Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Ultralow-intensity near-infrared light induces drug delivery by upconverting nanoparticles†
Shuqing
He
a,
Kristina
Krippes
a,
Sandra
Ritz
a,
Zhijun
Chen
a,
Andreas
Best
a,
Hans-Jürgen
Butt
a,
Volker
Mailänder
ab and
Si
Wu
*a
aMax Planck Institute for Polymer Research, Ackermannweg 10, 55128 Mainz, Germany. E-mail: wusi@mpip-mainz.mpg.de
bIII. Medical Clinic, University Medicine of the Johannes-Gutenberg University Mainz, Langenbeckstr. 1, 55131 Mainz, Germany
First published on 11th November 2014
Abstract
Mesoporous silica coated upconverting nanoparticles are loaded with the anticancer drug doxorubicin and grafted with ruthenium complexes as photoactive molecular valves. Drug release was triggered by 974 nm light with 0.35 W cm−2. Such low light intensity minimized overheating problems and prevented photodamage to biological samples.
Light has been used for therapy for thousands of years. In modern nanomedicine, light is used to trigger drug release from photosensitive nanocarriers.1 For most photosensitive nanocarriers, photoreactions are induced by UV or visible light.1 Compared to UV and visible light, near-infrared (NIR) light is more suitable for biomedical applications since NIR light is able to penetrate deeper into tissues and causes less damage. One approach to achieve NIR-triggered drug release is to use simultaneous two-photon absorption to induce photoreactions in nanocarriers. However, this approach is inefficient even when high-intensity femtosecond lasers (pulse intensity >106 W cm−2)2 are used because of the low two-photon absorption cross sections of typical chromophores.
An alternative approach to achieve NIR light-triggered drug release is based on lanthanide-doped upconverting nanoparticles (UCNPs).3 UCNPs convert NIR light (∼980 nm) to UV and visible light that can subsequently trigger useful photoreactions (Fig. 1a). This process is called UCNP-assisted photochemistry (Fig. 1a).3d Compared to simultaneous two-photon absorption, UCNPs can be excited by continuous-wave NIR laser diodes with relatively low intensity. There are two requirements for UCNP-assisted photochemistry. First, upconversion requires an excitation intensity which exceeds a certain threshold.4 Second, a suitable photosensitive compound absorbs upconverted light. The reported excitation intensity for UCNP-assisted photochemistry is usually several to several hundred W cm−2 (Table S1, ESI†).3b,c,e,5 However, according to the American national standard for safe use of lasers, NIR lasers with the intensity of several W cm−2 are dangerous.6 Under continuous exposure to 980 nm light, the maximum permissible exposure of skin is 0.726 W cm−2.6 High-intensity NIR light can cause overheating problems and photodamage to biological samples. For example, the overheating effect induced by 980 nm light at 6 W cm−2 can cause cell death within 5 min.7 The required high excitation intensity of UCNP-assisted photochemistry hinders its biomedical applications. To prevent damage to biological samples due to the NIR laser, it is highly desirable to reduce the light intensity for UCNP-assisted photochemistry.
Herein, we demonstrate a strategy to reduce the NIR light intensity for UCNP-assisted photochemistry to a medically harmless dose. We show that 974 nm light with 0.35 W cm−2 is able to trigger drug release based on UCNP-assisted photochemistry. To the best of our knowledge, 0.35 W cm−2 is the lowest reported intensity for UCNP-assisted photochemistry (Table S1, ESI†). This light intensity is also lower than the maximum permissible exposure of skin (0.726 W cm−2). Overheating problems are minimized and photodamage to biological samples is prevented at such a low light intensity. We fabricated a drug delivery system by loading of mesoporous silica coated UCNPs with doxorubicin. As photoactive molecular valves, blue-light-cleavable ruthenium (Ru) complexes are grafted to the surface of the nanoparticles. The required NIR intensity was that low because a three-photon process is sufficient to create the required blue photons and to induce the photocleavage of Ru complexes. Usually, a four-photon NIR-to-UV upconversion is applied to induce photoreactions of UV-sensitive compounds.
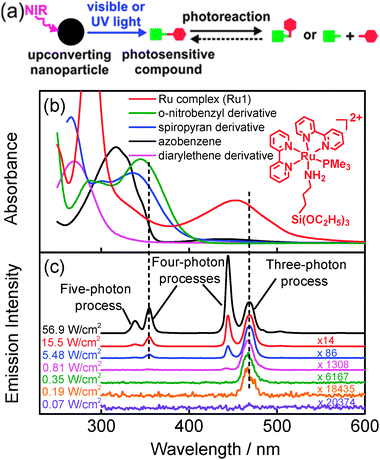
The concept in UCNP-assisted photochemistry entails that a photosensitive compound absorbs the upconverted light. Therefore, the absorption of photosensitive compounds should overlap with the emission of UCNPs (Fig. 1a). We studied absorption of five commonly used photosensitive compounds (Fig. 1b); their chemical structures are shown in Fig. S1 (ESI†). The metal-to-ligand charge transfer band of the Ru complex (Ru1) is at ∼453 nm (Fig. 1b). Photocleavage of Ru1 can be induced by blue light (Fig. S12, ESI†).8 Like most reported photosensitive compounds,1 the absorption bands of the other four compounds are in the UV region. Their photoreactions are induced by UV light.
We synthesized β-phase NaYF4:TmYb@NaYF4 UCNPs (core = NaYF4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 mol% Tm3+
0.5 mol% Tm3+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 mol% Yb3+; shell = NaYF4) (Fig. S2–S5, ESI†), which are some of the most efficient UCNPs known to date.9 At high excitation intensities (≥5.48 W cm−2) with a 974 nm laser, the UCNPs emit both blue and UV light (Fig. 1c). At low excitation intensities (0.19–0.81 W cm−2), only blue light is emitted (Fig. 1c). The relative intensity of the emission depends on the excitation intensity because the photon numbers for excitation of different emissions vary.10 The excitation thresholds for 1I6–3F4 transition (340 nm, a five-photon process10), 1D2–3H6 transition (360 nm, a four-photon process10), and 1G4–3H6 transition (470 nm, a three-photon process10) are 5.48, 2.22, and 0.19 W cm−2, respectively (Fig. 1c and Fig. S6, ESI†). Thus, blue emission at 470 nm still retains and UV emission completely vanishes at excitation intensities below 2.22 W cm−2 and above 0.19 W cm−2. Out of the five photosensitive compounds, the upconverted blue light at 470 nm overlaps with the absorption band of Ru1 only (Fig. 1b and c). Therefore, it is only possible to excite the photoreaction of Ru1 at low excitation intensity e.g. 0.35 W cm−2.
30 mol% Yb3+; shell = NaYF4) (Fig. S2–S5, ESI†), which are some of the most efficient UCNPs known to date.9 At high excitation intensities (≥5.48 W cm−2) with a 974 nm laser, the UCNPs emit both blue and UV light (Fig. 1c). At low excitation intensities (0.19–0.81 W cm−2), only blue light is emitted (Fig. 1c). The relative intensity of the emission depends on the excitation intensity because the photon numbers for excitation of different emissions vary.10 The excitation thresholds for 1I6–3F4 transition (340 nm, a five-photon process10), 1D2–3H6 transition (360 nm, a four-photon process10), and 1G4–3H6 transition (470 nm, a three-photon process10) are 5.48, 2.22, and 0.19 W cm−2, respectively (Fig. 1c and Fig. S6, ESI†). Thus, blue emission at 470 nm still retains and UV emission completely vanishes at excitation intensities below 2.22 W cm−2 and above 0.19 W cm−2. Out of the five photosensitive compounds, the upconverted blue light at 470 nm overlaps with the absorption band of Ru1 only (Fig. 1b and c). Therefore, it is only possible to excite the photoreaction of Ru1 at low excitation intensity e.g. 0.35 W cm−2.
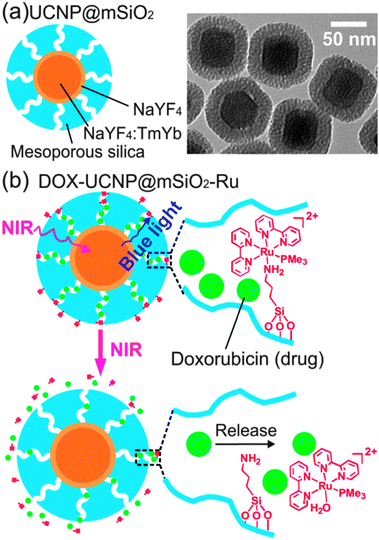
Based on our hypothesis, we prepared a NIR light-triggered drug delivery system by grafting mesoporous silica coated UCNPs with Ru1 (Fig. 2). UCNPs were coated with mesoporous silica (UCNP@mSiO2) by a sol–gel reaction.11 The mesoporous silica is the drug carrier.11 The grafted Ru complexes act as molecular valves.12 The average diameter of UCNP@mSiO2 measured by TEM and dynamic light scattering was 92 nm and 89 nm, respectively (Fig. 2a and Fig. S2 and S3, ESI†). The average thickness of the mesoporous silica shell was 21 nm (Fig. S2, ESI†). Powder X-ray diffraction confirmed that the UCNPs are hexagonal in phase (Fig. S4, ESI†). The average pore size and the Brunauer–Emmett–Teller (BET) surface area of UCNP@mSiO2 were 2.6 nm and 316 m2 g−1, respectively (Fig. S7, ESI†). UCNP@mSiO2 nanoparticles were loaded with the anticancer drug doxorubicin and grafted with Ru1 to form DOX-UCNP@mSiO2-Ru (Fig. 2b). The drug loading efficiency, measured by fluorescence spectroscopy and UV/Vis absorption spectroscopy, was 2.36% and 2.52% (23.6 and 25.2 μg doxorubicin in 1 mg nanoparticles), respectively (Fig. S17, ESI†). The successful grafting of UCNP@mSiO2 with Ru1 was confirmed by 29Si MAS solid-state NMR spectroscopy, UV/Vis absorption spectroscopy and FTIR spectroscopy (Fig. S8–S10, ESI†). UV/Vis absorption spectroscopy shows that 7.6 μg Ru1 was grafted on 1 mg of nanoparticles (Fig. S11, ESI†).
To demonstrate absorption of upconverted blue light by the Ru complex, we compared upconversion luminescence spectra of UCNP@mSiO2 to UCNP@mSiO2-Ru (Ru1 grafted UCNP@mSiO2) nanoparticles. The intensity of upconversion luminescence at 470 nm decreased significantly in the spectrum of UCNP@mSiO2-Ru (Fig. 3a). In contrast, the emission at ∼800 nm, a spectral region where the Ru complex has no absorption, still remained. This result proved efficient absorption of the upconverted blue light by the Ru complex. UV/Vis absorption spectroscopy further confirmed that upconverted blue light can trigger cleavage of the Ru complex (Fig. 3b). 974 nm light induced a slight redshift of the absorption band of the Ru complex on UCNP@mSiO2-Ru (Fig. 3b). This spectral change is identical to observations for solutions of Ru1 or similar Ru complexes which were exposed to blue light to directly trigger photocleavage (Fig. S12, ESI†).8b,c Exposure of Ru1 to 974 nm light in the absence of UCNPs had no influence on the absorption spectrum (Fig. S13, ESI†), thus demonstrating that the photocleavage of the Ru complex is triggered by upconversion. Measurements by UV/Vis absorption spectroscopy showed that 974 nm light (0.35 W cm−2) cleaved 59% Ru complexes from UCNP@mSiO2-Ru after 5 hour irradiation (Fig. S14, ESI†).
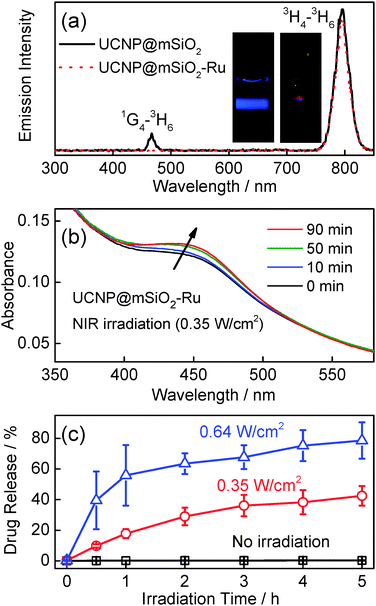 | ||
| Fig. 3 (a) Emission spectra (λex = 974 nm, 0.35 W cm−2) of UCNP@mSiO2 and Ru1 grafted UCNP@mSiO2 (UCNP@mSiO2-Ru) nanoparticles. Inset: photograph of UCNP@mSiO2 (left) and UCNP@mSiO2-Ru (right) nanoparticles upon a 974 nm laser exposure. The concentration of Ru complexes in UCNP@mSiO2-Ru was higher than that in DOX-UCNP@mSiO2-Ru. (b) UV/Vis absorption spectra of UCNP@mSiO2-Ru upon 974 nm light exposure (0.35 W cm−2). (c) Doxorubicin release profile for PEG- and FA-modified DOX-UCNP@mSiO2-Ru nanoparticles in the dark and upon 974 nm light exposure. The release profile was measured by fluorescence spectroscopy (Fig. S19, ESI†). | ||
NIR light-triggered drug release from doxorubicin loaded nanoparticles DOX-UCNP@mSiO2-Ru was studied by “mini dialysis” (Fig. S19, ESI†). Release of doxorubicin was not detected without NIR irradiation. Approximately 42% of doxorubicin was released after 974 nm light irradiation with 0.35 W cm−2 for 5 hours (Fig. 3c). The release rate increased and ∼78% of doxorubicin was released after 974 nm light irradiation with 0.64 W cm−2 for 5 hours.
As a control experiment, UCNP@mSiO2 nanoparticles are grafted with a UV-sensitive azobenzene compound instead of the Ru complex (Fig. S30, ESI†). The control experiment showed that no drug release from the azobenzene-grafted nanoparticles could be induced by 974 nm light with 0.35 W cm−2. Approximately 27% of doxorubicin was released after 974 nm light irradiation with 7 W cm−2 for 5 hours (Fig. S30, ESI†). This result shows that the required light intensity for drug release in DOX-UCNP@mSiO2-Ru is much lower. Thus, combining blue-light sensitive Ru complexes with UCNPs could efficiently reduce the excitation intensity for UCNP-assisted photochemistry.
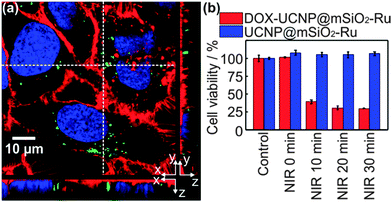
Encouraged by the successful drug release, we investigated NIR-induced drug delivery in cancer cells. For all drug release and cell culture experiments, nanoparticles were modified with poly(ethylene glycol) (PEG) and folic acid (FA) to enhance uptake by HeLa cells.13 TEM analyses demonstrated retention of nanoparticle morphology after surface modification (Fig. S15, ESI†). In cellular studies, HeLa cells were incubated with fluorescence-labeled UCNP@mSiO2-Ru nanoparticles for 3–6 hours before irradiation. Confocal laser scanning microscopy confirmed uptake of nanoparticles by HeLa cells (Fig. 4a and Fig. S21, ESI†). Incubation of HeLa cells with UCNP@mSiO2, UCNP@mSiO2-Ru or the doxorubicin loaded nanoparticles (DOX-UCNP@mSiO2-Ru) in the dark did not cause a decrease of cell viability, hereby excluding leakage or non-incorporated doxorubicin (Fig. S22, ESI†). This result is in accordance with the extracellular drug release profile which states that nearly no drugs can be released from the nanoparticles without irradiation (Fig. 3c).
Subsequently, we exposed HeLa cells, incubated for 3–6 h with DOX-UCNP@mSiO2-Ru, to 974 nm light with 0.35 mW cm−2. Afterwards, the cells were incubated further for 24 hours. NIR irradiation of DOX-UCNP@mSiO2-Ru significantly inhibited the growth of cancer cells (Fig. 4b). Irradiation for 10–30 min reduced cell viability to a value of 40–29%. For comparison, control experiments under identical conditions using UCNP@mSiO2-Ru, UCNP@mSiO2 or no nanoparticles instead of DOX-UCNP@mSiO2-Ru were extensively performed (Fig. 4b and Fig. S23 and S24, ESI†): NIR light irradiation did not cause significant decrease of cell viability for these control samples demonstrating that cytotoxicity to cancer cells (Fig. 4b) was caused by release of doxorubicin.
We also studied the photothermal effect of 974 nm light by exposure of water to the laser. The water temperature increased only 2.5 °C upon irradiation of 974 nm light under 0.35 W cm−2. In contrast, irradiation of 974 nm light under 2 W cm−2 causes a temperature increase of 14 °C (Fig. S20, ESI†). This result demonstrates that low NIR intensity can minimize overheating problems caused by NIR light.
We tested the amount of photodamage of pork tissue under 974 nm light irradiation for 20 min (Fig. S27, ESI†). Serious burn wounds were observed when the light intensity is higher than 3 W cm−2. When the light intensity is lower than 1 W cm−2, no obvious burn wound was observed. Therefore, using low-intensity NIR light is important to prevent tissue damages.
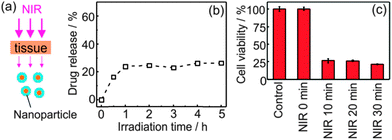
We verified if low-intensity 974 nm light could induce drug delivery after passing through tissue. We placed a pork tissue between the laser and the nanoparticles. Approximately 26% of doxorubicin was released after 974 nm light irradiation with 0.64 W cm−2 for 5 hours (Fig. 5b). The released doxorubicin also inhibited the growth of cancer cells (Fig. 5c). The used light intensity (0.64 W cm−2) is also lower than the maximum permissible exposure of skin (0.726 W cm−2). Thus, low-intensity NIR light could be used to trigger drug delivery and minimize tissue damage in our system.
In conclusion, we demonstrate a strategy to reduce excitation intensity for UCNP-assisted photochemistry. The reported concept for constructing photoresponsive systems that are sensitive to ultralow-intensity NIR light is novel and should be generally applicable to various biomedical applications. We apply this novel concept for low-intensity NIR light-triggered drug delivery. Ru complexes on UCNP@mSiO2-Ru nanoparticles are cleaved by 974 nm light with intensity as low as 0.35 W cm−2. Comparing UCNP@mSiO2-Ru to other recently developed systems based on UCNP-assisted photochemistry shows that 0.35 W cm−2 is the lowest intensity reported to the best of our knowledge (Table S1, ESI†). In particular, the intensity is lower than the maximum permissible exposure of skin (0.726 W cm−2). We expect the suitability of not only Ru complexes but also other blue-light-sensitive compounds for UCNP-assisted photochemistry with low excitation intensity.14 Low excitation intensity in UCNP-assisted photochemistry minimizes the overheating effect and causes less photodamage to biological samples, both of which are important for biomedical applications.
This work is supported by the Deutsche Forschungsgemeinschaft (DFG, SPP1313 program MA 3271/3-1 and SFB1066). S.H. was supported by the joint program of MPG and CAS. V. M. and S. W. are the senior authors for the biology and chemistry parts, respectively. We thank H. Menges for upconverting luminescence spectra, R. Graf for NMR spectra, and G. Waschatko for preparing tissue.
Notes and references
- R. Tong and D. S. Kohane, Wiley Interdiscip. Rev.: Nanomed. Nanobiotechnol., 2012, 4, 638 CrossRef CAS PubMed.
- M. Álvarez, A. Best, S. Pradhan-Kadam, K. Koynov, U. Jonas and M. Kreiter, Adv. Mater., 2008, 20, 4563 CrossRef.
- (a) B. Yan, J. C. Boyer, N. R. Branda and Y. Zhao, J. Am. Chem. Soc., 2011, 133, 19714 CrossRef CAS PubMed; (b) J. Liu, W. Bu, L. Pan and J. Shi, Angew. Chem., Int. Ed., 2013, 52, 4375 CrossRef CAS PubMed; (c) Y. Yang, B. Velmurugan, X. Liu and B. Xing, Small, 2013, 9, 2937 CrossRef CAS PubMed; (d) M. L. Viger, M. Grossman, N. Fomina and A. Almutairi, Adv. Mater., 2013, 25, 3733 CrossRef CAS PubMed; (e) Y. Dai, H. Xiao, J. Liu, Q. Yuan, P. Ma, D. Yang, C. Li, Z. Cheng, Z. Hou, P. Yang and J. Lin, J. Am. Chem. Soc., 2013, 135, 18920 CrossRef CAS PubMed.
- F. Auzel, Chem. Rev., 2004, 104, 139 CrossRef CAS PubMed.
- (a) C. J. Carling, J. C. Boyer and N. R. Branda, J. Am. Chem. Soc., 2009, 131, 10838 CrossRef CAS PubMed; (b) J. C. Boyer, C. J. Carling, B. D. Gates and N. R. Branda, J. Am. Chem. Soc., 2010, 132, 15766 CrossRef CAS PubMed; (c) C. J. Carling, F. Nourmohammadian, J. C. Boyer and N. R. Branda, Angew. Chem., Int. Ed., 2010, 49, 3782 CrossRef CAS PubMed; (d) W. Wu, L. M. Yao, T. S. Yang, R. Y. Yin, F. Y. Li and Y. L. Yu, J. Am. Chem. Soc., 2011, 133, 15810 CrossRef CAS PubMed; (e) J. V. Garcia, J. Yang, D. Shen, C. Yao, X. Li, R. Wang, G. D. Stucky, D. Zhao, P. C. Ford and F. Zhang, Small, 2012, 8, 3800 CrossRef CAS PubMed; (f) M. K. G. Jayakumar, N. M. Idris and Y. Zhang, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 8483 CrossRef CAS PubMed; (g) Y. M. Yang, Q. Shao, R. R. Deng, C. Wang, X. Teng, K. Cheng, Z. Cheng, L. Huang, Z. Liu, X. G. Liu and B. G. Xing, Angew. Chem., Int. Ed., 2012, 51, 3125 CrossRef CAS PubMed; (h) J. Shen, G. Chen, T. Y. Ohulchanskyy, S. J. Kesseli, S. Buchholz, Z. Li, P. N. Prasad and G. Han, Small, 2013, 9, 3213 CAS; (i) D. Yang, P. Ma, Z. Hou, Z. Cheng, C. Li and J. Lin, Chem. Soc. Rev., 2014 10.1039/c4cs00155a.
- (a) American National Standard for safe use of lasers, Laser Institute of America, Orlando, FL, 2000 Search PubMed; (b) Laser Safety Handbook, Northwestern University, 2011 Search PubMed.
- X. Xie, N. Gao, R. Deng, Q. Sun, Q. H. Xu and X. Liu, J. Am. Chem. Soc., 2013, 135, 12608 CrossRef CAS PubMed.
- (a) V. S. Miguel, M. Álvarez, O. Filevich, R. Etchenique and A. del Campo, Langmuir, 2012, 28, 1217–1221 CrossRef PubMed; (b) L. Zayat, M. G. Noval, J. Campi, C. I. Calero, D. J. Calvo and R. Etchenique, ChemBioChem, 2007, 8, 2035–2038 CrossRef CAS PubMed; (c) R. E. Goldbach, I. Rodriguez-Garcia, J. H. van Lenthe, M. A. Siegler and S. Bonnet, Chem. – Eur. J., 2011, 17, 9924 CrossRef CAS PubMed.
- F. Wang and X. G. Liu, Chem. Soc. Rev., 2009, 38, 976 RSC.
- (a) G. F. Wang, W. P. Qin, L. L. Wang, G. D. Wei, P. F. Zhu and R. J. Kim, Opt. Express, 2008, 16, 11907 CrossRef CAS; (b) J. F. Suyver, J. Grimm, M. K. van Veen, D. Biner, K. W. Krämer and H. U. Güdel, J. Lumin., 2006, 117, 1 CrossRef CAS PubMed.
- (a) D. Tarn, C. E. Ashley, M. Xue, E. C. Carnes, J. I. Zink and C. J. Brinker, Acc. Chem. Res., 2013, 46, 792 CrossRef CAS PubMed; (b) S. A. Mackowiak, A. Schmidt, V. Weiss, C. Argyo, C. von Schirnding, T. Bein and C. Bräuchle, Nano Lett., 2013, 13, 2576 CrossRef CAS PubMed; (c) S. Angelos, E. Johansson, J. F. Stoddart and J. I. Zink, Adv. Funct. Mater., 2007, 17, 2261 CrossRef CAS; (d) C. Li, D. Yang, P. Ma, Y. Chen, Y. Wu, Z. Hou, Y. Dai, J. Zhao, C. Sui and J. Lin, Small, 2013, 9, 4150 CrossRef CAS PubMed.
- (a) N. Ž. Knežević, B. G. Trewyn and V. S. Y. Lin, Chem. Commun., 2011, 47, 2817 RSC; (b) M. Frasconi, Z. Liu, J. Lei, Y. Wu, E. Strekalova, D. Malin, M. W. Ambrogio, X. Chen, Y. Y. Botros, V. L. Cryns, J.-P. Sauvage and J. F. Stoddart, J. Am. Chem. Soc., 2013, 135, 11603 CrossRef CAS PubMed.
- G. Baier, D. Baumann, J. M. Siebert, A. Musyanovych, V. Mailänder and K. Landfester, Biomacromolecules, 2012, 13, 2704 CrossRef CAS PubMed.
- Z. Shi, P. Peng, D. Strohecker and Y. Liao, J. Am. Chem. Soc., 2011, 133, 14699–14703 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4cc07489k |
| This journal is © The Royal Society of Chemistry 2015 |