare examples of naturally occurring β-diketone ligands. As such, they should be ideally suited to act as chelating ligands toward a variety of metals and to form stable complexes. The past 10 years have witnessed a dramatic increase in studies directed to the synthesis, characterization and biological investigation of metal curcumin complexes. Already it has become clear that the potential medicinal applications are as diverse as those of curcumin itself. The three most exciting areas appear to be selective cytotoxicity and anticancer activity, anti-Alzheimer's disease activity, and antioxidative/neuroprotective effects. However, highly promising results with metal curcumin complexes have also been reported in the fields of antiarthritic/antirheumatic activity, antimicrobial/antifungal activity, anti-viral/anti-HIV activity and biological imaging/radioimaging. As becomes evident from the list of references, many of these investigations have been published in journals which e.g. synthetic chemists normally do not have on their screen. Notably, more than 50% of the references cited in this review appeared between 2012 and 2015. Thus it is a major goal of this Tutorial Review to serve as a springboard for the readers to readily access the highly diverted literature in the area of metal curcumin complexes. Overall, this Tutorial Review tries to provide the first general overview of this rapidly expanding field and to outline the most exciting current trends.
2. The characteristics of curcumin-based ligands
The chemical structure of curcumin was first identified in 1910 by Miłobędzka, von Kostanecki and Lampe (Fig. 1). Through chemical derivatization, these authors clearly established the identity of curcumin as diferuloylmethane or 1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione.8 They also prepared two potentially useful derivatives, dicarbomethoxycurcumin and dicarboethoxycurcumin, by treatment of curcumin with two equivalents of methyl- or ethylchloroformate, respectively, in the presence of aqueous potassium hydroxide. These two derivatives are particularly valuable in curcumin chemistry as they are easily accessible in good yields and high purity.8 | ||
| Fig. 1 First paragraphs of the historic paper by Miłobędzka, von Kostanecki and Lampe. Reproduced from ref. 8. | ||
At the first glance, curcumin is just another typical substituted β-diketone ligand closely related to acetylacetone. Like acetylacetone, curcumin displays typical keto–enol-tautomerism as illustrated in Scheme 1.
However, unlike acetylacetone, the central β-diketone functionality is flanked by the sterically demanding unsaturated phenolic groups, –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH–C6H4(OH)(OMe)-3,4, which results in an unusually wide and flat β-diketonate ligand. Thus the overall shape of curcumin ligands with the two large wings attached to the β-diketone unit may be compared to that of an eagle. In addition, the phenolic –OH groups are additional centers of potential reactivity which could lead to complications in metal complex preparation (i.e. formation of insoluble polymers).
CH–C6H4(OH)(OMe)-3,4, which results in an unusually wide and flat β-diketonate ligand. Thus the overall shape of curcumin ligands with the two large wings attached to the β-diketone unit may be compared to that of an eagle. In addition, the phenolic –OH groups are additional centers of potential reactivity which could lead to complications in metal complex preparation (i.e. formation of insoluble polymers).
Curcumin exists in the keto form in acidic and neutral pH media and in the enol form in alkaline pH medium. A stable crystalline form of curcumin is long known in the literature. Recently, however, two new metastable polymorphs (enol form) of curcumin were reported that exhibit higher solubility as compared to the stable form. In the course of a recent study, the solubility and mechanical properties in these polymorphic systems have been investigated. It was found that the hardness (H) is inversely proportional to the solubility of a polymorph. Effectively, this study demonstrated that the hardness is a useful parameter (comparable to melting point and density) that correlates well with the solubility of a polymorph. To summarize, a softer polymorph is more soluble (Fig. 2). It was pointed out that such a correlation is helpful in systems like curcumin in which the Gibbs free energies of the polymorphs are close to one another.9
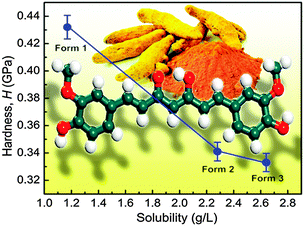 | ||
| Fig. 2 Correlation between hardness and solubility of the three known curcumin polymorphs. Reproduced from ref. 9. Copyright 2014 American Chemical Society. | ||
Possible variations of the basic curcumin ligand system are manifold. Various changes of the substituents at the phenyl rings are possible, e.g. by converting the phenolic –OH group into –OR (R = Me: Me2CurcH, R = Et: Et2CurcH, R = nBu: Bu2CurcH), –OC(O)Me (diacetylcurcumin = DACurcH), or –OC(O)OR (R = Me, Et) functionalities. Even β-D-glucopyranos-1-yloxy moieties have been successfully attached to the curcumin backbone.10 Methylation at the central carbon atom of the β-diketone moiety is also possible to give e.g. trimethylcurcumin (Me3CurcH). Table 1 lists some of the most common curcumin ligands and their abbreviations used in this Tutorial Review.
Most recently, in the course of our own studies of new metal curcumin complexes, we succeeded in the synthesis and structural characterization of the first triorganosilyl-substituted curcumin derivative, 1,7-bis(4-triphenylsiloxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione (=Ph3SiCurcH). This compound is formed in high yield upon treatment of an alkaline solution of curcumin with 2 equiv. of chlorotriphenylsilane. The bright yellow compound shows a reasonable solubility in a number of common organic solvents. X-Ray quality single-crystals of Ph3SiCurcH could be grown from hot toluene. Fig. 3 clearly shows that the two bulky triphenylsilyl substituents add a great deal of steric demand to the curcumin ligand system. Metal complex formation of this new curcumin ligand still awaits further exploration.
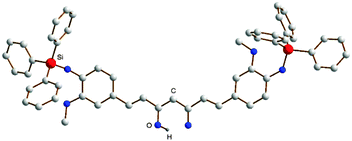 | ||
| Fig. 3 Molecular structure of 1,7-bis(4-triphenylsiloxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione (=Ph3SiCurcH). | ||
Furthermore, new curcumin-derived O,N-chelating ligands can be obtained by replacing one of the β-diketone oxygen atoms by ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–OH (curcuminoxime),
N–OH (curcuminoxime), ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–NHC(O)NH2 (curcuminsemi-carbazone), and
N–NHC(O)NH2 (curcuminsemi-carbazone), and ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–NHC(S)NH2 (curcuminthiosemicarbazone). Curcumin can also be modified in various other ways leading to potentially new ligand systems. For example, Knoevenagel condensation of the active central methylene group of curcumin with an aldehyde (e.g. salicylaldehyde) provides a non-enolizable β-diketone which can be further treated with suitable amines, e.g. thiosemicarbazide or substituted anilines, to afford the corresponding Schiff base derivatives. The resulting curcumin diketimine ligands have been successfully employed in the synthesis of a series of copper(II) complexes.11 An even more sophisticated extension of these ligand modifications is the design of a macrocyclic tetraaza diacetyl curcumin ligand. This work nicely demonstrates how far-reaching modifications of the curcumin ligand system are possible. The reaction sequence is illustrated in Scheme 2.12
N–NHC(S)NH2 (curcuminthiosemicarbazone). Curcumin can also be modified in various other ways leading to potentially new ligand systems. For example, Knoevenagel condensation of the active central methylene group of curcumin with an aldehyde (e.g. salicylaldehyde) provides a non-enolizable β-diketone which can be further treated with suitable amines, e.g. thiosemicarbazide or substituted anilines, to afford the corresponding Schiff base derivatives. The resulting curcumin diketimine ligands have been successfully employed in the synthesis of a series of copper(II) complexes.11 An even more sophisticated extension of these ligand modifications is the design of a macrocyclic tetraaza diacetyl curcumin ligand. This work nicely demonstrates how far-reaching modifications of the curcumin ligand system are possible. The reaction sequence is illustrated in Scheme 2.12
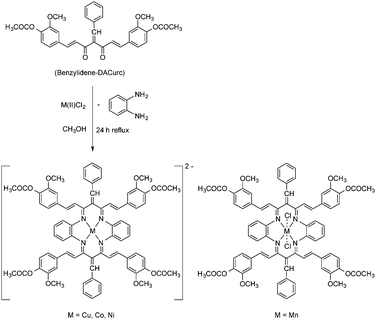 | ||
| Scheme 2 Synthetic route to metal(II) complexes containing a tetraazamacrocyclic diacetylcurcumin ligand. | ||
3. How to prepare and characterize metal curcumin complexes
Most studies dealing with metal curcumin complexes and their applications have been published within the past 10 years. Coordination compounds comprising curcumin and its derivatives as ligands have been reported for most metallic elements in the periodic table. Notably, well characterized alkali and alkaline earth metal curcumin complexes are scarce. Only very few examples have been described for Na, Be, Mg, Ca, and Ba. The paucity of well-defined alkali metal curcumin derivatives is particularly surprising in view of the fact that such compounds are often formed and used as intermediates upon deprotonation of free curcumins by alkali metal hydroxides, alkoxides, acetates etc. Among the other main group elements, boron derivatives form the longest known and most thoroughly investigated class of compounds.13,14 Several aluminum15–17 and gallium18–20 compounds with curcumin ligands have also been described, while there are only a few scattered examples for curcumin complexes of indium, tin, and lead.20 In the case of Groups 3–6, examples are known for Y, La, Zr, V, and Cr, with a significant prevalence of vanadyl species containing the V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O unit.20–23 However, there is a clear accumulation of compounds containing middle and late transition metals.24 Curcumin complexes have been described for all metals in the Groups 7–12 except for osmium. By far the most papers in this area deal with copper curcumin complexes,25–27 followed by zinc,28–30 nickel,31 iron,10,31 manganese,32,33 and ruthenium34,35 species. For the remaining middle and late transition metals, the number of well-characterized curcumin complexes is significantly smaller.36–38 Recently, even technetium complexes containing curcumin have been reported.39 Moreover, curcumin complexes have been prepared for several lanthanide elements2,40–43 as well as thorium and uranium, with the latter all being uranyl (O
O unit.20–23 However, there is a clear accumulation of compounds containing middle and late transition metals.24 Curcumin complexes have been described for all metals in the Groups 7–12 except for osmium. By far the most papers in this area deal with copper curcumin complexes,25–27 followed by zinc,28–30 nickel,31 iron,10,31 manganese,32,33 and ruthenium34,35 species. For the remaining middle and late transition metals, the number of well-characterized curcumin complexes is significantly smaller.36–38 Recently, even technetium complexes containing curcumin have been reported.39 Moreover, curcumin complexes have been prepared for several lanthanide elements2,40–43 as well as thorium and uranium, with the latter all being uranyl (O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) U
U![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) species.
O) species.
Pioneering work in this area was published in 1997 by Beck and co-workers at the Ludwig-Maximilians-Universität in Munich.31 As part of a series of publications entitled “Metal Complexes of Dyes”, no less than twenty-eight new transition metal complexes containing the ligands curcumin (Curc), bis-demethoxycurcumin (BDMCurc), dimethylcurcumin (Me2Curc), trimethylcurcumin (Me3Curc; this ligand contains a methyl group at the central methylene functionality), and diacetylcurcumin (DACurc) were synthesized and characterized. The general synthetic protocol employed in this early study is applicable to the preparation of virtually all metal complexes containing curcumin and its derivatives as ligands. The first step normally involves deprotonation of the free curcumin derivative by a suitable base such as ammonia, sodium hydroxide, sodium methoxide, sodium carbonate, sodium acetate or silver acetate. In the second step, the resulting anions are treated with metal halides in the appropriate molar ratios. This way, Beck et al. obtained the homoleptic complexes Pd(Curc)2 and M(Me2Curc)2 (M = Ni, Cu) from the metal(II) acetates, while homoleptic Fe(Curc)3 was made from FeCl3 and 3 equiv. of in situ-prepared NaCurc. An even larger series of new curcumin complexes was prepared starting from organotransition metal halides, including [(R3P)MCl2]2 (M = Pd, Pt; R = Et, nBu, Ph, tolyl), [(η5-C5Me5)MCl2]2 (M = Rh, Ir), [(η6-p-cymene)RuCl2]2 (p-cymene = p-isopropyltoluene) and [(η3-C3H5)PdCl]2. All these reactions yielded well-defined complexes containing one curcumin-type ligand per metal atom plus one or two different co-ligands (“spectator ligands”) such as allyl, phosphines, pentamethylcyclopentadienyl, or p-cymene. Characterization of the new complexes was based mainly on spectroscopic methods (1H and 13C NMR, IR, UV/Vis) and elemental analyses. Only in the case of (η6-p-cymene)RuCl(Me2Curc), X-ray quality single-crystals could be obtained.31 The closely related curcumin complex (η6-p-cymene)RuCl(Curc) was later prepared in a similar manner by reaction of [(η6-p-cymene)RuCl2]2 with curcumin in the presence of sodium methoxide, NaOMe. The compound forms an air- and moisture-stable solid which is soluble in chlorinated solvents, alcohols, acetone, acetonitrile, and DMSO, clearly a positive effect of the spectator ligand p-cymene. In both cases the X-ray analyses confirmed the formation of the expected half-sandwich complex containing an η6-arene ligand, one chloride and a chelating curcumin anion. Fig. 4 depicts the molecular structure of (η6-p-cymene)RuCl(Curc).35
 | ||
| Fig. 4 Molecular structure of (η6-p-cymene)RuCl(Curc). Reproduced from ref. 35. Copyright 2012 American Chemical Society. | ||
The reason why the early study by Beck et al. has been discussed here in more detail is the following: this work already revealed some of the important general characteristics and potential problems of transition metal curcumin chemistry. Most curcumin complexes are easily accessible using the general synthetic route with or without minor modifications. Yields are often very high, and in most cases the products are readily isolated in the form of precipitates. However, crystallinity of the products is often low, as is solubility in water and common organic solvents. Many curcumin complexes exhibit significant solubility only in highly polar solvents such as pyridine, dimethylformamide (DMF), or dimethylsulfoxide (DMSO). As a result, structural characterization of metal curcumin complexes through X-ray diffraction studies remains scarce. A SciFinder survey on the topic “metal complexes of curcumin” yielded ca. 150 publications in which only 11 crystal structures of such complexes were reported. The lack of structural information is particularly apparent for the homoleptic curcumin complexes. Homoleptic curcumin complexes of the type M(Curc)2 and M(Curc)3 are known for various middle and late transition metals as well as some of the rare-earth elements. Quite frequently, homoleptic transition metal or lanthanide curcumin complexes are described as orange, red, or brown precipitates which lack solubility in most organic solvents and are difficult to characterize.24 Accordingly, to our knowledge, none of these homoleptic curcumin complexes has been structurally authenticated by X-ray diffraction!
Now, what are the main lessons to be learned from the pioneering work by Beck and co-workers? Apparently, the crystallization of homoleptic transition metal complexes comprising only curcumin as ligands is severely hampered, presumably through the formation of polymeric arrays by interaction of the metal ions with the free phenolic –OH groups. This would be a possible explanation for the observed low solubility of such complexes in water and most of the common organic solvents. What can be done to overcome these obstacles without completely altering the curcumin ligand system? Basically, two different successful strategies can be envisaged which in many cases have already led to the formation of metal curcumin complexes which exhibit higher solubility and better crystallinity:
1. A logical approach would be the blocking of the free phenolic –OH groups at the 4-positions of the phenyl rings. As mentioned above, this can be achieved by converting the phenolic –OH groups into –OR, –OC(O)R, or –OC(O)OR (R = Me, Et) functionalities. Very recently, positive proof of this approach was provided by the successful synthesis and structural characterization of the copper(II) complexes Cu(Et2Curc)2(1,4-dioxane) (Et2CurcH = 1,7-bis(4-ethoxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) and homoleptic Cu(Bu2Curc)2 (Bu2Curc = 1,7-bis(4-n-butoxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione). These compounds were prepared in the usual manner from copper(II) acetate and 2 equiv. of the alkylated curcumins. Unlike previously reported copper(II) curcumin complexes, X-ray quality single-crystals of Cu(Et2Curc)2(dioxane) could be grown from ethyl acetate–1,4-dioxane solutions. As shown in Fig. 5, the central copper(II) ions are coordinated by two chelating diethylcurcumin ligands in a square-planar coordination geometry. As a result of the large π-conjugated electron system, the ligands are almost planar. The packing diagram of the complex showed that the adjacent molecules are stacked through two types of π–π interactions along the a and c axes with short distances of 3.846 and 3.897 Å. Single crystals of the n-butyl derivative Cu(Bu2Curc)2 could even be grown directly from ethyl acetate. The results clearly demonstrate that the solubility of homoleptic metal curcumin complexes can be significantly improved by blocking the phenolic –OH groups of the parent curcumin ligand.25,26
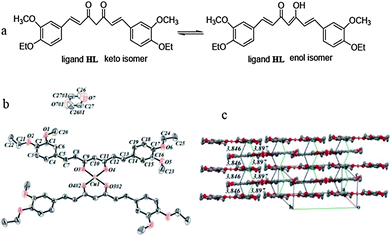 | ||
| Fig. 5 (a) Keto–enol tautomers of the ligand diethylcurcumin (1,7-bis(4-ethoxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione); (b) molecular and (c) crystal structure of the copper(II) curcumin complex Cu(Et2Curc)2(dioxane). Reproduced from ref. 25 and 26. | ||
2. The second potentially successful approach is the synthesis of heteroleptic complexes containing suitable spectator ligands in addition to curcumin. Chances to isolate soluble and crystallizable curcumin complexes apparently increase if only one curcumin ligand is present in such heteroleptic metal complexes. Suitable ligands to complement the coordination sphere of the metal ions in addition to curcumin include, but are not limited to, pyridine, 2,2′-bipyridine, phenanthroline, terpyridine derivatives, tertiary phosphines, cyclopentadienyl ligands and η6-coordinated arenes like cymene or hexamethylbenzene. Typical examples for this spectator ligand approach will be discussed in the following paragraphs.
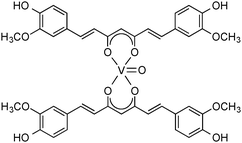
In recent years, the original synthetic protocol (deprotonation of curcumins, followed by treatment with suitable metal halide precursors) has been extended to the synthesis of a large number of other main-group and transition metal complexes containing curcumin and substituted curcumins as ligands. Only selected examples will be discussed here in more detail. A fairly large and well-investigated group of early transition metal curcumin complexes contains the vanadyl group (V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O). The parent complex, VO(Curc)2 (“vanadyl curcumin”), is easily accessible in high yields (89%) on a 20 g-scale by treatment of vanadyl acetylacetonate, VO(acac)2, with 2 equiv. of curcumin in dichloromethane solution in the presence of 2 M NaOH. VO(Curc)2 is a rust-colored solid, which was characterized by IR, MS, magnetic moment and elemental analysis (Scheme 3). This remarkable compound was found to exhibit both promising antiarthritic and anticancer activity (cf. Section 5).21
O). The parent complex, VO(Curc)2 (“vanadyl curcumin”), is easily accessible in high yields (89%) on a 20 g-scale by treatment of vanadyl acetylacetonate, VO(acac)2, with 2 equiv. of curcumin in dichloromethane solution in the presence of 2 M NaOH. VO(Curc)2 is a rust-colored solid, which was characterized by IR, MS, magnetic moment and elemental analysis (Scheme 3). This remarkable compound was found to exhibit both promising antiarthritic and anticancer activity (cf. Section 5).21
No other metal curcumin complex motif has been chemically modified to such an extent than vanadyl curcumin.20–23 In particular, a considerable number of vanadyl mono(curcumin) complexes comprising suitable co-ligands (spectator ligands) have been synthesized, characterized and investigated for their biological activity. Mainly bi- and tridentate N-donor ligands related to 2,2′-bipyridine, phenanthroline, bis(2-pyridyl-methyl)amine and terpyridine and their derivatives have been employed as spectator ligands. Most of these vanadyl complexes were prepared in a straightforward manner following a general synthetic protocol in which vanadyl sulfate, VOSO4, is first treated with barium chloride to give a solution of vanadyl chloride, VOCl2. After removal of the by-product barium sulfate, the filtrate is treated with equimolar amounts of curcumin and the respective bi- or tridentate ligand to afford chlorovanadyl complexes like e.g. VO(Curc)(phenanthroline)Cl as shown in Scheme 4a. Closely related cationic vanadyl mono(curcumin) complexes can be prepared from reactions of an equimolar mixture of VO(acac)2, the respective bi- or tridentate ligand and sodium perchlorate. A typical example containing a ferrocenylmethyl-substituent at the spectator ligand backbone is shown in Scheme 4b.22,23
 | ||
| Scheme 4 (a, b) Typical examples of vanadyl mono(curcumin) complexes containing tridentate co-ligands. | ||
Some technetium and rhenium curcumin complexes have recently been reported.39 It was found that curcumin reacts with the bromide precursors fac-[M(CO)3Br3]2− (M = Tc, Re) through the β-diketonate moiety to generate, under mild synthetic conditions, the aqua complexes fac-M(CO)3(H2O)(Curc). The aqua ligand in both complexes is labile and can be easily replaced at room temperature by triphenylphosphine to generate the corresponding tricarbonyl monophosphine complexes fac-M(CO)3(PPh3)(Curc) (M = Tc, Re). Excess of phosphine leads under reflux in methanol to the formation of the new dicarbonyl bis(phosphine) complexes cis–trans-M(CO)2(PPh3)2(Curc). Apparently, in the tricarbonyl complexes the phosphine ligand, which has a large trans effect, labilizes the trans carbonyl which is quantitatively replaced by a second phosphine ligand. The rhenium bis(phosphine) complex was also prepared using the precursor mer-trans-Re(CO)3(PPh3)2Cl. In this case, the incoming bidentate donor ligand curcumin displaces the Cl and one of the CO ligands. All complexes were isolated in high yield and characterized by elemental analysis, spectroscopic methods, and, in the case of cis–trans-M(CO)2(PPh3)2(Curc), X-ray crystallography (Fig. 6). All complexes were found to be soluble in dichloromethane, chloroform, benzene, and toluene, slightly soluble in methanol and ethanol, and insoluble in water. The bis(phosphine) complexes were even stable in solution for months as shown by HPLC and NMR. In solutions of the monophosphine complexes, however, a gradual release of the coordinated phosphine ligand was noted over time, more intense in solvents with coordinating potential like dimethylsulfoxide (DMSO). After all, this is another nice example illustrating how heteroleptic metal curcumin complexes with good solubility and crystallinity can be designed through the right choice of spectator ligands.39
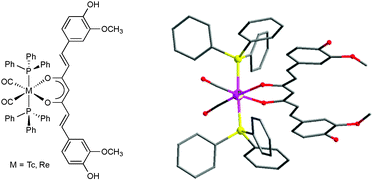 | ||
| Fig. 6 Schematic representation of the curcumin complexes cis–trans-M(CO)2(PPh3)2(Curc) (M = Tc, Re) and molecular structure of cis–trans-Re(CO)2(PPh3)2(Curc). Reproduced from ref. 39. Copyright 2014 American Chemical Society. | ||
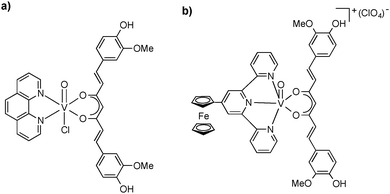
Among the late transition metals, ruthenium provides particularly instructive and successful examples for the spectator ligand approach to well-characterized and nicely crystalline mono(curcumin) complexes. Following the successful synthesis and structural characterization of (η6-p-cymene)RuCl(Me2Curc)35 (vide supra), the synthetic protocol was applied to a number of cationic ruthenium mono(curcumin) complexes containing not only the η6-bonded arene ligand (p-cymene or hexamethyl-benzene) but also the bulky phosphine ligand 1,3,5-triaza-7-phosphaadamantane (=PTA) (cf.Fig. 7a). In this case, the crystal and molecular structures of three new complexes, [(η6-p-cymene)Ru(Curc)(PTA)][SO3CF3], [(η6-hexamethylbenzene)Ru(Curc)(PTA)][SO3CF3], and [(η6-p-hexamethylbenzene)Ru(BDMCurc)(PTA)][SO3CF3] could be authenticated by X-ray diffraction! Fig. 7b depicts the molecular structure of the cation in [(η6-hexamethylbenzene)Ru(Curc)(PTA)][SO3CF3]. These results clearly emphasize the importance of careful spectator ligand design in the chemistry of metal curcumin complexes.34
 | ||
| Fig. 7 (a) Schematic representation of cationic (η6-arene)ruthenium mono(curcumin) complexes containing PTA as co-ligand; X− = PF6−, SO3CF3−; (b) Molecular structure of the cation in [(η6-hexamethyl-benzene)Ru(Curc)(PTA)][SO3CF3]. Reproduced from ref. 34. Copyright 2014 American Chemical Society. | ||
Low solubility and poor crystallinity also characterize the homoleptic lanthanide complexes of the type Ln(Curc)3 (Ln = Sc, Y, La–Lu).2,40–42 Here, too, the spectator ligand approach proved to be highly useful. Scheme 5 illustrates the successful combination of curcumin (+diglucosylcurcumin), anions (NO3−) and spectator ligands (4′-phenylterpyridine and 4′-(1-pyrenyl)terpyridine) which was assembled in the coordination sphere of the large lanthanide anions La3+ and Gd3+. Due to their large ionic radii, steric saturation of the coordination sphere of the lanthanide ions is even more important than in the case of transition metals. This is due to the fact that Ln3+ ions generally prefer high coordination numbers such as 8 or 9. As a result of this favorable ligand design, the crystal structures of both 4′-phenylterpyridine derivatives could be determined by X-ray diffraction.43
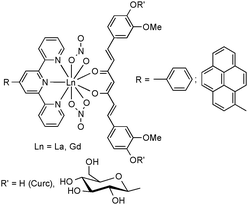 | ||
| Scheme 5 Schematic representation of lanthanide mono(curcumin) complexes containing 4′-phenylterpyridine and 4′-(1-pyrenyl)terpyridine as spectator ligands. | ||
4. Non-medicinal/analytical applications of metal curcumin complexes
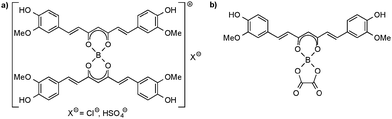
The oldest known application of curcumin complexes is related to the spectrophotometric detection of trace amounts of boron in various media,44 including biological materials45 (e.g. foodstuffs, plants and blood plasma), natural water and sea water, soil, iron and steel, as well as materials relevant to nuclear technology, such as uranium metal, uranium oxide and aluminum. As early as 1866, Schlumberger discovered an intensely red dye which he named “rosocyanine”. This dye was formed when curcumin reacted with boric acid, B(OH)3, in alcoholic solution in the presence of a strong mineral acid. At that time, the molecular structure of curcumin had not even been established. Today it is well established that rosocyanine is a cationic 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
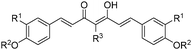
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex formed from curcumin and boric acid in which the central boron atom is tetrahedrally coordinated by two curcumin anions (Scheme 6). Anions can be chloride and hydrogensulfate.13 Similar complexes containing only curcumin ligands bonded to boron are formed e.g. with BF3·Et2O and boronic acids, RB(OH)2. Also notable in this context is a closely related compound named “rubrocurcumin”. This red dye is formed when the reaction of curcumin with boric acid or borates is carried out in the presence of oxalic acid. In contrast to the cationic rosocyanine, rubrocurcumin is a neutral compound in which boron is tetrahedrally coordinated by one curcumine and one oxalate ligand. Similar products can be obtained e.g. by replacing oxalic acid with citric acid.13,14
1 complex formed from curcumin and boric acid in which the central boron atom is tetrahedrally coordinated by two curcumin anions (Scheme 6). Anions can be chloride and hydrogensulfate.13 Similar complexes containing only curcumin ligands bonded to boron are formed e.g. with BF3·Et2O and boronic acids, RB(OH)2. Also notable in this context is a closely related compound named “rubrocurcumin”. This red dye is formed when the reaction of curcumin with boric acid or borates is carried out in the presence of oxalic acid. In contrast to the cationic rosocyanine, rubrocurcumin is a neutral compound in which boron is tetrahedrally coordinated by one curcumine and one oxalate ligand. Similar products can be obtained e.g. by replacing oxalic acid with citric acid.13,14
Solid rosocyanine forms green-black microcrystals with a metallic luster. Its intense red color allows the colorimetric detection of boron even at ppm levels. Recently, the method has been extended to develop a rapid and facile thin-layer chromatography staining method for the qualitative analysis of boronic acids and related boron-containing compounds. Here too, a characteristic red coloration is observed due to the formation of boron–curcumin complexes. In this case it was possible to confirm the deep red phenylboronic acid–curcumin complex by single-crystal X-ray diffraction. The X-ray study clearly showed that the curcumin acts as a chelating ligand to the tetrahedrally coordinated boron to produce a tetrahedral boronate to which one methanol molecule is added, consistent with the coordination geometry observed in rosocyanine (Fig. 8).46
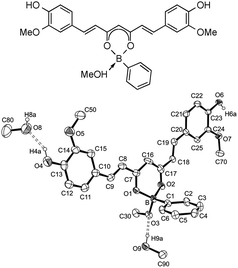 | ||
| Fig. 8 Schematic representation and molecular structure of the phenylboronic acid–curcumin complex. Reproduced from ref. 46. Copyright 2012 The Royal Society of Chemistry. | ||
Yet another potentially useful non-medicinal application of metal curcumin complexes, especially those of nickel, involves their use in the electrochemical modification of electrodes for oxidation reactions. The catalytic oxidation of methanol on a glassy carbon electrode which was electrochemically modified by a conductive Ni(II)–curcumin complex film was first reported in 1995.47 In the course of this study, it was found that a glassy carbon electrode modified by this Ni(II)–curcumin film can act as a very effective catalyst for the oxidation of alcohols. Deposition of the nickel(II) curcumin complex was simply performed by contacting the clean electrode surface with a freshly prepared solution of curcumin, a Ni(II) salt and 0.1 M NaOH. Electropolymerization of the nickel–curcumin complex and oxidation of methanol were studied by cyclic voltammetry. The modified electrode was shown to provide a durable catalytic surface which allows the voltammetric oxidation of methanol: in 0.1 M NaOH electrolyte the resulting anodic peak is at 0.59 V (vs. SCE). In contrast, electrooxidation was not possible at bare glassy carbon electrodes, at least before the onset of the electrolyte decomposition. Electrocatalytic oxidation of ethanol, propanol and butanol could be achieved quite efficiently in the same manner.47 The surface morphologies of the nano-structured Ni(II)–curcumin-modified carbon electrodes have been studied by scanning electron microscopy (SEM) and atomic force microscopy (AFM). The results (Fig. 9) showed that the deposited Ni(II)–curcumin films had a nano-globular structure in the range of 20–50 nm.48 More recently, Ni–curcumin-modified glassy carbon or carbon paste electrodes have been successfully employed in the electrocatalytic oxidation of various biologically and pharmaceutically relevant molecules and drugs. The substrates included glucose and fructose, several amino acids, the antibiotic amoxicillin, and some non-steroidal anti-inflammatory drugs such as indomethacin, mefenamic acid, and diclofenac. As a result, the modified electrodes could be used as sensors for the determination of glucose and fructose with good response at low detection limits. The method also provided a sensitive, simple, and time-saving amperometric procedure for the analysis of antibiotic and anti-inflammatory drugs in pharmaceutical preparations and biological media.49
 | ||
| Fig. 9 SEM images of a nano-structured Ni(II)–curcumin-modified carbon electrode at two different magnifications (50.000× and 60.000×). Reproduced from ref. 48. Copyright 2008 Elsevier B. V. | ||
There are also a few unusual applications of metal curcumin complexes in materials science. Notable is a recent report about the synthesis and characterization of ZnO nanoparticles via thermal decomposition of a zinc curcumin complex. Treatment of an ethanolic solution of zinc nitrate with curcumin in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio afforded the dinuclear zinc complex [Zn(Curc)(OH)]2 which could be employed as a single-source precursor for the production of ZnO nanoparticles. Thermal decomposition of the precursor was complete in less than 1 h at a heating rate of 10 °C min−1. Full characterization of the resulting nanoparticles revealed the formation of monodispersed hexagonal zincite structure with an average size of 117 ± 4 nm.30
2 molar ratio afforded the dinuclear zinc complex [Zn(Curc)(OH)]2 which could be employed as a single-source precursor for the production of ZnO nanoparticles. Thermal decomposition of the precursor was complete in less than 1 h at a heating rate of 10 °C min−1. Full characterization of the resulting nanoparticles revealed the formation of monodispersed hexagonal zincite structure with an average size of 117 ± 4 nm.30
5. Medicinal applications of metal curcumin complexes
Without any doubt the most exciting aspect of metal curcumin complexes is the finding that many of them exhibit very diverse and highly promising health effects similar to curcumin itself. Thus it is not surprising that this section constitutes the most important part of this Tutorial Review. One of the first biological investigations on metal curcumin complexes was published as early as 1987 and comprises an antiarthritic study of the orally active gold(III) curcumin complex [Au(Curc)2]Cl. This complex was prepared in a simple manner by mixing curcumin with gold(III) chloride in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio in ethanol and characterized by elemental analysis, magnetic moment, IR and electronic spectral studies. It remained unclear, however, whether or not the chloride ion is directly coordinated to gold, as the conductivity of the complex could not be determined due to its poor solubility in all common organic solvents. Antiarthritic studies on rats were carried out using three parameters: (1) assessment of remission in adjuvant-induced polyarthritis by measurement of paw volume, (2) X-ray studies to see the anatomical changes of the affected limb, and (3) changes in the serum acid phosphatase level during the disease course. The serum gold levels were also determined at different dose levels.37 Today, virtually all metal curcumin complexes are being investigated with biological and medicinal studies in mind. Notable is an impressive increase of relevant contributions to this field in the past 3–5 years.
1 molar ratio in ethanol and characterized by elemental analysis, magnetic moment, IR and electronic spectral studies. It remained unclear, however, whether or not the chloride ion is directly coordinated to gold, as the conductivity of the complex could not be determined due to its poor solubility in all common organic solvents. Antiarthritic studies on rats were carried out using three parameters: (1) assessment of remission in adjuvant-induced polyarthritis by measurement of paw volume, (2) X-ray studies to see the anatomical changes of the affected limb, and (3) changes in the serum acid phosphatase level during the disease course. The serum gold levels were also determined at different dose levels.37 Today, virtually all metal curcumin complexes are being investigated with biological and medicinal studies in mind. Notable is an impressive increase of relevant contributions to this field in the past 3–5 years.
5.1. (Photo)cytotoxicity and anticancer activity
Not surprisingly, this area has become the hottest topic in the field in the past three years, although initial findings on anticancer activity of metal curcumin complexes date back until the year 1998. In the following years, several transition metal complexes of curcumin and curcuminoid ligands have been prepared and successfully tested in vitro and in vivo for antitumor effects. Among a series of vanadyl (V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), Co(II), Ni(II) and Cu(II) curcumin complexes, the copper(II) derivatives turned out to exhibit the highest selective cytotoxicity and also showed significant reduction in solid tumor volume in ascites tumor-bearing mice. IC50 values of 6.6–11.1 μM in Ehrlich ascites tumor cells were reported for such copper(II) curcumin complexes. Also notable among these earlier results is the readily accessible and versatile vanadyl compound VO(Curc)2 (“vanadyl curcumin”) which was first reported in 2004. This simple though highly remarkable compound showed antirheumatic activity in synoviocytes, angiogenesis inhibition in smooth muscle cells and promising anticancer potential in mouse lymphoma cells (L1210). At the same time, VO(Curc)2 proved to be exceptionally non-toxic and showed no negative symptoms during a 1 month treatment period at doses up to 2.0 mmol kg−1 day−1.21 Tests of the closely related vanadyl curcuminoid complexes VO(DMCurc)2, VO(BDMCurc)2, VO(DACurc)2, and VO(DABDMCurc)2 in all cases revealed similar cytotoxicity in mouse lymphoma cells as for vanadyl curcumin.20
O), Co(II), Ni(II) and Cu(II) curcumin complexes, the copper(II) derivatives turned out to exhibit the highest selective cytotoxicity and also showed significant reduction in solid tumor volume in ascites tumor-bearing mice. IC50 values of 6.6–11.1 μM in Ehrlich ascites tumor cells were reported for such copper(II) curcumin complexes. Also notable among these earlier results is the readily accessible and versatile vanadyl compound VO(Curc)2 (“vanadyl curcumin”) which was first reported in 2004. This simple though highly remarkable compound showed antirheumatic activity in synoviocytes, angiogenesis inhibition in smooth muscle cells and promising anticancer potential in mouse lymphoma cells (L1210). At the same time, VO(Curc)2 proved to be exceptionally non-toxic and showed no negative symptoms during a 1 month treatment period at doses up to 2.0 mmol kg−1 day−1.21 Tests of the closely related vanadyl curcuminoid complexes VO(DMCurc)2, VO(BDMCurc)2, VO(DACurc)2, and VO(DABDMCurc)2 in all cases revealed similar cytotoxicity in mouse lymphoma cells as for vanadyl curcumin.20
Until now, anticancer activity has been reported for metal curcuminoid complexes of the Group 13 elements Al, Ga and In, the first-row transition metals V and Fe–Zn, the second-row transition metals Ru and Pd, as well as some rare-earth metals. The majority of the complexes studied are vanadyl (V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) and copper(II) derivatives, followed by zinc complexes. Notable among the main-group metal complexes is a study reporting the synthesis and characterization of 68Ga-labeled curcumin and curcuminoid complexes as potential radiotracers for cancer imaging. The complexes 68Ga(Curc)+ and 68Ga(DACurc)+ as well as a related complex containing bis(dehydroxy)curcumin were evaluated for their uptake by A549 lung cancer cells. All three complexes showed a quite high uptake in the lung cancer cells which was at least equivalent to the respective free curcuminoids, thus confirming their potential applications as cancer-detecting radiotracers.18 As mentioned in Section 3, a considerable number of vanadyl curcumin complexes comprising a variety of spectator ligands have been synthesized and fully characterized. These complexes have been extensively and successfully studied for the photoinduced anticancer activity.22,23 Photodynamic therapy is a promising and non-invasive method for treatment of various cancers. The treatment involves the combined use of a photosensitizer, visible light and molecular oxygen to generate reactive oxygen species (=ROS, cf. also Section 5.3) which are cytotoxic. Under optimal conditions, the ROS will damage only the photoexposed cancer cells while leaving the unexposed healthy cells unaffected.22 Very recently, the utility of heteroleptic vanadyl curcumin complexes as photodynamic therapy agents has been explored in a series of studies with highly promising results. Virtually all complexes studied showed remarkable photocytotoxicity in selected cancer cells (e.g. HeLa, a cell line often used in cancer research, as well as Hep G2 and 3T3 cells) in visible light, while being relatively non-toxic in the dark. Mechanistic studies revealed either singlet oxygen (1O2) or hydroxyl radicals as the reactive oxygen species. The results also suggested light-induced cell death by the curcumin complexes via an apoptotic pathway.22,23
O) and copper(II) derivatives, followed by zinc complexes. Notable among the main-group metal complexes is a study reporting the synthesis and characterization of 68Ga-labeled curcumin and curcuminoid complexes as potential radiotracers for cancer imaging. The complexes 68Ga(Curc)+ and 68Ga(DACurc)+ as well as a related complex containing bis(dehydroxy)curcumin were evaluated for their uptake by A549 lung cancer cells. All three complexes showed a quite high uptake in the lung cancer cells which was at least equivalent to the respective free curcuminoids, thus confirming their potential applications as cancer-detecting radiotracers.18 As mentioned in Section 3, a considerable number of vanadyl curcumin complexes comprising a variety of spectator ligands have been synthesized and fully characterized. These complexes have been extensively and successfully studied for the photoinduced anticancer activity.22,23 Photodynamic therapy is a promising and non-invasive method for treatment of various cancers. The treatment involves the combined use of a photosensitizer, visible light and molecular oxygen to generate reactive oxygen species (=ROS, cf. also Section 5.3) which are cytotoxic. Under optimal conditions, the ROS will damage only the photoexposed cancer cells while leaving the unexposed healthy cells unaffected.22 Very recently, the utility of heteroleptic vanadyl curcumin complexes as photodynamic therapy agents has been explored in a series of studies with highly promising results. Virtually all complexes studied showed remarkable photocytotoxicity in selected cancer cells (e.g. HeLa, a cell line often used in cancer research, as well as Hep G2 and 3T3 cells) in visible light, while being relatively non-toxic in the dark. Mechanistic studies revealed either singlet oxygen (1O2) or hydroxyl radicals as the reactive oxygen species. The results also suggested light-induced cell death by the curcumin complexes via an apoptotic pathway.22,23
Notable among late transition metal curcumin complexes exhibiting antitumor activity are particularly ruthenium, palladium, copper and zinc derivatives.26–29,34–36 Well-defined ruthenium complexes for which antitumor activity has been reported, include (η6-p-cymene)RuCl(Curc) and (η6-p-cymene)RuCl(DMCurc)35 as well as the novel cationic ruthenium mono(curcumin) complexes containing an η6-bonded arene ligand (p-cymene or hexamethylbenzene) and the bulky phosphine ligand 1,3,5-triaza-7-phosphaadamantane (=PTA) in addition to the curcuminoid ligands (cf.Fig. 7).34 When tested in vitro, both (η6-p-cymene)RuCl(Curc) and (η6-p-cymene)RuCl(DMCurc) displayed good antitumor activity in colon-rectal tumor (HCT116), breast (MCF7) and ovarian carcinoma cell lines (A2780 and A2780cisR), whereas human glioblastoma (U-87) and lung carcinoma (A549) cells were less sensitive. This indication of selectivity was seen as a positive feature for future developments. A DNA docking study of (η6-p-cymene)RuCl(Curc) revealed the same mechanism of action that has been established in Pt chemotherapy.35 The ruthenium η6-arene-PTA–curcumin complexes showed not only superior solubility properties due to the sophisticated ligand design but also superior cytotoxicities compared to other classes of related compounds. On the cancer cell lines tested (human ovarian carcinoma A2780 cells and the A2780cisR variant with acquired resistance to cisplatin), these new Ru compounds were found to be ca. 100-fold more cytotoxic, with IC50 values being typically ≤1 μM, while maintaining an excellent selectivity index (i.e., they are considerably less cytotoxic to non-tumorous human embryonic kidney cells). It is interesting to note, that in this case the presence or absence of peripheral methoxy groups in curcumin and the different arene rings did not strongly influence the biological activity. However, the PTA ligand appears to significantly improve the pharmacological properties of the curcumin-modified ruthenium(II)–arene complexes. It has been pointed out that the superior cytotoxicity and selectivity index in comparison to clinically used cisplatin clearly warrants further development of these complexes.34 In the case of palladium, antitumoral effects of the heteroleptic cationic complex [(bipy-9)Pd(Curc)][CF3SO3] (bipy-9 = dinonyl-2,2′-bipyridine) have been studied in more detail. This compound was synthesized with the aim to determine a synergism around the palladium metal center in order to improve the performances of the single components, namely the bipyridine derivative and the curcumin, which are both biologically active ligands. In fact, the study revealed a greatly increased biological activity of the palladium complexes. The combined test data confirmed/validated the significance of curcumin complexation to a metal center and its conjugation to another functionalized bioactive ligand in the apoptosis signal transduction and enhancement of cell death in human prostate cancer cell lines (LnCaP, PC3, and DU145) and suggested the potential of this design strategy in the improvement of the metal-based drugs cytotoxicity.36 The copper(II) complexes Cu(Et2Curc)2(1,4-dioxane) (Et2CurcH = 1,7-bis(4-ethoxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) and Cu(Bu2Curc)2 (Bu2Curc = 1,7-bis(4-n-butoxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) have already been mentioned in Section 3 because of their increased solubility and crystallinity due to blocking of the phenolic –OH groups through alkylation. Both complexes showed significantly enhanced antitumor activity against three human cancer cell lines (ASPC-1 (pancreatic carcinoma), MCF-7 (breast cancer) and HeLa (cervical cancer)) in comparison with the free ligands. These findings once again underlined the importance of exploring the coordination chemistry of curcumin derivatives as an effective method to improve their antitumor activity.26Scheme 7 illustrates three advanced copper curcumin complexes comprising N-ferrocenylmethyl-L-amino acids as co-ligands. The DNA photocleavage activity, photocytotoxicity and cellular localization in HeLa and MCF-7 cancer cells of these complexes have been studied. All three complexes exhibited high photocytotoxicity with low dark toxicity, thus showing a remarkable photodynamic effect leading to apoptosis of the cancer cells.27
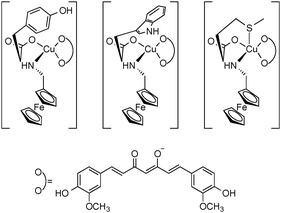 | ||
| Scheme 7 Schematic representation of copper(II) curcumin complexes comprising N-ferrocenylmethyl-L-amino acids as spectator ligands. | ||
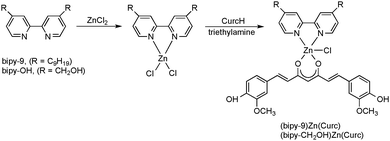
Highly promising results have also been obtained with heteroleptic zinc curcumin complexes as non-classical anticancer agents. In this case, special disubstituted 2,2′-bipyridine ligands were employed as spectator ligands to provide good solubility and crystallinity. The complexes shown in Scheme 8 were synthesized by treatment of the chloro precursors with curcumin in the presence of triethylamine. The dinonyl-2,2′-bipyridine derivative could be structurally authenticated through X-ray diffraction, being yet another successful example of the spectator ligand approach (cf. Section 3).28,29
When tested in vitro towards different human prostate cancer cell lines (DU145, LNCaP and PC-3), both new Zn(II) curcumin complexes showed promising and selective anticancer properties. In particular the dinonyl-2,2′-bipyridine Zn(II) curcumin complex showed the strongest growth inhibition in all cell lines, being even more effective than free curcumin in a neuroblastoma cell line (LAN-5). Furthermore, the curcumin ligand makes the complexes fluorescent, a feature enabling investigation of their interaction with DNA through optical methods. The presence of intrinsic fluorescence in (bipy-9)Zn(Curc) and (bipy-CH2OH)Zn(Curc) allows the combination of anticancer properties with an excellent tool for investigating their mechanism of action through optical methods in a single molecule, without additional external agents.28,29 Based on these encouraging results, a series of new Zn(II)–curcumin-based heteroleptic complexes have been synthesized and fully characterized, with the aim to improve the bioactivity of the potential antitumor agent (bipy-9)Zn(Curc). Several structural changes were made starting from the reference complex (bipy-9)Zn(Curc), in order to introduce new functionalities, such as electrostatic and/or covalent interactions. Keeping the dinonyl-2,2′-bipyridine ligand, two different Zn(II) species were obtained: a tetracoordinated Zn(II) cation with BF4− as counterion and a dimeric neutral complex bridged by a sulfate anion. Moreover, other N,N′-chelating ligands such as phenanthroline (=phen) were employed. The antitumor activity of all new Zn(II) complexes was tested in vitro against a human neuroblastoma cell line (SH-SY5Y). The results showed that all complexes exhibited strong cytotoxic activity. In particular, the ionic tetrafluoroborate Zn(II) complex [(bipy-9)Zn(Curc)]BF4 and the neutral phenanthroline-based Zn(II) derivative (phen)Zn(Curc)Cl (Scheme 9) showed the strongest growth inhibition, being even more effective than the model complex (bipy-9)Zn(Curc).29
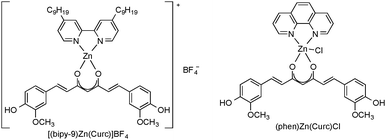 | ||
| Scheme 9 Schematic representation of the Zn(II)–curcumin-based antitumor agents [(bipy-9)Zn(Curc)]BF4 and (phen)Zn(Curc)Cl. | ||
Unprecedented photoactivated anticancer activity has also been reported for the lanthanide complexes depicted in Scheme 5. It was found that these lanthanide curcumin complexes display remarkable photocytotoxicity in HeLa cancer cells giving IC50 values that were comparable to that of the established photodynamic therapy (PDT) drug Photofrin®. The glycosylated curcumin complexes showed higher solubility in aqueous media but had only moderate photocytotoxicity in visible light. These curcumin complexes are essentially non-toxic in the dark but become highly cytotoxic upon photoactivation. Moreover, coordination of curcumin to Ln(III) ions increases the hydrolytic stability and photocytotoxicity of curcumin. Clearly this work opens up a new area of research on the photochemotherapeutic applications of curcumin and its lanthanide complexes. It was shown that nuclear localization of the complexes in HeLa cells could enable the complexes to bind the chromosomal DNA and damage the nuclear DNA on photoactivation. With a significant photodynamic therapy effect, the curcumin lanthanide complexes could serve the dual purpose of detecting a tumor while remaining dormant inside the cells in the dark until subjected to photoactivation causing apoptotic cellular death selectively in the photo-exposed cells while leaving the unexposed cells unaffected. In contrast, lanthanide-based MRI agents can be used only for tumor detection but not for any tumor damage.43
5.2. Anti-Alzheimer's disease activity
Another important area of research involving metal curcumin complexes is directed toward the imaging/radioimaging and possible treatment of Alzheimer's disease (AD). Alzheimer's disease is a progressive, degenerative, and devastative disorder which attacks the brain's nerve cells (neurons), resulting in progressive loss of memory, thinking, language skills, and judgment. AD is the most common cause of dementia among people of age 65 or older. It appears well established that the gradual loss of brain function characteristic for AD is connected to two main forms of nerve damage. One form is the development of so-called neurofibrillary tangles or β-sheet-rich fibrils inside the neurons. These are insoluble twisted fibers formed by aggregation of the tau protein. Outside and around the neurons, so-called β-amyloid plaques, sticky clumps of cellular material and fragments of the amyloid-β-peptide called Aβ, can form. The aggregation can be induced by multiple factors, including the presence of free radicals, oxidative stress, and the presence of excessive metal ions. Metal ions which are believed to be potential risk co-factors in neurodegenerative disorders like AD include Al3+, Mn2+, Fe3+, Cu2+ and Zn2+. The involvement of these metal ions in neurodegenerative disorders has been confirmed by post-mortem analyses of brain tissues. Apparently, Al3+ has the most harmful effect in terms of fibrillation and β-amyloid plaque formation.15 During the past 10 years, strong evidence has been accumulated that curcumin is an efficient natural drug for treatment of AD either by scavenging free radicals or by blocking the Aβ-aggregation.19 Due to its lipophilic character, curcumin can cross the blood–brain barrier, destabilize the preformation of β-amyloid fibrils, and eventually bind to plaques and inhibit formation of β-amyloid fibrils.17 Being a natural β-diketone derivative, curcumin is also well established to strongly chelate the metal ions Al3+, Mn2+. Fe3+, Cu2+ and Zn2+. Thus a number of recent studies has been directed to the use of metal curcumin complexes as inhibitors of metal-induced neurotoxicity with the goal of developing new diagnostic tools for AD and eventually new therapeutic agents against this disease that affects millions of patients worldwide.One interesting aspect in this area of research is the development of potential radiotracers for imaging of Alzheimer's disease based on metal curcumin complexes. In this respect, several gallium, technetium, and rhenium complexes of curcumin and some of its derivatives have been investigated. In the case of gallium, 68Ga-labeled complexes of curcumin as well as diacetylcurcumin (DACurcH) and bis(dehydroxy)curcumin have been synthesized and fully characterized. Gallium-68 was chosen because it is a generator-produced radionuclide with suitable energy and half-life (T1/2 = 67.7 min) useful for many applications in nuclear medicine. The radiotracers were prepared by reaction of 68Ga3+ obtained from a 68Ge/68Ga generator with 1 mg mL−1 curcumin ligand solutions. A first evaluation of their affinity to synthetic β-amyloid fibrils was performed to show the potential application of these new labeled curcuminoids in this diagnostic field. The results were encouraging since 68Ga(Curc)+ and 68Ga(DAC)+ maintained a high affinity for synthetic β-amyloid fibrils.18 Promising results have also been obtained with technetium and rhenium. The synthesis and characterization of curcumin complexes of the [M(CO)3]+ (M = Re, 99mTc) core and imidazole or isocyanocyclohexane as monodentate spectator ligands have been reported. The complexes were synthesized by treatment of the [NEt4]2[Re(CO)3Br3] precursor with curcumin to generate the intermediate aqua complex fac-Re(CO)3(Curc)(H2O), followed by replacement of the labile coordinated water by the monodentate ligand. The chemistry was successfully transferred at 99mTc tracer level. Three of these curcumin complexes were successfully tested for selective staining of β-amyloid plaques of Alzheimer's disease. The binding affinity for β-amyloid plaques was examined following standard staining procedures. In a closely related study, both the monophosphine and bis(phosphine) curcumin rhenium complexes fac-Re(CO)3(PPh3)(Curc) and cis–trans-Re(CO)2(PPh3)2(Curc) were also found to show selective binding to β-amyloid plaques of Alzheimer's disease. Fig. 10 shows the results of the in vitro staining of human post-mortem AD fixed brain sections with the complexes fac-Re(CO)3(PPh3)(Curc) (A) and cis–trans-Re(CO)2(PPh3)2(Curc) (B) as they appear under the fluorescence microscope. The results of staining with plain curcumin (C) are also shown as a positive control. It was found that both complexes bind selectively to the plaques, allowing clear visualization of both diffused and dense core ones. Even though quantitative comparison of binding affinities between the curcumin complexes and curcumin is not possible because of differences in fluorescence intensity, it is clear that the rhenium complexes fac-Re(CO)3(PPh3)(Curc) and cis–trans-Re(CO)2(PPh3)2(Curc) stain amyloid plaques in a mode similar to curcumin. These results indicated that rhenium tricarbonyl and dicarbonyl curcumin complexes retain affinity for β-amyloid plaques in the presence of suitable monodentate ligands in their coordination sphere and may serve as a scaffold for the development of a 99mTc-radiodiagnostic for Alzheimer's disease. The fact that the complexes maintain the affinity of the mother compound curcumin for β-amyloid plaques prompts for further exploration of their chemistry and biological properties as radioimaging probes.39
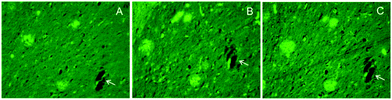 | ||
| Fig. 10 Fluorescence microscopy images (magnification 20×) of almost adjacent closely located sections from Alzheimer's disease brain stained with (A) complex fac-Re(CO)3(PPh3)(Curc), (B) complex cis–trans-Re(CO)2(PPh3)2(Curc), and (C) curcumin. The arrows indicate cross sections of a blood vessel running through the tissue that serve as the anatomical mark for positioning the plaques on the slice. Reproduced from ref. 39. Copyright 2013 American Chemical Society. | ||
Other metal curcumin complexes that have been investigated as potential agents to combat Alzheimer's disease contain the metals Al,15–17 Ga, Cu and Zn.16,19 Several Al(III)–curcumin complexes, e.g. [Al(Curc)(EtOH)2](NO3)2, have been synthesized and characterized by various spectroscopic and analytical methods. It was revealed that curcumin strongly interacts with the Al3+ ion. Curcumin is thus capable of scavenging Al3+ and preventing this metal ion from interacting with proteins like Aβ, thereby weakening the Aβ toxicity and oxidative stress.16,17 Silk protein has been used as a model protein to study the influence of curcumin on the Al(III)-induced fibrillation of neurodegenerative proteins. Using the silk fibroin model, it was shown that Al(III)–curcumin complexes could effectively inhibit the β-sheet formation and even reverse the conformation of the silk protein, which suggests that Al(III)–curcumin complexes could be used as novel agents to prevent proteins from formation of amyloid fibrils and perhaps even removing the preformed amyloid deposits.15 Several copper(II) and zinc(II)–curcumin complexes have also been investigated in order to gain a better understanding of the possible role of curcumin in the AD treatment e.g. by scavenging reactive oxygen species (ROS), blocking Aβ-aggregation and chelating metal ions.15,16
5.3. Antioxidative/neuroprotective activity
Closely related to the anti-Alzheimer's disease activity of metal curcumin complexes is their activity as innovative and relatively non-toxic antioxidants. It is well established that insufficient levels of antioxidants in the human body cause oxidative stress which means the damage of cell structures and cell functions through highly reactive oxygen species (ROS), in particular free radicals. Typical examples for ROS include the superoxide radical anion (O2−˙), the hydroxyl radical (OH˙), and the perhydroxyl radical (HO2−˙), as well as hydrogen peroxide (H2O2) and nitric oxide (NO). For example, excessive production of the free radical NO in the brain has been shown to induce neurotoxicity. Oxidative stress seems to play a crucial role in neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS) as well as Parkinson's, Alzheimer's and Huntington's disease. Typical for neurodegenerative diseases is progressive loss of the structure or function of neurons and eventually even the death of neurons. Thus, antioxidants are considered to be important as neuroprotective agents. In this respect, it is well established that curcumin can act as a potent antioxidant by effectively scavenging free radicals. As a logical extension, several metal curcumin complexes have also been investigated for their antioxidant capacity. Thus far, antioxidant properties have been reported for curcumin complexes of gallium, indium and vanadyl,19,20 as well as manganese32 and copper.33 Particularly instructive from a chemist's point of view is an earlier study describing the synthesis and characterization of dual function vanadyl, gallium and indium curcumin complexes.20 In the course of this study, eight new metal curcumin complexes were synthesized, characterized and tested for biological effects such as antioxidant potential, cytotoxicity, and glucose-lowering activity. In addition to the known vanadyl complex VO(Curc)2 (“vanadyl curcumin”), the analogous vanadyl complexes VO(DMCurc)2, VO(BDMCurc)2, VO(DACurc)2, and VO(DAB-DMCurc)2 were prepared. The new Group 13 metal derivatives comprised homoleptic Ga(Curc)3 and Ga(DACurc)3 as well as In(Curc)3 and In(DACurc)3. The basic idea behind choosing to prepare these pairs of compounds was to test the hypothesis that free radical scavenging would be at least partially impaired in the acetylated complexes as compared to those containing the parent curcuminoid ligands. The question was, whether the blocking of the phenolic –OH groups by acetylation would affect the cytotoxicity and the antioxidant potential of the complexes. In the case of antioxidant activity, the answer is Yes, while the acetylation did not affect the cytotoxicity. All complexes were also tested for their antioxidant activity using the so-called Trolox (6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid) equivalent antioxidant capacity (TEAC) assay. This assay is based on ABTS+˙ (2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)-diammonium salt) radical cation decolorization, as measured spectrophotometrically.20 Comparison of the antioxidant potential revealed that the ligand was the predominant determinant for antioxidative potential. It was indeed found that the activity of VO(DACurc)2 and VO(DABDMCurc)2 was very low as compared to the other vanadyl complexes. The acetylated gallium and indium complexes Ga(DACurc)3 and In(DACurc)3 also exhibited only one-third of the antioxidant activity of the parent curcumin complexes Ga(Curc)3 and In(Curc)3. The results clearly confirmed the initial hypothesis that free aromatic ring –OH groups would be important for high antioxidant capacity of such complexes.20A predominant cellular free radical is the superoxide radical anion (O2−˙). It is involved in various degenerative changes, particularly at low antioxidant concentrations. An important naturally occurring antioxidant is the metalloenzyme superoxide dismutase (SOD), which controls the formation of the superoxide radical anion and catalyzes its conversion to hydrogen peroxide, which can then be reduced to water by the enzymes catalase and/or peroxidase. In this context, several curcumin complexes of copper and manganese have been examined for their SOD activity, free radical-scavenging ability and antioxidant potential. In the case of copper(II) complexes, the distorted orthorhombic 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex Cu(Curc)(OAc)(OH) (OAc = acetate) and the square-planar homoleptic 1
1 complex Cu(Curc)(OAc)(OH) (OAc = acetate) and the square-planar homoleptic 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 complex Cu(Curc)2 have been investigated. It was found that, depending on the coordination geometry, these two complexes exhibit different SOD activities, free radical-neutralizing abilities and antioxidant potentials. The 1
2 complex Cu(Curc)2 have been investigated. It was found that, depending on the coordination geometry, these two complexes exhibit different SOD activities, free radical-neutralizing abilities and antioxidant potentials. The 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cu(II)–curcumin complex Cu(Curc)(OAc)(OH) with the larger distortion from the square-planar structure showed a nearly ten times higher SOD activity than the 1
1 Cu(II)–curcumin complex Cu(Curc)(OAc)(OH) with the larger distortion from the square-planar structure showed a nearly ten times higher SOD activity than the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 complex Cu(Curc)2. From this study it was concluded that the 1
2 complex Cu(Curc)2. From this study it was concluded that the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex would be able to undergo and sustain the distortion from square planar geometry to the distorted tetrahedral one during its reaction with superoxide radical. This allows for the compound to remain intact and undergo many redox cycles and hence act as a very efficient antioxidant. On the other hand, the homoleptic 1
1 complex would be able to undergo and sustain the distortion from square planar geometry to the distorted tetrahedral one during its reaction with superoxide radical. This allows for the compound to remain intact and undergo many redox cycles and hence act as a very efficient antioxidant. On the other hand, the homoleptic 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 complex Cu(Curc)2 is planar but rigid and hence cannot undergo the distortions and therefore is a less powerful antioxidant. These findings clearly underline the need for further synthetic studies and the fine-tuning of the biological activities of metal curcumin complexes through ligand variations.33 In a series of related studies, several manganese(II) curcumin complexes have been evaluated for their neuroprotective activity, including SOD activity and radical scavenging ability.32 A central role among the compounds employed in these studies played the easily accessible manganese(II) complexes Mn(Curc)(OAc)(H2O) and Mn(DACurc)2. The results supported an important role of manganese in importing SOD activity and the enhancement of radical scavenging activity. For example, both Mn(II) complexes showed more effective NO radical scavenging than the respective ligands curcumin and diacetylcurcumin. Therefore it was concluded that Mn(Curc)(OAc)(H2O) and Mn(DACurc)2 could have potential advantages as neuroprotective agents in the treatment of acute brain pathologies associated with NO-induced neurotoxicity and oxidative-stress-induced neuronal damages such as epilepsy, stroke and traumatic brain injury. Both manganese complexes also catalyzed the conversion of superoxide to hydrogen peroxide and oxygen. Questions remained, however, if their stability is good enough for therapeutic purposes. Thus in the Mn(II) case, too, further fine-tuning of the biological activities through careful ligand design appears highly desirable.32
2 complex Cu(Curc)2 is planar but rigid and hence cannot undergo the distortions and therefore is a less powerful antioxidant. These findings clearly underline the need for further synthetic studies and the fine-tuning of the biological activities of metal curcumin complexes through ligand variations.33 In a series of related studies, several manganese(II) curcumin complexes have been evaluated for their neuroprotective activity, including SOD activity and radical scavenging ability.32 A central role among the compounds employed in these studies played the easily accessible manganese(II) complexes Mn(Curc)(OAc)(H2O) and Mn(DACurc)2. The results supported an important role of manganese in importing SOD activity and the enhancement of radical scavenging activity. For example, both Mn(II) complexes showed more effective NO radical scavenging than the respective ligands curcumin and diacetylcurcumin. Therefore it was concluded that Mn(Curc)(OAc)(H2O) and Mn(DACurc)2 could have potential advantages as neuroprotective agents in the treatment of acute brain pathologies associated with NO-induced neurotoxicity and oxidative-stress-induced neuronal damages such as epilepsy, stroke and traumatic brain injury. Both manganese complexes also catalyzed the conversion of superoxide to hydrogen peroxide and oxygen. Questions remained, however, if their stability is good enough for therapeutic purposes. Thus in the Mn(II) case, too, further fine-tuning of the biological activities through careful ligand design appears highly desirable.32
Finally, an innovative recent approach to new curcumin-based antioxidants involves the in situ synthesis and surface functionalization of gold nanoparticles (AuNPs) with curcumin and their antioxidant properties. The curcumin-functionalized AuNPs were synthesized by direct reduction of HAuCl4 using curcumin in the aqueous phase without the need of using any other reducing agents. It was found that under these conditions curcumin acts both as a reducing and capping agent, stabilizing the gold sol for many months. Moreover, these curcumin-capped AuNPs also showed good antioxidant activity which was confirmed by the 2,2-diphenyl-1-picrylhydrazyl radical test. Thus, the surface functionalization of AuNPs with curcumin may pave a new way of using the curcuminoids towards possible drug delivery and therapeutics. Moreover, the conjugation of curcumin appears to be a possibility to increase the bioavailability of curcumin.38
5.4. Other medicinal applications of metal curcumin complexes
A literature survey of the field revealed a surprising variety of other promising and innovative medicinal applications of metal curcumin complexes containing very different metals. Chances are that metal curcumin complexes will become “multi-anti” agents like curcumin itself. The following paragraph intends to give a brief overview of the diverse potential applications. Each entry is followed by a list of the metals which have thus far been employed as well as a short description of one or two characteristic studies.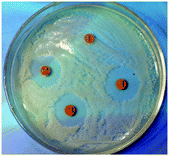 | ||
| Fig. 11 Antibacterial diameters of curcumin (1) and the heteroleptic lanthanide curcumin complexes Ln(Curc)3L (Ln = Eu, Sm, Dy; L = 1,10-phenanthroline-5,6-dione). Reproduced from ref. 40. Copyright 2008 Elsevier B. V. | ||
An innovative aspect in this field is the construction of novel antimicrobial agents from metal nanoparticles and curcumin. In a recent study it was clearly pointed out that the most important future research in antimicrobial therapy is to develop novel materials which could work as effective antimicrobials. In this context, using a direct reduction method (reaction of cobalt(II) chloride with NaBH4 in aqueous solution), 10 nm Co nanoparticles were synthesized and further functionalized with curcumin. The characterization showed FCC (=face-centered-cubic) structured Co nanoparticles to be electrostatically bound to curcumin using the hydroxyl linkage, retaining the magnetic moment of Co as well as the diaryl heptanoid chromophore group of curcumin with the loading percentage of ∼89% (Fig. 12a and b). These samples were then subjected to the gram negative bacterium Escherichia coli, and the antimicrobial activity of nano-Co:curcumin was found to be much enhanced as compared to only Co and only curcumin (Fig. 12c). The synthesized system exhibited a combination of the ability of curcumin to penetrate the cell barrier and render antimicrobial action along with the chelating capacity of the nano-Co:curcumin complex. Thus it was shown that in the same way that strong metal–ligand bonds are vital for the efficiency of metal chelators towards toxic metals, these strongly bound metal complexes may prove to be better antimicrobial agents, binding them to the bacterial cell walls.50
 | ||
| Fig. 12 Interaction between curcumin and Co nanoparticles (black circular cartoon). (a) shows the interaction at the hydroxyl site of curcumin wherein the diaryl-heptanoid group is shown by dotted circles. (b) shows the type of bonding. (c) shows the culture plates for nano-Co:curcumin at t = 0 (a), 3 h (b), 6 h (c), 9 h (d) and finally 12 h (e). A systematic decrease in the microbial growth is observed, which almost reaches 0% after 12 h of dosage. Reproduced from ref. 50. Copyright 2011 Elsevier B. V. | ||
 | ||
| Fig. 13 Fluorescent imaging of MCF-7 breast cancer cells labeled with the Eu(III) complex Eu(Curc)3(pyridine)2 (green) and PI (red) at different times under continuous light exposure (all the scale bars represent 10 mm). Reproduced from ref. 42. Copyright 2012 The Royal Society of Chemistry. | ||
Other promising uses of metal curcumin complexes which have been documented in only a handful of recent studies include e.g. drug formulation with ceramic particles (Ba), the treatment of gastric ulcers (Zn) and iron deficiency (Fe),10 protein determination (La) and the development of innovative vaginal gels (Cu). Some of these medicinal applications are in their infant stage, but all of them are definitely worth further exploration.
6. Conclusions
In summary, this Tutorial Review provides the first general overview of the emerging and rapidly expanding field of metal curcumin complexes and their various applications. Innovative synthetic protocols leading to soluble and crystallizable metal curcumin complexes are highlighted. Although numerous complexes of curcuminoid ligands with various main group, d-transition and f-elements have been reported, there is still a striking paucity of structurally authenticated representatives. Here the spectator ligand approach is expected to provide valuable new insights in the future. In recent years, highly exciting and promising medicinal applications of metal curcumin complexes have been reported. The three most important areas are selective (photo)cytotoxicity and anticancer activity, anti-Alzheimer's disease activity, and antioxidative/neuroprotective effects. Curcumin metal complexes could serve the dual purpose of detecting cancer and causing apoptotic cellular death selectively in the photo-exposed cells leaving the unexposed cells unaffected. In contrast, the lanthanide-based MRI agents are only useful for tumor detection but not for any tumor damage. Metal–curcumin complexes already play an important role in the treatment of Alzheimer's disease e.g. by scavenging reactive oxygen species (ROS), blocking Aβ-aggregation and chelating neuro-toxic metal ions. Curcumin–metal complexes as well as surface functionalized metal nanoparticles with curcumin may pave a new way of using the curcuminoids towards possible drug delivery, therapeutics and excellent anti-oxidants. Moreover, antiarthritic/antirheumatic, antimicrobial/antifungal, antiosteoporotic and antiviral activities have been reported for certain metal curcumin complexes, indicating that these compounds have the potential of becoming “multi-anti” agents in the future like curcumin itself.Acknowledgements
Financial support of the authors' own work by the Otto-von-Guericke-Universität Magdeburg is gratefully acknowledged. Special thanks are due to Ms Desirée Schneider and Christoph Seidel for preparing the ChemDraw Schemes. We thank Professor Edgar Haak for valuable discussions and for critically reading the manuscript.Notes and references
- B. B. Aggarwal, C. Sundaram, N. Malani and H. Ichikawa, Curcumin: The Indian Solid Gold, Springer, New York, 2001 Search PubMed.
- Y. Mawani and C. Orvig, J. Inorg. Biochem., 2014, 132, 52–58 CrossRef CAS PubMed.
- G. P. Lim, T. Chu, F. Yang, W. Beech, S. A. Frautschy and G. M. Cole, J. Neurosci., 2001, 21, 8370–8377 CAS.
- R. Waranyoupalin, S. Wongnawa, M. Wongnawa, C. Pakawatchai, P. Panichayupakaranant and P. Sherdshoopongse, Cent. Eur. J. Chem., 2009, 7, 388–394 CrossRef CAS PubMed.
- A. Goel, A. B. Kunnumakkara and B. B. Aggarwal, Biochem. Pharmacol., 2009, 75, 787–809 CrossRef PubMed.
- M. M. Yallapu, M. Jaggi and S. C. Chauhan, Drug Discovery Today, 2012, 17, 71–80 CrossRef CAS PubMed , and references cited therein.
- P. Anand, A. B. Kunnumakkara and B. B. Aggarwal, Mol. Pharmaceutics, 2007, 4, 807–818 CrossRef CAS PubMed.
- J. Miłobędzka, S. von Kostanecki and V. Lampe, Ber. Dtsch. Chem. Ges., 1910, 43, 2163–2170 CrossRef PubMed.
- M. K. Mishra, P. Sanphui, U. Ramamurty and G. R. Desiraju, Cryst. Growth Des., 2014, 14, 3054–3061 CAS , and references cited therein.
- E. Ferrari, B. Arezzini, M. Ferrali, S. Lazzari, F. Pignedoli, F. Spagnolo and M. Saladini, BioMetals, 2009, 22, 701–710 CrossRef CAS PubMed.
- J. Annarjai, S. Srinivasan, K. M. Ponvel and P. R. Athappan, J. Inorg. Biochem., 2005, 99, 669–676 CrossRef PubMed , and references cited therein.
- J. Rajesh, A. Gubendran, G. Rajagopal and P. Athappan, J. Mol. Struct., 2012, 1010, 169–178 CrossRef CAS PubMed.
- H. J. Roth and B. Miller, Arch. Pharm., 1964, 297, 660–673 CrossRef CAS PubMed , and references cited therein.
- Z. Sui, R. Salto, J. Li, C. Craik and P. R. Ortiz de Montellano, Bioorg. Med. Chem., 1993, 1, 415–422 CrossRef CAS.
- T. Jiang, G.-R. Zhou, Y.-H. Zhang, P.-C. Sun, Q.-M. Du and P. Zhou, RSC Adv., 2012, 2, 9106–9113 RSC.
- T. Jiang, L. Wang, S. Zhang, P.-C. Sun, C.-F. Ding, Y.-Q. Chu and P. Zhou, J. Mol. Struct., 2014, 1004, 163–173 CrossRef PubMed.
- F. Ahmadi, A. A. Alizadeh, N. Shahabadi and M. Rahimi-Nasrabadi, Spectrochim. Acta, Part A, 2011, 79, 1466–1474 CrossRef CAS PubMed.
- E. Ferrari, R. Benassi, S. Sacchi, F. Pignedoli, M. Asti and M. Saladini, J. Inorg. Biochem., 2014, 139, 38–48 CrossRef CAS PubMed.
- M. Asti, E. Ferrari, S. Croci, G. Atti, S. Rubagotti, M. Iori, P. C. Capponi, A. Zerbini, M. Saladini and A. Versari, Inorg. Chem., 2014, 53, 4922–4933 CrossRef CAS PubMed.
- K. Mohammadi, K. H. Thompson, B. O. Patrick, T. Storr, C. Martins, E. Polishchuk, V. G. Yuen, J. H. McNeill and C. Orvig, J. Inorg. Biochem., 2005, 99, 2217–2225 CrossRef CAS PubMed.
- K. H. Thompson, K. Böhmerle, E. Polishchuk, C. Martins, P. Toleikis, J. Tse, V. Yuen, J. H. McNeill and C. Orvig, J. Inorg. Biochem., 2004, 98, 2063–2070 CrossRef CAS PubMed , and references cited therein.
- B. Balaji, B. Balakrishnan, S. Perumalla, A. A. Karande and A. R. Chakravarty, Eur. J. Med. Chem., 2014, 85, 458–467 CrossRef CAS PubMed , and references cited therein.
- B. Banik, K. Somyajit, G. Nagaraju and A. R. Chakravarty, Dalton Trans., 2014, 43, 13358–13369 RSC , and references cited therein.
- M. S. Refat, Spectrochim. Acta, Part A, 2013, 105, 326–337 CrossRef CAS PubMed , and references cited therein.
- Z. Pi, J. Wang, B. Jiang, G. Cheng and S. Zhou, Mater. Sci. Eng., C, 2015, 46, 565–571 CrossRef CAS PubMed , and references cited therein.
- J. Wang, D. Wei, B. Jiang, T. Liu, J. Ni and S. Zhou, Transition Met. Chem., 2014, 39, 553–558 CrossRef CAS , and references cited therein.
- T. K. Goswami, S. Gadadhar, B. Gole, A. A. Karande and A. R. Chakravarty, Eur. J. Med. Chem., 2013, 63, 800–810 CrossRef CAS PubMed.
- D. Pucci, T. Bellini, A. Crispini, I. D'Agnano, P. F. Liguori, P. Garcia-Orduña, S. Pirillo, A. Valentini and G. Zanchetta, Med. Chem. Commun., 2012, 3, 462 RSC.
- D. Pucci, A. Crispini, B. Sanz Mendiguchía, S. Pirillo, M. Ghedini, S. Morelli and L. De Bartolo, Dalton Trans., 2013, 42, 9679–9687 RSC , and references cited therein.
- M. I. Khalil, M. M. Al-Qunaibit, A. M. Al-zahem and J. P. Labis, Arabian J. Chem., 2014, 7, 1178–1184 CrossRef CAS PubMed.
- F. Kühlwein, K. Polborn and W. Beck, Z. Anorg. Allg. Chem., 1997, 623, 1211–1219 CrossRef PubMed.
- Y. Sumanont, Y. Murakami, M. Tohda, O. Vajragupta, H. Watanabe and K. Matsumoto, Biol. Pharm. Bull., 2007, 30, 1732–1739 CAS , and references cited therein.
- A. Barik, B. Mishra, A. Kunwar, R. M. Kadam, L. Shen, S. Dutta, S. Padhye, A. K. Satpati, H.-Y. Zhang and K. I. Priyadarsini, Eur. J. Med. Chem., 2007, 42, 431–439 CrossRef CAS PubMed , and references cited therein.
- R. Pettinari, F. Marchetti, F. Condello, C. Pettinari, G. Lupidi, R. Scopelliti, S. Mukhopadhyay, T. Riedel and P. J. Dyson, Organometallics, 2014, 33, 3709–3715 CrossRef CAS , and references cited therein.
- F. Caruso, M. Rossi, A. Benson, C. Opazo, D. Freedman, E. Monti, M. B. Gariboldi, J. Shaulky, F. Marchetti, R. Pettinari and C. Pettinari, J. Med. Chem., 2012, 55, 1072–1081 CrossRef CAS PubMed.
- A. Valentini, F. Conforti, A. Crispini, A. De Martino, R. Condello, C. Stellitano, G. Rotilio, M. Ghedini, G. Federici, S. Bernardini and D. Pucci, J. Med. Chem., 2009, 52, 484–491 CrossRef CAS PubMed.
- K. K. Sharma, S. Chandra and D. K. Basu, Inorg. Chim. Acta, 1987, 135, 47–48 CrossRef CAS.
- D. K. Singh, R. Jagannathan, P. Khandelwal, P. M. Abraham and P. Poddar, Nanoscale, 2013, 5, 1882–1893 RSC.
- C. Triantis, T. Tsokatos, C. Tsoukalas, M. Sagnou, C. Raptopoulou, A. Terzis, V. Psycharis, M. Pelecanou, I. Pirmettis and M. Papadopoulos, Inorg. Chem., 2013, 52, 12995–13003 CrossRef CAS PubMed.
- Y.-M. Song, J.-P. Xu, L. Ding, Q. Hou, J.-W. Liu and Z.-L. Zhu, J. Inorg. Biochem., 2009, 103, 396–400 CrossRef CAS PubMed.
- M. A. Subhan, K. Alam, M. S. Rahaman, M. A. Rahman and M. R. Awal, J. Sci. Res., 2014, 6, 97–109 CAS.
- S.-S. Zhou, X. Xue, J.-F. Wang, Y. Dong, B. Jiang, D. Wei, M.-L. Wan and Y. Jia, J. Mater. Chem., 2012, 22, 22774–22780 RSC.
- A. Hussain, K. Somyajit, B. Banik, S. Banerjee, G. Nagaju and A. R. Chakravarty, Dalton Trans., 2013, 42, 182–195 RSC.
- J. Atnarez, A. Bonilla and J. C. Vidal, Analyst, 1983, 108, 368–373 RSC.
- I. Ikeuchi and T. Amano, Chem. Pharm. Bull., 1978, 26, 2619–2623 CrossRef CAS.
- K. Lawrence, S. E. Flower, G. Kociok-Köhn, C. G. Frost and T. D. James, Anal. Methods, 2014, 4, 2215–2217 RSC.
- A. Ciszewski, G. Milczarek, B. Lewandowska and K. Krutowski, Electroanalysis, 2003, 15, 518–523 CrossRef CAS PubMed , and references cited therein.
- M. Y. Elahi, M. F. Mousavi and S. Ghasemi, Electrochim. Acta, 2008, 54, 490–498 CrossRef CAS PubMed.
- R. Ojani, J.-B. Raoof and S. Zamani, Bioelectrochemistry, 2012, 85, 44–49 CrossRef CAS PubMed , and references cited therein.
- S. Hatamie, M. Nouri, S. K. Karandir, A. Kulkarni, S. D. Dhole, D. M. Phase and S. N. Kale, Mater. Sci. Eng., C, 2012, 32, 92–97 CrossRef CAS PubMed.
Footnote |
| † Dedicated to Professor Karl-Heinz Thiele on the occasion of his 85th birthday. |
| This journal is © The Royal Society of Chemistry 2015 |