Asymmetric Kita spirolactonisation catalysed by anti-dimethanoanthracene-based iodoarenes†
Abstract
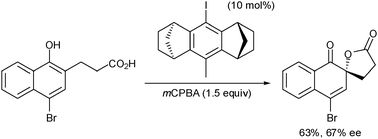
Enantiopure C2-symmetric iodoarenes based on the rigid all-carbon anti-dimethanoanthracene framework are shown to catalyse the asymmetric oxidative Kita spirolactonisation of propanoic acid-tethered 1-naphthols with significant levels of asymmetric induction of up to 67% ee.

 Please wait while we load your content...
Please wait while we load your content...