Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
One-step synthesis of patterned polymer brushes by photocatalytic microcontact printing†
Friederike
Kettling
a,
Benjamin
Vonhören
a,
Jennifer A.
Krings
a,
Susumu
Saito
b and
Bart Jan
Ravoo
*a
aOrganic Chemistry Institute, Westfälische Wilhelms-Universität Münster, Corrensstrasse 40, 48149 Münster, Germany. E-mail: b.j.ravoo@uni-muenster.de; Fax: +49 251 83 36557; Tel: +49 251 83 33287
bGraduate School of Science and Institute for Advanced Research, Nagoya University, Chikusa, Nagoya 464-8602, Japan. E-mail: saito.susumu@f.mbox.nagoya-u.ac.jp; Fax: +81-52-789-5945; Tel: +81-52-789-5945
First published on 24th November 2014
Abstract
A novel method to prepare microstructured polymer brushes using TiO2 nanoparticles and photocatalytic microcontact printing is described. It is shown that ethanol amine can be polymerized to linear polyethyleneimine (PEI) driven by the photocatalytic action of TiO2. Upon UV irradiation during microcontact printing of ethanol amine with a stamp coated with TiO2 nanoparticles, patterned polymer brushes with a length of around 50 nm are obtained.
Microcontact printing (μCP) is a well-known method to produce structured surfaces in a simple, rapid and cost efficient way.1–3 Although μCP typically relies on chemisorption of ink molecules to a substrate, it has recently been shown that catalysis can also play an important role in surface patterning. An early example of catalytic μCP is the use of the acidic nature of an oxidized polydimethylsiloxane (PDMS) stamp to deprotect acid labile silylether-terminated self-assembled monolayers (SAMs) to produce patterned alcohol surfaces on gold.4 Similarly, PDMS stamps can be coated with sulfonic acid functionalized gold nanoparticles (NPs) to pattern an acid labile SAM on gold5 or functionalized covalently with sulfonic acids to pattern N-hydroxysuccinimide-terminated SAMs on germanium surfaces by deprotection.6 In addition it has been shown that enzyme functionalized stamps can be used for biocatalytic μCP.7 Furthermore, heterogeneous catalysis with metal coated stamps enabled micropatterned Heck reactions8 as well as CuI catalyzed azide–alkyne click chemistry9 by μCP.
To the best of our knowledge, heterogeneous catalysis combined with μCP has never been used to prepare patterned polymer brushes in one step. Polymer brushes are an important subject of research since many years10 and their remarkable properties on various surfaces leads to a range of applications. Polymer brushes are used to stabilize colloids11 and switch the character of a surface from hydrophobic to hydrophilic.12 It is possible to control cell adhesion on surfaces treated with polymer brushes.13 Polymer brushes can be synthesized using a “grafting to” or a “grafting from” approach. Also the patterning of polymer brushes on surfaces is of great interest for various applications. Typically, the patterned “grafting-from” approach makes use of some lithography method, e.g. micromolding,14 photolithography,15 electron beam lithography16 or μCP17 to pattern an initiator onto the surface. Afterwards a surface initiated polymerization is used to build up polymer brushes from solution. The new method presented here requires only a single step for patterning and grafting (Scheme 1). In order to grow polymer brushes of linear polyethyleneimine (PEI) from ethanol amine, the photocatalytic properties of TiO2 NPs are exploited. Previously, TiO2 was used as photoinitiator for surface initiated Cu-mediated ATRP to prepare polymer brushes and hydrogels.18 In spite of the large body of literature on photocatalysis by TiO2 NPs, the photocatalytic step-growth of polymer brushes is unprecedented.
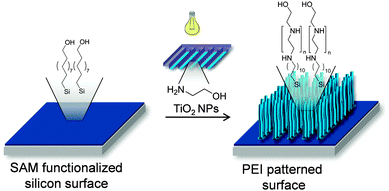 | ||
| Scheme 1 Photocatalytic polymerization by printing of ethanol amine with TiO2 NPs on a 11-(trichlorosilyl)undecan-1-ol SAM. | ||
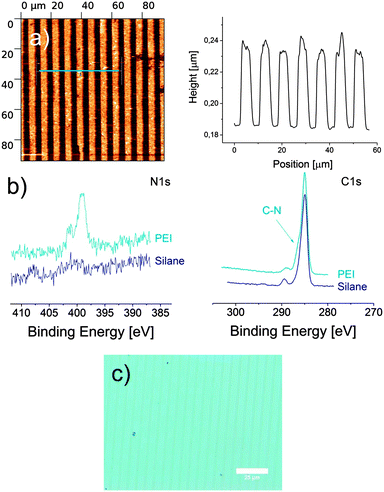
TiO2 NPs were prepared via sol–gel synthesis from TiCl4 and water in diethyleneglycol and have an average diameter of around 5 nm according to dynamic light scattering (see ESI†). μCP was performed with a striped PDMS stamp (preparation see ESI†) which was incubated with 30 μL of a methanolic solution of TiO2 NPs and ethanol amine. After 1 min the stamp was dried in a stream of argon and placed on a silicon (or glass) surface, which was functionalized with a SAM of 11-(trichlorosilyl)undecan-1-ol (for preparation see ESI†). The transparent stamp and surface were irradiated with UV light for 1 to 45 min.19 Afterwards the stamp was removed and the surface was washed and sonicated in dichloromethane, ethanol and milliQ water for 5 min each to remove physisorbed material, in particular residual TiO2 NPs. AFM measurements (Fig. 1a) show microstructured brushes with an average height of around 58 nm for an irradiation time of 20 min. As shown in Fig. S1 in the ESI† the length of the polymer brushes tends to increase with the printing time although no strict linear correlation can be observed.
If μCP was performed without TiO2 NPs, no structure was observed on the surface at all. Even optical microscopy images (Fig. 1c) clearly show the formation of a microstructure on the surface. Fig. 1b compares the X-ray photoelectron spectroscopy (XPS) analysis of the surface before (silane SAM) and after (PEI brushes) μCP with a flat PDMS stamp and confirms the immobilization of nitrogen in the polymer. The C1s signal is also present before the printing process due to the alkyl chain of the SAM, but a detailed view reveals a shoulder in the C1s signal of the polymer at around 286 eV. This signal is known for the C–N bond of PEI and indicates the presence of PEI on the surface.20 The XPS spectrum after printing shows also some remaining Ti on the surface, which might be due to some TiO2 particles that could not be washed from the surface and were embedded in the polymer brushes. The proposed mechanism of the photocatalytic polymerization can be found in Scheme 2. We suggest that under UV irradiation the holes (h+) in TiO2 oxidize compound 1 (n = 0; R = Si(CH2)9) as well as ethanol amine to the corresponding aldehyde 2 (step I). This aldehyde can react with ethanol amine to imine 3 (step II), which can in turn be reduced by the electrons generated from TiO2 to the secondary amine 4 (step III) and finally re-enter the cycle with the unaffected alcohol function as 1 (step IV).
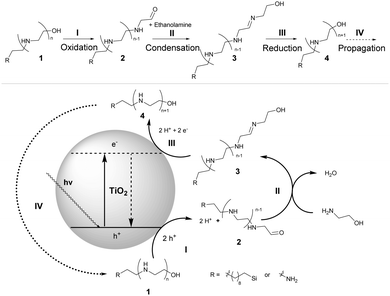 | ||
| Scheme 2 Proposed reaction mechanism of the photocatalytic step-growth polymerization of ethanol amine. | ||
To verify whether the immobilized polymer consists of secondary amines or imines, the following experiments were performed. If the imine was not reduced by protons and electrons generated in the TiO2 NPs it should be possible to hydrolyze the polymer under acidic conditions, yet stirring and even boiling in acetic acid at pH 3 (60 mM in water) did not destroy neither the structure nor reduce the height of the brushes on the surface.
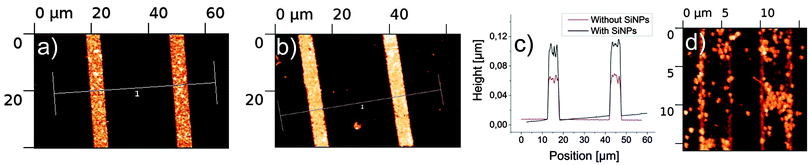
Moreover the cationic character of the polymer brushes could be confirmed. To this end, immobilization tests with negatively charged SiO2 nanoparticles (SNPs) with a size of around 60 nm were performed.21 If indeed the polymers are (protonated) amines, not imines, the SNPs will bind strongly to the stripes and can be detected by AFM. In Fig. 2 the AFM measurements of the patterned surfaces before and after incubation with SNPs can be found. Fig. 2a shows the bare polymer brushes before binding at pH 7. Fig. 2d displays the binding behaviour of the SNPs after the standard μCP process at pH 7. As mentioned above the polymer brushes were washed with water. Because PEI is a weak base, around 15–20%22 of the secondary amine groups will be protonated at pH 7. This leads to positive charges in the polymer brushes, which attract the negatively charged SNPs. If the polymer brushes are sonicated in HCl at pH 3 for 15 min, the amount of protonated amines rises to 70–80%23 and, as can be seen in Fig. 2b, the aggregation of SNPs increases as well. Comparison of the height profiles of Fig. 2a and b clearly demonstrates the attachment of the SNPs (Fig. 2c). The difference between the calculated height (length of polymer plus size of particles) of around 110 nm and the measured one around 90 nm might be due to the polymer brushes being slightly compacted upon binding the SNPs.
Finally, to demonstrate that this photocatalytic μCP is a versatile method to modify substrates that display free alcohol functions, the polymerization of ethanol amine was also performed on cellulose surfaces. These were fabricated by spin coating of a solution of cellulose in dimethylacetamide with LiCl on a clean silicon wafer.23,24 After washing and drying a layer thickness of around 100 nm was observed by profilometry (Fig. S2, ESI†). μCP was performed as described above with a printing time of 30 min, but the cleaning process was reduced to washing with dichloromethane to protect the cellulose coating. In this experiment a dotted stamp (3 μm spots spaced by 5 μm) was used. Fig. 3 displays formation of polymer brushes on the cellulose surface. The profiles in Fig. 3c indicate that also on cellulose polymers with length of 40 nm can be achieved. XPS data confirm the successful immobilization (Fig. S3, ESI†). The N1s signal clearly indicates the immobilization of PEI on cellulose while the C1s signal confirms the presence of C–N bonds overlaying the C–O bond of the cellulose.
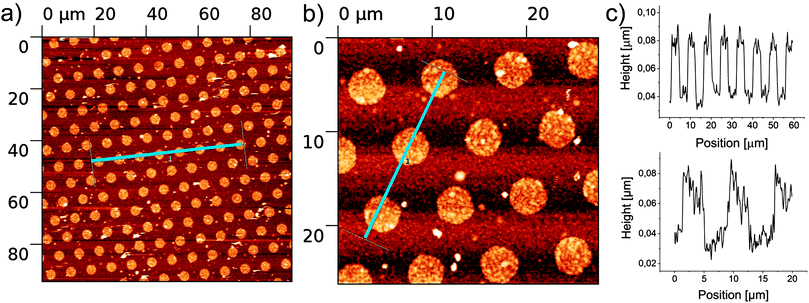 | ||
| Fig. 3 AFM measurement of ethanol amine polymerized (30 min) onto cellulose; (a) overview; (b) zoom in; (c) profile of blue traces in (a) and (b). | ||
In this study the photocatalytic polymerization of ethanol amine by μCP with TiO2 NPs was confirmed by microscopy, AFM and XPS. Using TiO2 NPs it is possible to polymerize ethanol amine on a surface to polyethyleneimine brushes with a length around 50 nm. The use of μCP leads to a well-defined pattern of the polymer brushes. To underline the versatility of this reaction the brushes were printed on SAMs of 11-(trichlorosilyl)undecan-1-ol and also on spin-coated cellulose surfaces, which raises the prospect of micropatterning of PEI on paper. Further investigations regarding the photocatalytic PEI synthesis in solution are ongoing.
We thank the DFG (IRTG 1143 Münster-Nagoya) and the Sustainable Chemical Systems (SusChemSys) program cofinanced by the European Regional Development Fund (ERDF) and the state of North Rhine-Westphalia, Germany, under the Operational Programme “Regional Competitiveness and Employment” (2007–2013) for funding.
Notes and references
- Y. Xia and G. M. Whitesides, Angew. Chem., Int. Ed., 1998, 37, 550–575 CrossRef CAS.
- C. Wendeln and B. J. Ravoo, Langmuir, 2012, 28, 5527–5538 CrossRef CAS PubMed.
- A. Perl, D. N. Reinhoudt and J. Huskens, Adv. Mater., 2009, 21, 2257–2268 CrossRef CAS.
- X.-M. Li, M. Péter, J. Huskens and D. N. Reinhoudt, Nano Lett., 2003, 3, 1449–1453 CrossRef CAS.
- X.-M. Li, V. Paraschiv, J. Huskens and D. N. Reinhoudt, J. Am. Chem. Soc., 2003, 125, 4279–4284 CrossRef CAS PubMed.
- C. J. Morris, A. A. Shestopalov, B. H. Gold, R. L. Clark and E. J. Toone, Langmuir, 2011, 27, 6486–6489 CrossRef CAS PubMed.
- P. W. Snyder, M. S. Johannes, B. N. Vogen, R. L. Clark and E. J. Toone, J. Org. Chem., 2007, 72, 7459–7461 CrossRef CAS PubMed.
- H. Mizuno and J. M. Buriak, ACS Appl. Mater. Interfaces, 2010, 2, 2301–2307 CAS.
- J. M. Spruell, B. A. Sheriff, D. I. Rozkiewicz, W. R. Dichtel, R. D. Rohde, D. N. Reinhoudt, J. F. Stoddart and J. R. Heath, Angew. Chem., Int. Ed., 2008, 47, 9927–9932 CrossRef CAS PubMed.
- S. T. Milner, Science, 1991, 251, 905–914 CrossRef CAS PubMed.
- D. Maillard, S. K. Kumar, A. Rungta, B. C. Benicewicz and R. E. Prud'homme, Nano Lett., 2011, 11, 4569–4573 CrossRef CAS PubMed.
- A. Sidorenko, S. Minko, K. Schenk-Meuser, H. Duschner and M. Stamm, Langmuir, 1999, 15, 8349–8355 CrossRef CAS.
- (a) O. Hyeong Kwon, A. Kikuchi, M. Yamato and T. Okano, Biomaterials, 2003, 24, 1223–1232 CrossRef CAS; (b) C. Rodriguez-Emmenegger, C. M. Preuss, B. Yameen, O. Pop-Georgievski, M. Bachmann, J. O. Mueller, M. Bruns, A. S. Goldmann, M. Bastmeyer and C. Barner-Kowollik, Adv. Mater., 2013, 25, 6123–6127 CrossRef CAS PubMed.
- C. A. Escobar, T. J. Cooksey, M. P. Spellings and G. K. Jennings, Adv. Mater. Interfaces, 2014 DOI:10.1002/admi.201400055.
- O. Prucker, M. Schimmel, G. Tovar, W. Knoll and J. Rühe, Adv. Mater., 1998, 10, 1073–1077 CrossRef CAS.
- S. J. Ahn, M. Kaholek, W.-K. Lee, B. LaMattina, T. H. LaBean and S. Zauscher, Adv. Mater., 2004, 16, 2141–2145 CrossRef CAS.
- (a) P. Xiao, J. Gu, J. Chen, D. Han, J. Zhang, H. Cao, R. Xing, Y. Han, W. Wang and T. Chen, Chem. Commun., 2013, 49, 11167–11169 RSC; (b) O. Roling, A. Mardyukov, S. Lamping, B. Vonhören, S. Rinnen, H. F. Arlinghaus, A. Studer and B. J. Ravoo, Org. Biomol. Chem., 2014, 12, 7828–7835 RSC.
- (a) B. Li, B. Yu and F. Zhou, Macromol. Rapid Commun., 2014, 35, 1287–1292 CrossRef CAS PubMed; (b) J. Yan, B. Li, F. Zhou and W. Liu, ACS Macro Lett., 2013, 2, 592–596 CrossRef CAS; (c) D. Zhang, J. Yang, S. Bao, Q. Wu and Q. Wang, Sci. Rep., 2013, 3, 1399 Search PubMed.
- C. Wendeln, S. Rinnen, C. Schulz, H. F. Arlinghaus and B. J. Ravoo, Langmuir, 2010, 26, 15966–15971 CrossRef CAS PubMed.
- E. P. Dillon, C. A. Crouse and A. R. Barron, ACS Nano, 2008, 2, 156–164 CrossRef CAS PubMed.
- L. M. Rossi, L. Shi, F. H. Quina and Z. Rosenzweig, Langmuir, 2005, 21, 4277–4280 CrossRef CAS.
- J. Suh, H. J. Paik and B. K. Hwang, Bioorg. Chem., 1994, 22, 318–327 CrossRef CAS.
- K. Han, K.-S. Kang and J. Kim, J. Micromech. Microeng., 2009, 19, 035010 CrossRef.
- K. Rahn, M. Diamantoglou, D. Klemm, H. Berghmans and T. Heinze, Angew. Makromol. Chem., 1996, 238, 143–163 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Materials, methods and additional experiments and analysis. See DOI: 10.1039/c4cc08646e |
| This journal is © The Royal Society of Chemistry 2015 |