Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
pH switchable anion transport by an oxothiosquaramide†
Robert B. P.
Elmes‡§
a,
Nathalie
Busschaert‡¶
b,
Dawid D.
Czech
b,
Philip A.
Gale
 *b and
Katrina A.
Jolliffe
*a
*b and
Katrina A.
Jolliffe
*a
aSchool of Chemistry (F11), The University of Sydney, 2006 NSW, Australia. E-mail: kate.jolliffe@sydney.edu.au; Fax: +61 2 9351 3329; Tel: +61 2 9351 2297
bChemistry, University of Southampton, Southampton, SO17 1BJ, UK. E-mail: philip.gale@soton.ac.uk; Tel: +44 (0)23 8059 3332
First published on 22nd May 2015
Abstract
An oxothiosquaramide was shown to bind to chloride through hydrogen bonding interactions in DMSO and found to exhibit pH switchable choride transport across phospholipid bilayers via an antiport transport mechanism.
The transport of anions across cellular membranes is an important biological function often mediated by specialised proteins embedded within cellular lipid bilayers.1,2 Intense effort is currently focussed on the development of small synthetic molecules that can mimic the role of such proteins. These have potential to treat diseases characterised by faulty anion transport3,4 or to induce cellular processes such as apoptosis.5,6 We have developed a number of simple hydrogen bonding receptors for anion transport containing urea, thiourea and squaramide groups.7–9 Transporters containing squaramides have proven particularly effective and mediate higher fluxes of anions than the analogous ureas or thioureas due to significantly higher anion affinities.9,10 More recently, we have shown that thiosquaramides display pH-dependent anion transport behaviour mimicking the gating/switching behaviour seen in biological systems.11 Thiosquaramides are significantly more acidic than their oxosquaramide analogues, and at pH > 7 are present as deprotonated species, unable to bind to or transport anions. Lowering the pH reprotonates the thiosquaramide and switches on anion transport, providing a potential method for targeting acidic environments in biological systems, without the need for external stimulation.
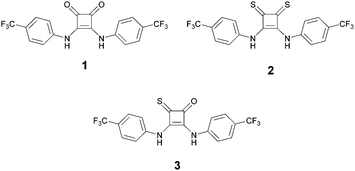
Given the significant differences in anion binding strength and pKa between oxo- and thiosquaramides,11–13 we were intrigued by the possibility that a mixed oxothiosquaramide derivative might exhibit intermediate behaviour between the two extremes, with ‘switch-ON’ anion transport behaviour occurring at a different pH to that observed for 2, which could be more relevant to switching in biological systems. We therefore report here the synthesis, chloride binding and transport properties of the mixed oxothiosquaramide 3.
We previously found that conversion of oxosquaramide 1 to the thiosquaramide analogue 2 was most readily achieved using an excess of the crystalline P4S10·pyridine thionating agent14 in MeCN.11 We reasoned that treatment of 1 with 1 equivalent of this reagent should yield a statistical mixture of compounds 1–3,15 and found that this mixture was readily separable by flash chromatography to give 3 in 26% yield (see ESI†).
Mixed oxothiosquaramides have not previously been investigated as anion binding motifs. In order to evaluate the chloride binding ability of 3, 1H NMR spectroscopic titrations at 298 K in 0.5% H2O in DMSO-d6 were performed by addition of tetrabutylammonium chloride (TBACl). The data (Δδ) was fitted to a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding model using Hyperquad© (see ESI† for fitted data).16 The association constants (Ka) of 1–3 in 0.5% H2O in DMSO-d6 with Cl− are summarised in Table 1. Both NH signals of 3 underwent significant downfield shifts (1.31 and 1.75 ppm) upon titration with chloride, indicating that both protons are involved in hydrogen bonding. Compound 3 exhibited similar affinity for Cl− to that of its squaramide counterpart 1, suggesting that incorporation of a single sulfur atom does not significantly alter the anion binding capacity of these compounds. This contrasts with the behaviour of thiosquaramide 2, which binds only weakly to Cl− in this solvent.
1 binding model using Hyperquad© (see ESI† for fitted data).16 The association constants (Ka) of 1–3 in 0.5% H2O in DMSO-d6 with Cl− are summarised in Table 1. Both NH signals of 3 underwent significant downfield shifts (1.31 and 1.75 ppm) upon titration with chloride, indicating that both protons are involved in hydrogen bonding. Compound 3 exhibited similar affinity for Cl− to that of its squaramide counterpart 1, suggesting that incorporation of a single sulfur atom does not significantly alter the anion binding capacity of these compounds. This contrasts with the behaviour of thiosquaramide 2, which binds only weakly to Cl− in this solvent.
To compare the acidity of the NH protons in compounds 1–3, pKa values were determined by pH-spectrophotometric titrations in a mixture of acetonitrile/water (9/1 v/v; in the presence of 0.1 M TBAPF6) as described previously.11 Under these conditions, 3 was observed to undergo a single deprotonation event, characterised by a hypochromism at ∼372 nm and a concomitant hyperchromism at ∼396 nm (see Fig. 1). The titration profiles comparing 1–3 are shown in Fig. 1. The obtained pKa values corresponding to a single deprotonation event are summarised in Table 1. As expected, the mixed oxothiosquaramide 3 has a pKa intermediate between those of 1 and 2. However, the pKa is much closer to that of the dithionated derivative 2 than to that of 1. Importantly, the pKa value of 6.6 suggests that 3 also exists in its deprotonated form at neutral pH and may therefore be useful as a pH-switchable anion transporter.
In order to evaluate the chloride transport properties of compound 3, previously reported vesicle-based procedures17 were employed under conditions allowing direct comparison to the reported chloride transport properties of 1 and 2.9,11 Unilamellar 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC) liposomes loaded with a NaCl solution (489 mM) buffered to pH 7.2 were prepared and suspended in an isotonic NaNO3 solution (489 mM) buffered to pH 7.2. A small volume of a DMSO solution of 3 was added to these liposomes and the resulting chloride efflux was monitored using an ion selective electrode (ISE). An analogous series of experiments were also performed at pH 4.0. The results for oxothiosquaramide 3 (Fig. 2) indicate that, like the thiosquaramide analogue 2, 3 possesses significantly enhanced anion transport ability at pH 4.0, compared to that observed at pH 7.2 (the results for the oxo-/thiosquaramides can be found in the ESI†). This is consistent with the pKa value determined for 3 (6.6), which implies that at pH 7.2 a significant proportion of this receptor will be deprotonated and thus negatively charged. Interestingly, the transport behaviour of 3 at low loading (0.1 mol%) showed a more pronounced pH switching effect compared to higher loadings (1 mol%). This suggests that the ‘trigger’ to switch on anion transport may occur at a higher pH for 3 compared to that required for receptor 2.
Hill analysis18 of the anion transport of 3 was performed at both pH 7.2 and pH 4.0, and revealed that 3 is a poor anion transporter at pH 7.2 but is an efficient anion transporter at pH 4.0 (Table 2). The mixed oxothiosquaramide derivative 3 was found to exhibit chloride transport efficiencies intermediate between those of 1 and 2. At pH 7.2, transport activity was observed to follow the trend 1 > 3 > 2 which is consistent with the percentage of deprotonation as indicated by the trend in pKa values. A similar dependence of anion transport on the percentage of protonated receptor at physiological pH has also been observed for prodigiosenes.19 At pH 4.0, transport activity follows the general trend 2 > 3 > 1 and is consistent with a combination of lipophilicity (as determined by HPLC), which is an important factor in transport ability, and the expected anion binding ability of the receptors in their fully protonated forms.20–23
| EC50 at pH 7.2a (mol%) | EC50 at pH 4.0a (mol%) | Retention timeb (min) | Apparent pKac | |
|---|---|---|---|---|
| a Concentration of transporter (mol% with respect to lipid) needed to achieve 50% chloride efflux in 270 s from POPC vesicles filled with NaCl and buffered to pH 7.2 or pH 4.0. b Retention time of the compounds on a reversed-phase HPLC column. c pKa value obtained by performing anion transport studies at various pH (see main text for details). d Value taken from ref. 11. | ||||
| 1 | 0.06d | 0.08d | 26.0 | 11.2 (±0.2) |
| 2 | 0.68d | <0.0125d | 26.6 | 6.0 (±0.2) |
| 3 | 0.22 | 0.03 | 26.7 | 6.9 (±0.1) |
The chloride transport mediated by compound 3 may be either via the formation of membrane-spanning channels or via a mobile carrier mechanism. Oxo and thiosquaramides have previously been shown to function primarily via a mobile carrier mechanism9,11 and a series of vesicle-based experiments point towards 3 operating via a similar mechanism (see ESI†). Calcein leakage assays24 revealed that the large highly anionic dye calcein does not leak from POPC vesicles at pH 7.2 or pH 4.0 in the presence of 3 (see ESI†), excluding the formation of pores or channels with large internal diameter and vesicle disruption induced by the transporters.
The charge imbalance created by transmembrane anion transport can be compensated by means of an antiport process or a symport process. The chloride efflux shown in Fig. 2 is therefore most likely the result of either a pure ‘switch-on’ Cl−/NO3− antiport mechanism or the occurrence of a H+/Cl− symport process at lower pH (or a combination of both). To investigate which mechanism is dominant for receptor 3, POPC liposomes containing a NaCl solution and suspended in a Na2SO4 solution were prepared at pH 7.2 and pH 4.0. The double negatively charged sulfate anion is considerably more hydrophilic than nitrate (ΔGhydr(SO42−) = −1080 kJ mol−1; ΔGhydr(NO3−) = −300 kJ mol−1)25 and is therefore more challenging to transport across a lipid bilayer. It was found that under these conditions the efficiency of chloride transport was significantly reduced with a minimal amount of chloride being transported out of the liposomes at both pH 7.2 and pH 4.0 (see ESI†). While these results strongly suggest that an antiport mechanism is the dominant process for the chloride transport facilitated by 3, in principle HCl symport may account for some of the anion transport efficiency. A series of vesicle-based experiments were thus conducted using the pH-dependent fluorescence properties of 8-hydroxy-1,3,6-pyrenetrisulfonate (HPTS) in order to clarify this effect (see ESI†).24 Initially, POPC liposomes were prepared containing NaCl (489 mM) and HPTS (1 mM) buffered to pH 7.2 or pH 6.0, and suspended in a NaNO3 (489 mM) solution buffered to pH 7.2 or pH 6.0. The results revealed that 3 is capable of effecting large changes in intravesicular pH even in the absence of a pH gradient (although to a lesser extent). The ability of 1 and 2 to change intravesicular pH was previously observed, however in the absence of a pH gradient the intravesicular pH did not change significantly upon the addition of these transporters (see ESI†).11 Taken together, these results indicate that the chloride effluxes shown in Fig. 2 are mainly the result of Cl−/NO3− antiport processes but that H+/Cl− symport (or OH−/Cl− antiport) may also be an underlying mechanism. In conclusion, it appears that the chloride transport ability of receptor 3 is mainly the result of a Cl−/NO3− antiport mechanism that can be switched on by lowering the pH, while H+/Cl− symport may also be contributing to the overall transport ability of 3.
To probe whether the pH-switch of the thiosquaramides mirrors the pKa values obtained through spectrophotometric titrations, the Cl−/NO3− experiments depicted in Fig. 2 were repeated at a range of pHs for receptors 1–3 (see ESI† for details on the different pHs and buffers used). The exact position of the pH switch should correspond to the pKa value of the receptor under the conditions of the anion transport experiments (‘apparent pKa’).11 In order to calculate the apparent pKa of 3, we plotted the normalised initial rate of chloride efflux versus pH and fitted the data to a sigmoidal function (Fig. 3). The values obtained using this method are summarised in Table 2 and largely agree with the pKa values determined above. These results clearly show that the chloride transport ability of 3 can be switched on at pH < 7, with the major switch occurring at pH ≈ 7.0. Fig. 3 also confirms that the chloride transport by the analogous (thio)squaramides 1 and 2 is also pH-dependent but in different pH windows. Clearly, squaramide 1 displays potent transmembrane anion transport activity across a large pH range (pH 4–10), while thiosquaramide 2 is inactive above pH ≈ 6 and oxothiosquaramide 3 shows activity over an intermediate pH range.
 | ||
| Fig. 3 pH dependence of the chloride transport ability of oxo/thio/mixed squaramides 1–3 (1 mol%). Chloride transport experiments were conducted as described in Fig. 2 but at various different pHs. The obtained data was fitted to an asymptotic function and the initial rate of chloride efflux (kini) was calculated (see ESI† for details) and plotted as a function of pH after normalising by the maximum rate achievable at pH 4.0. Apparent pKa values could subsequently be estimated by fitting the obtained plot to the following sigmoidal function: y = max + ((min − max)/(1 + 10(pKa−x))). | ||
In summary we have reported the first use of a mixed oxothiosquaramide as an efficient chloride receptor and pH-switchable chloride transporter. Receptor 3 has similar anion binding properties to oxosquaramide 1, forming strong complexes with chloride anions, as indicated by proton NMR titrations in DMSO-d6. However, its pKa is closer to that of thiosquaramide 2. Vesicle studies indicated that the trans-membrane anion transport ability of 3 is switched OFF at pH > 7 and switched ON at pH < 7. Importantly, the effect of the addition of one sulfur to the squaramide structure is large enough to cause the desired pH switch for transport while addition of a second sulfur moves the switch to lower pH. This paper provides further evidence for the controllable and switchable anion transport nature of synthetic receptors containing the squaramide moiety. We are currently studying these and related systems for the development of future biologically active anion transporters.
We thank the EPSRC (EP/J009687/1) and the ARC (DP140100227) for funding. PAG thanks the Royal Society and the Wolfson Foundation for a Royal Society Wolfson Research Merit Award. PAG and KAJ thank the European Cooperation in Science and Technology (COST) action CM1005 “Supramolecular Chemistry in Water” for support.
Notes and references
- J. T. Davis, O. Okunola and R. Quesada, Chem. Soc. Rev., 2010, 39, 3843–3862 RSC.
- F. M. Ashcroft, Ion Channels and Disease, Academic Press, San Diego, 2000 Search PubMed.
- P. A. Gale, R. Pérez-Tomás and R. Quesada, Acc. Chem. Res., 2013, 46, 2801–2813 CrossRef CAS PubMed.
- N. Busschaert and P. A. Gale, Angew. Chem., Int. Ed., 2013, 52, 1374–1382 CrossRef CAS PubMed.
- S.-K. Ko, S. K. Kim, A. Share, V. M. Lynch, J. Park, W. Namkung, W. Van Rossom, N. Busschaert, P. A. Gale, J. L. Sessler and I. Shin, Nat. Chem., 2014, 6, 885–892 CrossRef CAS PubMed.
- J. T. Davis, in Topics in Heterocyclic Chemistry, ed. P. A. Gale and W. Dehaen, Springer, Berlin, Heidelberg, 2010, vol. 24, pp. 145–176 Search PubMed.
- L. E. Karagiannidis, C. J. E. Haynes, K. J. Holder, I. L. Kirby, S. J. Moore, N. J. Wells and P. A. Gale, Chem. Commun., 2014, 50, 12050–12053 RSC.
- C. J. E. Haynes, N. Busschaert, I. L. Kirby, J. Herniman, M. E. Light, N. J. Wells, I. Marques, V. Felix and P. A. Gale, Org. Biomol. Chem., 2014, 12, 62–72 CAS.
- N. Busschaert, I. L. Kirby, S. Young, S. J. Coles, P. N. Horton, M. E. Light and P. A. Gale, Angew. Chem., Int. Ed., 2012, 51, 4426–4430 CrossRef CAS PubMed.
- S. J. Edwards, H. Valkenier, N. Busschaert, P. A. Gale and A. P. Davis, Angew. Chem., Int. Ed., 2015, 54, 4592–4596 CrossRef CAS PubMed.
- N. Busschaert, R. B. P. Elmes, D. D. Czech, X. Wu, I. L. Kirby, E. M. Peck, K. D. Hendzel, S. K. Shaw, B. Chan, B. D. Smith, K. A. Jolliffe and P. A. Gale, Chem. Sci., 2014, 5, 3617–3626 RSC.
- X. Ni, X. Li, Z. Wang and J.-P. Cheng, Org. Lett., 2014, 16, 1786–1789 CrossRef CAS PubMed.
- R. B. P. Elmes, P. Turner and K. A. Jolliffe, Org. Lett., 2013, 15, 5638–5641 CrossRef CAS PubMed.
- J. Bergman, B. Pettersson, V. Hasimbegovic and P. H. Svensson, J. Org. Chem., 2011, 76, 1546–1553 CrossRef CAS PubMed.
- M. B. Onaran, A. B. Comeau and C. T. Seto, J. Org. Chem., 2005, 70, 10792–10802 CrossRef CAS PubMed.
- P. Gans, A. Sabatini and A. Vacca, Talanta, 1996, 43, 1739–1753 CrossRef CAS.
- B. D. Smith and T. N. Lambert, Chem. Commun., 2003, 2261–2268 RSC.
- A. V. Hill, Biochem. J., 1913, 7, 471–480 CAS.
- E. Marchal, S. Rastogi, A. Thompson and J. T. Davis, Org. Biomol. Chem., 2014, 12, 7515–7522 CAS.
- N. Busschaert, S. J. Bradberry, M. Wenzel, C. J. E. Haynes, J. R. Hiscock, I. L. Kirby, L. E. Karagiannidis, S. J. Moore, N. J. Wells, J. Herniman, G. J. Langley, P. N. Horton, M. E. Light, I. Marques, P. J. Costa, V. Félix, J. G. Frey and P. A. Gale, Chem. Sci., 2013, 4, 3036–3045 RSC.
- H. Valkenier, C. J. E. Haynes, J. Herniman, P. A. Gale and A. P. Davis, Chem. Sci., 2014, 5, 1128–1134 RSC.
- N. Busschaert, M. Wenzel, M. E. Light, P. Iglesias-Hernández, R. Pérez-Tomás and P. A. Gale, J. Am. Chem. Soc., 2011, 133, 14136–14148 CrossRef CAS PubMed.
- V. Saggiomo, S. Otto, I. Marques, V. Félix, T. Torroba and R. Quesada, Chem. Commun., 2012, 48, 5274–5276 RSC.
- V. Sidorov, F. W. Kotch, G. Abdrakhmanova, R. Mizani, J. C. Fettinger and J. T. Davis, J. Am. Chem. Soc., 2002, 124, 2267–2278 CrossRef CAS PubMed.
- Y. Marcus, J. Chem. Soc., Faraday Trans., 1991, 87, 2995–2999 RSC.
Footnotes |
| † Electronic supplementary information (ESI) available: Synthesis and characterisation of the receptors, experimental details and figures relating to stability, association constant determination, stack plots and fitplots of the 1H NMR titrations, pH-spectrophotometric titrations, various vesicle assays methods and Hill plots. The data underlying this communication is available online at http://dx.doi.org/10.5258/SOTON/376697. See DOI: 10.1039/c5cc03625a |
| ‡ These authors contributed equally to this work. |
| § Current address: Department of Chemistry, Maynooth University, National University of Ireland, Maynooth, Co. Kildare, Ireland. |
| ¶ Current address: Department of Chemistry, Chemistry Research Laboratory, University of Oxford, 12 Mansfield Road, Oxford, OX1 3TA, UK. |
| This journal is © The Royal Society of Chemistry 2015 |