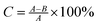The bifunctional regulation of interconnected Zn-incorporated ZrO2 nanoarrays in antibiosis and osteogenesis
Haoyan
Cheng†
a,
Lin
Mao†
b,
Xing
Xu
c,
Yan
Zeng
d,
Danni
Lan
a,
Hao
Hu
a,
Xu
Wu
a,
Huihui
You
b,
Xu
Yang
b,
Rui
Li
*b and
Zhihong
Zhu
*a
aInstitute of Nano-Science and Nano-Technology, College of Physical Science and Technology, Central China Normal University, Wuhan, 430079, China. E-mail: zhzhu@phy.ccnu.edu.cn; Fax: +86-2767861185; Tel: +86-2767861185
bHubei Key Laboratory of Genetic Regulation and Integrative Biology, School of Life Sciences, Central China Normal University, Wuhan, 430079, China. E-mail: ruili@mail.ccnu.edu.cn; Fax: +86-2767861185; Tel: +86-2767861185
cSchool of Life Sciences, Central China Normal University, Wuhan, 430079, China
dCollege of Chemistry and Technology, Central China Normal University, Wuhan, 430079, China
First published on 16th March 2015
Abstract
New generation biomaterials should be designed to bear long-term antibacterial activity, biocompatibility and even osteogenesis facilitation. In this study, the bifunctional regulation of antibiosis and osteogenesis is realized by the highly-ordered and interconnecting Zn-incorporated ZrO2 nanoarrays, which are prepared by a hydrothermal approach with the precursor hydrolyzed in situ and allow long-term controllable Zn release. The content of incorporated Zn can be easily controlled by pH of the HCl solution used for etching. All Zn-incorporated nanoarrays show good antibacterial properties against both Escherichia coli (E. coli, Gram-negative) and Staphylococcus aureus (S. aureus, Gram-positive), as indicated by high antibacterial rates and the apparent inhibition zone. Analysis of the biocompatibility confirms that the hybrid nanoarrays could cause varying degrees of promotion for the adhesion and spreading of MC3T3-E1 cells. Zn incorporated ZrO2 nanotubes balance antibiosis and osteogenesis delicately, as proved by the up-regulated MTT and ALP activities, as well as the increase of bone-related gene expression (the mRNA level of OCN, OPN and BMP-2). The novel bone implant materials with better antibacterial properties can promote the osteogenesis, and have promising applications in biomedical devices and antibacterial control systems.
1. Introduction
Implant materials which possess both the long-term antibacterial ability and excellent biocompatibility need to be tailored due to the fact that the implant-associated infections remain among the most serious clinical complications.1 Generally, E. coli and S. aureus are typically susceptible bacteria because of their biofilm mode in growth during and after implant surgery.2 The biofilm makes the bacteria highly resistant to the host defense and antibacterial agents, thereby leading to persistent and chronic infections.3 Accordingly, improving the antibacterial characteristics on the surface of implants and reducing the probable infections would increase the success rate of bone transplantation surgery.A large number of studies have been done, mainly focused on factors affecting surface bacteria adhesion by modifying the surface with organic antibacterial agents such as hydrogels, chitosan and gentamicin.4–6 However, the fabrication process is usually complex and costly. Specifically, the adhesion between the Ti surface and organic agents with bioactivity is unstable, particularly at high temperatures or pressures. By contrast, inorganic antibacterial materials such as metal oxides are robust and durable. Therefore they have key advantages of stability and longer shelf life.7 ZnO has gained increased interest for its application in biomedical therapy over the last decade. Nanoscaled ZnO in aqueous solution can generate active oxygen radicals (ROS) such as hydroxyl radicals, hydrogen peroxide and superoxide,8,9 which will result in oxidative damage of bacterial cells, making it outstanding for application as a bactericide in daily life. The antimicrobial activity of ZnO nanoparticles has been studied with different pathogenic and nonpathogenic bacteria such as S. aureus and E. coli.10,11 The released zinc ions are also commonly added as an antimicrobial agent to many oral healthcare products for its multiple inhibitory activities in bacteria, such as glycolysis, transmembrane proton translocation and acid tolerance.12 It was documented that Zn-incorporated coatings showed excellent in vitro antibacterial activity against S. aureus and E. coli, and the ability to inhibit bacteria could be improved by increasing the Zn content in the coatings.1 In addition, previous studies have demonstrated that Zn showed strong anti-inflammatory potential by decreasing levels of the pro-inflammatory mediator interleukin-8 and the matrix metalloproteinase-9.13
Besides its outstanding antibacterial performance, as an essential trace element, Zn is reported to be involved in bone metabolism, and it plays key roles in osteoblast activity as evidenced by the increased cell adhesion and proliferation.14,15 Furthermore, Zn exerts a direct effect on bone mineralization because of its effect on nucleation and mineral growth, which is of great significance for skeletal development.16 Numerous studies in vivo have documented that the Zn-modified implant exhibited excellent osseointegration between the bone and the implant,17 while the extracellular matrix calcification in osteoblasts was inhibited in the absence of Zn, which resulted in the decrease of matrix proteins, type I collagen and ALP activity.18 Likewise, nucleic acid metabolism, cell growth, division and tissue repair also require sufficient supply of Zn.14
Evidence of the beneficial effects brought by Zn in antibiosis and osteogenesis has motivated us to incorporate Zn into titanium-based implants for skeletal tissue applications. Nevertheless, fast release of Zn is adverse to long-term antibacterial activity and overdosage of Zn is a potential hazardous factor for osteoblasts. The balance between antibacterial properties and osteogenesis is important for surgical implantation. Therefore, it is of vital importance to develop suitable nanoreservoirs with an efficient Zn release system for long-term antibacterial activity, bio-safety and osteogenesis. Depositing antibacterials into TiO2 nanotubes is known to be the most common method.19–21 However, it is still a challenge to uniformly deposit an appropriate amount of antibacterial drugs into TiO2 nanotubes because deposition at the pore entrance is easier than that in the internal part of the nanotubes due to the existence of surface tension. Therefore, it is difficult to control the release of antibacterial drugs and eliminate the cytotoxicity caused by high concentration antibacterial drugs.22 In order to realize the controlled release of Zn, in our experiment, highly ordered ZnO nanorods with compatible density and dimensionality were prepared on a titanium substrate first, and then zirconia (ZrO2) was further coated onto the ZnO nanorods using an in situ hydrolyzed precursor. Similar to TiO2, ZrO2 nanotubes are also promising implant materials for orthopedic applications such as artificial knees and dental implants owing to their strong corrosion resistance and long-term stability,23 as well as in vivo biocompatibility.24,25 It was found that the adhesion and spreading of mesenchymal stem cells were enhanced on ZrO2 nanotubes with a diameter of ∼15–30 nm.26 The structure of ZrO2 nanoarrays was controlled by the regulation of the sacrificial template, and the Zn content was controlled by the dissolution conditions in our experiments. The antibacterial activity of ZrO2 nanoarrays with different Zn contents was elucidated along with cell–material interactions.
2. Materials and methods
2.1. Sample fabrication
Firstly, Ti foils with the dimension of 3 cm × 3 cm were ultrasonically cleaned with acetone, ethanol and deionized water sequentially. Then, the hybrid nanorods were synthesized by three steps as shown in Fig. 1: (1) hydrothermal synthesis of ZnO nanorods (ZnO-NR) on Ti foils, (2) coating of ZrO2 shell on the ZnO nanorods by precursor hydrolyzing in situ and (3) dissolution of ZnO. Firstly, ZnO nanorods (ZnO-NR) were prepared as per previous work via the seed-assisted hydrothermal method.27 Briefly, zinc acetate (0.025 mol) and diethanol amine (0.06 mol) were dissolved in ethanol (100 mL) solution at 60 °C under constant intense stirring for 1 h to yield a clear and homogeneous sol. Then, the sol was spin-coated onto the treated Ti foils and annealed at 500 °C for 1 h to produce a ZnO seed layer. The seeded substrates were hydrothermally treated in a precursor solution (50 mL) containing zinc nitrate (10 mM), ammonium fluoride (20 mM) and ammonium hydroxide (2.5 mL) at 70 °C for 8 h to produce ZnO-NR. Next, for the synthesis of the ZrO2 coating layer, ZnO-NR grown on Ti was dipped into a mixed solution (4 mL) of ethanol and zirconium(IV) propoxide (v![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v = 10
v = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) for 1 h and then the immersed products were annealed at 500 °C for 1 h, resulting in the core–shell structure nanorods (CS-NR). Finally, samples with the top-away structure and nanotube structure denoted as TA-NR and ZrO2-NT containing different contents of Zn can be obtained by etching CS-NR in 100 mL diluted HCl acid (pH = 3 and pH = 2.5) for 24 h.
1) for 1 h and then the immersed products were annealed at 500 °C for 1 h, resulting in the core–shell structure nanorods (CS-NR). Finally, samples with the top-away structure and nanotube structure denoted as TA-NR and ZrO2-NT containing different contents of Zn can be obtained by etching CS-NR in 100 mL diluted HCl acid (pH = 3 and pH = 2.5) for 24 h.
 | ||
| Fig. 1 Schematic illustration of the fabrication of Zn-incorporated nanoarrays and biological assessment. | ||
2.2. Surface characterization
The obtained products were characterized by X-ray diffraction (XRD, X'Pert PRO MRD, PANalytical, Netherlands), field-emission scanning electron microscopy (SEM; JEOL, JSM-6700F, Japan), and transmission electron microscopy (TEM; JEM-2100 (HR), 200 kV). The EDS test was carried out on the energy dispersive X-ray spectrometer (EDS) which was equipped with the SEM of QUANT200. The aqueous wettability of different substrates was analyzed by contact angle measurements using a Model 200 video-based optical system. The images of water drops on the sample surface were recorded using a digital CCD camera, and then analyzed with software supplied by the manufacturer.2.3. The Zn ion release
To detect the release time profile of the Zn ion in vitro, each sample with the dimension of 1 cm × 1 cm was immersed in 5 mL PBS for 1 day and then removed and immersed in another 5 mL fresh PBS. This process was repeated for 21 days to generate solutions at different time points. The amounts of Zn released from different samples were analyzed by inductively-coupled plasma atomic emission spectrometry (ICP-AES, IRIS Advantage ER/S).2.4. Antibacterial assay
E. coli and S. aureus were both selected to evaluate the antibacterial activities of different samples. E. coli was cultured in the Luria–Bertani (LB) medium, and S. aureus was cultured in beef extract-peptone (BEP) medium at 37 °C for one day. The bacterial suspension was diluted to an initial concentration of approximately 1 × 106 CFU mL−1. All samples were sterilized by autoclaving at 121 °C for 30 min before the experiment. Each specimen was exposed to 200 μL bacterial suspension at 37 °C for one day. At the end of the incubation period, the culture medium was collected to determine the viable counts of planktonic bacteria through the dilution method of plate counting. The antibacterial rate for planktonic bacteria was calculated based on the following formula:where A indicates the colony forming units (CFUs) of the control group with no specimen, and B is the CFUs of the experimental group. Bacteria adhered on each specimen were fixed with 3% glutaraldehyde at 4 °C for 1 h, then dehydrated in a graded ethanol series, and examined by scanning electron microscopy. As for bacteriostatic ring assay, E. coli and S. aureus were adjusted to a concentration of 1 × 107 CFU mL−1. 100 μL bacterial suspensions were spread onto agar plates and then the samples were lightly placed on the inoculated agar plates and incubated at 37 °C for 24 h, 48 h and 72 h, respectively. A series of representative pictures were taken with an optical camera (Panasonic, DMC-FZ50).
2.5. Cell culture
Newborn mouse calvaria-derived MC3T3-E1 cells were obtained from a preservation center for typical culture in Wuhan University (Wuhan, China). Cells were cultured in a-MEM (HyClone) containing 10% (v/v) fetal bovine serum (FBS; Gibco) and 1% penicillin/streptomycin and incubated in a humidified atmosphere of 5% CO2 at 37 °C. The cell culture medium was changed every three days. The cells of passages 2–4 were used in the experiments.2.6. Cell morphology observation
Cell morphology was observed by SEM (JSM-6700F) and CLSM (Leica, Germany) to evaluate the attachment behaviors of MC3T3-E1 cells on different materials. MC3T3-E1 cells were seeded in 96-well plates at an initial density of 1 × 104 cells per well. After 6 h culturing, cells were rinsed with phosphate-buffered saline, fixed with 3% glutaraldehyde, dehydrated in a graded ethanol series, freeze-dried, sputter coated with gold, and observed by SEM. For the immunofluorescence microscopy observation, after incubation for 24 h on different samples at an initial density of 1 × 104 cells per well, the attached cells were fixed in 4% paraformaldehyde for 30 min at 4 °C. Then, 0.1% Triton X-100 was added to the samples for 5 min before staining the actin cytoskeletons. Samples were incubated with FITC-Phalloidin (Sigma) at room temperature in the dark for 60 min. Finally, the nuclei were stained with 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI, Sigma). The stained samples were finally mounted with 90% glycerinum for CLSM observation and the cell number on each sample was also counted.2.7. Cell viability detection
The MTT assay was used to evaluate the viability of MC3T3-E1 cells. MC3T3-E1 cells were plated on each sample in 96-well plates at an initial seeding density of 1 × 104 cells per well. After 24 h, 48 h and 72 h of culture, cells were rinsed uisng PBS. Subsequently, fresh cell culture media was added to the targeted wells, and the MTT dye agent was added in an amount equal to 10% of the culture media volume, according to the manufacturer's instructions (MTT kit, Sigma, USA). Then plates were incubated at 37 °C for another 4 h to form formazan, which was later dissolved by addition of dimethyl sulfoxide after removing the MTT containing medium. The absorbance of the final solution was measured using a microplate reader (DNM-9602) at a wavelength of 492 nm.2.8. Lactate dehydrogenase activity assay
Lactate dehydrogenase (LDH) released by cells into the culture media was measured to investigate the cytotoxicity. MC3T3-E1 cells were seeded onto different samples at a density of 1 × 104 cells per well. After incubation for 24 h, 48 h and 72 h respectively, the culture media was collected and centrifuged, and the activity of LDH in the supernatant was determined following the manufacturer's instructions.2.9. Alkaline phosphatase (ALP) assay
Alkaline phosphatase (ALP) activity was determined to assess the bone forming ability of MC3T3-E1 cells on the experimental substrate surfaces. After being cultured for 7 and 14 days, respectively, MC3T3-E1 cells on different substrates were lysed using 1% Triton X-100 overnight and then broken using a sonic oscillator for 30 min. The cell lysates were centrifuged at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 rpm for 4 min. The ALP activity in the supernatant was determined with the ALP assay kit by measuring the absorbance at 405 nm. The bicinchoninic acid (BCA) protein assay kit was used to calculate the total protein content, and the absorbance was measured at a wavelength of 560 nm using a microplate reader (DNM-9602). Finally, the ALP activity was normalized by the total intracellular protein production.
500 rpm for 4 min. The ALP activity in the supernatant was determined with the ALP assay kit by measuring the absorbance at 405 nm. The bicinchoninic acid (BCA) protein assay kit was used to calculate the total protein content, and the absorbance was measured at a wavelength of 560 nm using a microplate reader (DNM-9602). Finally, the ALP activity was normalized by the total intracellular protein production.
2.10. Quantitative reverse transcription polymerase chain reaction (qRT-PCR)
The expression levels of osteogenesis related genes were determined by qRT-PCR. Cells were seeded onto different samples at a density of 2 × 105 cells per well in the 6-well plates, then cultured for 2 weeks and harvested using TRIzol (Invitrogen) to extract the RNA. The obtained RNA was reverse transcribed into complementary DNA using the PrimeScript RT reagent kit (Takara). SYBR green chemistry was used to perform quantitative determinations of the mRNAs for type I collagen (Col-I), osteocalcin (OCN), osteopontin (OPN), bone morphogenetic protein-2 (BMP-2) and a housekeeping gene M-actin following the optimized protocol. The double stranded DNA-specific dye SYBR Green I was incorporated into the PCR buffer provided in the SYBR Premix Ex Taq™ reagent. The temperature profile of the reaction was 95 °C for 10 min, 40 cycles of denaturation at 95 °C for 10 s, annealing at 60 °C (Col-I, OCN, OPN and BMP-2) for 20 s, and extension at 72 °C for 20 s. The M-actin gene was used to correct for differences in RNA isolation, RNA degradation, and the efficiencies of the reverse transcription.2.11. Statistical analysis
The results were representative of at least three independent experiments and all data were expressed as mean ± standard deviation for n = 5. The statistical analysis was performed with the statistical software Origin7.5. p < 0.05 was considered to be significant and p < 0.01 was regarded to be highly significant.3. Results
3.1. Surface characterization
The Zn-incorporated nanoarrays were synthesized in three steps as shown in Fig. 1. The SEM images of ZnO nanorods (ZnO-NR), core–shell structure nanorods (CS-NR), top-away structure nanorods (TA-NR) and Zn-incorporated ZrO2 nanotubes (ZrO2-NT) are demonstrated in Fig. 2, respectively. It was revealed that highly ordered ZnO nanorods (ZnO-NR) grew on Ti substrates uniformly with the length of 4 μm and a diameter of 60–120 nm (Fig. 2a). The sufficient space between ZnO nanorods was crucial for the formation of the ZrO2 shell. Fig. 2b revealed that the integration of ZrO2 into the ZnO array made the nanorod surface coarser without damaging the ordered structure. The ZrO2 shell can be distinguished from the ZnO core (Fig. 2c) after CS-NR being etched by diluted HCl solution (pH = 3.0) for 24 h. Zn-incorporated ZrO2 nanotubes (Fig. 2d) were fabricated after 24 h in diluted HCl (pH = 2.5). | ||
| Fig. 2 The SEM of the samples for low and high magnifications. ZnO-NR (a1, a2, a3), CS-NR (b1, b2, b3), TA-NR (c1, c2, c3), ZrO2-NT (d1, d2, d3). | ||
The samples were further investigated by TEM. As shown in Fig. 3a, each ZnO nanorod was shelled by the ZrO2 layer with a thickness of approximately 15 nm. Both the top-away structure and nanotube structure of the as-prepared products can be more clearly viewed in Fig. 3b and c, in which the outer diameter was shown to be almost the same as the diameter of ZnO nanorod template used, further illustrating the structure inheritance during the process. The selected-area electron diffraction (SAED) pattern in Fig. 3d showed the polycrystalline structure of the ZrO2 nanotubes.
 | ||
| Fig. 3 The TEM analysis of (a) CS-NR, (b) TA-NR, (c) ZrO2-NT and (d) the selected area electron diffraction (SAED) pattern of ZrO2-NT. | ||
The chemical compositions of CS-NR were characterized by energy dispersive X-ray spectroscopy (EDS) (Fig. 4A). About 18.4% of O, 2.42% of F, 7.29% of Zr, 4.05% of Ti and 67.84% (mass percent) of Zn were found in the CS-NR substrate. A strong signal of the Zr element with atomic percentage of 3.22% confirmed that ZnO was successfully coated with ZrO2. The presence of fluorine in the EDS result was derived from the precursor solution containing the fluoride salt when fabricating ZnO nanorods. It has been reported that fluoride was widely used as a highly effective anticaries agent in dental implant fields, and fluorine ions can affect bacterial metabolism as an enzyme inhibitor.28 What is more, a small amount of fluorine had a stronger stimulating effect on cell proliferation and differentiation.29
In the XRD spectra (Fig. 4B), as for ZnO-NR, the peak at 2θ = 33° (002) was dominant, with an intensity much higher than that of other peaks, revealing the high c-axis growth orientation of the product. The XRD pattern in Fig. 4B unambiguously disclosed the peak of ZnO in CS-NR, TA-NR and ZrO2-NT samples, which had great significance for antibacterial activity. However, for ZrO2-NT, this peak drops sharply due to the decrease of the ZnO amount.
Surface hydrophilicity affects biological functions such as cell adhesion and spreading.21 As shown in Fig. 5, the water contact angle of ZnO-NR significantly decreased from 78° to 14° compared with that of the Ti plate, and the contact angles of CS-NR, TA-NR and ZrO2-NT were less than 40°. The nanoarrays showed a significantly smaller water contact angle than pristine Ti discs, indicating a much higher hydrophilicity. Changes in the water contact angles stem from the surface morphology and chemistry, which was confirmed by Yu et al.30
 | ||
| Fig. 5 Water contact angles on different samples. (A) the optical pictures and (B) statistical diagram. Error bars represent mean ± SD for n = 5, **p < 0.01 compared to the Ti. | ||
3.2. The Zn ion release
The non-accumulated Zn ion released from the CS-NR, TA-NR, ZrO2-NT was measured by ICP-AES (Fig. 6). It was noteworthy that stable Zn ion release profiles were observed from all three substrates in a time span up to 21 days, which was crucial for long-term antibacterial activity. Thereinto, a smaller amount of Zn ion released from ZrO2-NT than that from CS-NR and TA-NR. This, to a great extent, could be attributed to the smallest quantity of ZnO in ZrO2-NT among the three samples.3.3. The antibacterial effect in vitro
The morphology of S. aureus and E. coli on different substrates was observed by SEM after the samples being exposed to 200 μL bacteria suspension at 37 °C for 24 h. Both S. aureus and E. coli cultured on flat Ti displayed an integrated surface and exhibited a healthy binary fission (yellow arrows in Fig. 7a1 and a2) without notable rupture or released intracellular component on the cell surface. In contrast, S. aureus and E. coli cultured on ZnO-NR, CS-NR, TA-NR and ZrO2-NT emerged as a wide range of abnormalities with various degrees of twist and deformation (red arrows in Fig. 7), indicating that all nanoarrays with the Zn content exhibited a great ability to inhibit bacteria. | ||
| Fig. 7 SEM views of S. aureus (a1–e1) and E. coli (a2–e2) seeded on Ti (a1–a2), ZnO-NR (b1–b2), CS-NR (c1–c2), TA-NR (d1–d2), ZrO2-NT (e1–e2). | ||
In order to assess the antibacterial activity of the samples, at the end of the incubation period of 24 h, the culture medium was collected and re-cultivated on agar plates according to the method of plate counting. Fig. 8A illustrated the representative photographs of the bacteria colony number in the control group, Ti, ZnO-NR, CS-NR, TA-NR and ZrO2-NT, respectively. Total plate counts of the largest number were detected on the Ti for both S. aureus and E. coli, whereas the colony forming units were significantly reduced on other experimental groups. It was noteworthy that neither E. coli colony nor S. aureus colony could be found on the agar plate of ZnO-NR, suggesting that ZnO had a strong antibacterial effect on both bacteria. Besides, the antibacterial rates of ZnO-NR against S. aureus and E. coli were approximately 100%. In comparison, the antibacterial rates measured on CS-NR, TA-NR and ZrO2-NT were all higher than 95% (Fig. 8B and C), illustrating their good antibacterial effect. It can also be inferred that the antibacterial capability was improved with the increase of Zn content. On the contrary, the percentage reduction of S. aureus and E. coli seeded on Ti reached 18% and 38% below zero, supporting the fact that Ti could significantly multiply the bacteria.
To further investigate the long-term antibacterial properties of all substrates, the inhibition zones of ZnO-NR, CS-NR, TA-NR and ZrO2-NT against both S. aureus and E. coli were detected after 24 h, 48 h and 72 h of culture, respectively. As shown in Fig. 9A and B, there was no inhibition zone around the pure Ti at any time for both bacteria, while ZnO-NR exhibited superior antibacterial performance with the largest inhibition zone among those of all samples under the same conditions. In general, diameters of the inhibition zone for both bacteria increased gradually with the culture time elongating (Fig. 9C and D), accounting for sustained antibacterial activities of all nanoarrays.
3.4. Cell morphology
To evaluate cell adhesion and growth on different samples, the morphologies of MC3T3-E1 cells were monitored by SEM after 6 h of culture (Fig. 10). Clearly, MC3T3-E1 cells cultured on ZnO-NR appeared to be round (Fig. 10b1–b3) without any lamellipodia or thin filopodia, illustrating that there was no spread on the ZnO nanorods. The lack of lamellipodia is probably due to an inability of cells to establish strong initial adhesion to the substrate, thereby altering the dynamics of cell spreading, which causes cell death.31 Besides, an overdose of the Zn ion would also lead to cytotoxicity.32 In contrast, most of the MC3T3-E1 cells were observed to have much more pronounced protrusion of filopodia with significantly longer configuration and a higher degree of contact on pure Ti, CS-NR and TA-NR, especially on ZrO2-NT. | ||
| Fig. 10 SEM images of MC3T3-E1 cells cultured for 6 h on Ti (a1–a3), ZnO-NR (b1–b3), CS-NR (c1–c3), TA-NR (d1–d3), ZrO2-NT (e1–e3). | ||
Immunofluorescent images of cytoskeletal actin (green) with the corresponding DAPI nuclear staining (blue) for cells cultured on different substrates are displayed in Fig. 11. Both the morphology and number (Fig. 12A) of adhered cells revealed significantly different surface properties between flat Ti and other nanoarrays. Fluorescent images demonstrated a similar trend of cell spreading on ZnO-NR compared with SEM results. MC3T3-E1 cells on CS-NR and flat Ti were relatively poor in spreading into a spindle shape, behaving as if they were undifferentiated quiescent cells. On the other hand, those cells on TA-NR and ZrO2-NT showed a clear topological configuration with more abundant and well-established filopodia extensions after an identical 24-hour culture, exhibiting a typical polygonal osteoblastic shape. The diversity of cell morphology results from different Zn contents of the substrates. Besides, compared with the Ti substrate, the nanotopography of the nanoarrays with larger surface areas facilitates cell growth in many different ways, including cell adhesion, cell morphology and proliferation. Previous research indicated that among cell responses to different nanopatterns, ZrO2 nanotube arrays could provide a better environment for cell adhesion and growth,25 which is why ZrO2-NT is better than other substrates.
3.5. Cell viability
An MTT assay was performed to estimate the proliferation and activity of viable cells on the experimental samples. Fig. 12B showed the optical density (OD) of reaction products of the MTT working solution with MC3T3-E1 cells cultured on different substrates after 24 h, 48 h, and 72 h, respectively. Overall, the cell viability increased with the elongation of culture time. MC3T3-E1 cells cultured onto the ZnO-NR substrate displayed the lowest cell viability when compared with others, indicating again the adverse influence of ZnO arrays on cytocompatibility. On the other hand, ZrO2-NT was proved to be a shelter with comparatively higher cell viability than Ti, CS-NR and TA-NR at any time, suggesting that the enhanced cell viability by ZrO2-NT should be mainly related to a smaller amount of Zn content.3.6. Cytotoxicity
It is known that Zn is an essential trace element that has stimulatory effects on osteogenetic activity and bone formation at a low concentration, while Zn overdose would lead to cytotoxicity.15 Therefore, the level of LDH released from cells cultured on these samples was measured to assess cytotoxicity. As shown in Fig. 12C, at each duration interval, ZnO-NR showed higher LDH activity than flat Ti, demonstrating strong cytotoxicity. The LDH activity of CS-NR and TA-NR had no significant difference compared with flat Ti, whereas ZrO2-NT exhibited much lower LDH activity than Ti did, illustrating no cytotoxicity or even better cytocompatibility.3.7. ALP activity
In order to determine the early differentiation of MC3T3-E1 cells, the ALP activity normalized according to the total protein content was analyzed after the cells were cultured on different substrates for 7 and 14 days, respectively. As shown in Fig. 12D, the ALP activities on all samples showed a time dependent pattern. MC3T3-E1 cells cultured on CS-NR and TA-NR generated more ALPs than those on Ti, but the statistical difference was not obvious. For ZrO2-NT, ALP was significantly up-regulated at either day 7 or 14 (p < 0.05 or p < 0.01), implying that the ZrO2-NT had an advantage over others as a bioactive implant for it stimulated early bone formation better than others.3.8. Osteogenesis-related gene expressions
To determine the osteogenic potential of MC3T3-E1 cells on different substrates, the gene expression of osteogenic markers, type I collagen (Col-I), osteocalcin (OCN) and osteopontin (OPN) and bone morphogenetic protein 2 (BMP-2), were evaluated by real time-polymerase chain reaction (RT-PCR) after 14 days of culture.The gene intensity of Col-I, one of the most important components of the extracellular matrix in natural bone tissue,33 was found to be reduced on all the Zn-incorporated nanoarrays when compared with that on the Ti substrate after 14 days of incubation, although there was no significant statistical difference between them (Fig. 13A). The mRNA level of OCN, a late marker for osteogenic differentiation,34 was up-regulated on ZrO2-NT when compared with pure Ti and other groups (Fig. 13B). A similar trend was also found in the mRNA level of OPN (Fig. 13C), one of the most abundant proteins in the bone matrix. Remarkably, the gene expression of BMP-2, which plays a key role in skeletal development by inducing bone formation and regeneration, exhibited the highest level on the ZrO2-NT substrate among all experimental groups (Fig. 13D).
 | ||
| Fig. 13 Relative expressions of (A) Col-I, (B) OCN, (C) OPN and (D) BMP-2 by MC3T3-E1 cells cultured on different substrates for 2 weeks, all values normalized to M-actin. **p < 0.01 compared to Ti. | ||
4. Discussion
Orthopaedic implant operation requires new materials with suitable antibacterial properties and sufficient osteogenetic activities to prevent millions of implant-associated infections worldwide. In order to obtain long-term antibacterial effect, the widely used approach in the preparation of titanium-based antimicrobial drugs carrier system can be described as the preparation of nanotubes by the anodic oxidation method, followed by drug-loading into nanotubes by complex ways.35–37 However, it still remains a challenge to deposit drugs uniformly into nanotubes and maintain its invariant morphology at the same time. On the other hand, excessive drugs accumulating out of nanotubes often leads to eruptible release and causes cell toxicity.21In this study, we report novel bone implant materials with both favorable antibacterial control systems and high osteogenic activity, and the schematic results can be seen in Fig. 14. Highly ordered ZnO nanorods with compatible density and dimensionality grown vertically on the Ti substrate were employed as the morphology template. ZrO2 was then integrated into the ZnO nanorods uniformly as a nanosized shell layer in the form of quasi-continuous nanoparticulates, with a large number of mesopores for Zn ion penetration and transportation, making it possible to provide a long-term antibacterial effect. ZrO2 coating not only precluded the direct contact between the cell and ZnO nanorods, but also constructed suitable extracellular microenvironments for the cell and enhanced the biocompatibility of implants. The rough coating surface could strongly support osteoblast adhesion and proliferation as reported by Wang et al.38 As demonstrated in the SEM image (Fig. 2d), there was more plenitudinous interconnecting space between the ZrO2 nanotubes (∼500 nm) than common ZrO2 nanotubes prepared by electrochemical anodization.39 The high connectivity was more conducive to cell adhesion and proliferation for the reason that the structure makes it easy for nutrient supply, gas diffusion, as well as metabolic waste removal, all of which contribute greatly for new bone formation.40
As can be seen clearly in the TEM images (Fig. 3), the content of ZnO could be easily controlled by the conditions of the solution. The released Zn ion greatly influenced the antibacterial properties, biocompatibility and the expression of osteogenesis-related gene expression as depicted in Fig. 14. Actually, it can be inferred from this study that simultaneously good antibiosis and cytocompatibility is hardly achieved if the Zn dosage is relatively high. Nevertheless a delicate balance between antibiosis and biocompatibility could be successfully reached by simply controlling the ZnO content and the Zn ion release.
The antibacterial effect was examined with both S. aureus and E. coli. It is well accepted that the great amount of active oxygen species produced by ZnO would disrupt the cell membranes.41 Indeed, the SEM analysis (Fig. 7) confirmed that most of both bacteria species cultured with ZnO-NR, CS-NR, TA-NR and ZrO2-NT were extensively injured on cell membranes. The integrity loss of the membranes further leads to malfunction of the permeability barrier, which ultimately caused the cell death.42 It was reported that the release of zinc ions could pierce the cell membranes and disturb bacterial functions by inhibition of the enzyme formation.12 Typical pictures of recultivated S. aureus and E. coli colonies on agar plates manifest that all groups incorporated Zn show obvious inhibition on both bacteria.
As proved by previous research,43 the reason for bacterial infections was the formation of biofilm on the surface of orthopedic implants. Reducing the number of viable bacteria is the most direct way to prevent biofilm formation. Making the interface of implant be rapidly covered with bistiocyte could prevent the implant from potential microbe adhesion effectively due to a living space competition.44 MC3T3-E1 cells were cultured on different substrates to assess the cell–surface interactions. SEM (Fig. 10) images of adherent MC3T3-E1 cells on CS-NR, TA-NR and ZrO2-NT revealed that cells spread well with noticeable filopodia extensions after incubation for the initial 6 hours. As one of the key signals for cell adhesion, filopodia is highly regulated by the reorganization of actin cytoskeleton and focal adhesion.45 The analysis with fluorescence microscopy (Fig. 11) after 24 h demonstrated that MC3T3-E1 cells adhered to TA-NR and ZrO2-NT substrates were typically polygonal, while they tended to be spindle on CS-NR and round on ZnO-NR. The cell density on ZnO-NR was much less than that on TA-NR and ZrO2-NT. These results suggested that ZnO nanorods were strong in antibacterial properties, but they also exhibited toxicity to cells as they dissuaded the MC3T3-E1 cell adhesion and reduced the cell viability as well, which was consistent with previous report that cells were unable to assemble focal adhesions and stress fibers on ZnO nanorods.31
For CS-NR, though ZnO was coated with ZrO2 completely, the amount of ZnO incorporated was still more than that in TA-NR and ZrO2-NT. Thus there were more Zn ions penetrating the ZrO2 coating and accumulating in the culture medium, leading to slight cytotoxicity as demonstrated by the lower level of MTT formation and the higher level of released LDH compared to those of the Ti. It was suggested that the effect of Zn was dose dependent, as low dose Zn enhances bone formation while ultrahigh doses induce toxicity.46 Previous researches revealed that Zn ions at a concentration of 0.08–0.3 ppm will have a positive effect on osteoblast functions and bone formation,47,48 while in the range of 2–8 ppm, Zn ions can inhibit normal osteoblasts function via oxidative stress.49
In comparison with other substrates, ZrO2-NT showed a significant increase in cell adhesion, activity and the lower LDH level. What is more, the early differentiation marker ALP activity detected in the ZrO2-NT group was higher relative to other substrates. Due to the smallest amount of ZnO incorporated in the ZrO2-NT, limited Zn ions were slowly released into the cell medium through the nanotubular structure, forestalling cytotoxicity-related problems observed in both ZnO-NR and CS-NR groups as described above. It was found that the dosage as well as the possible biological effect of Zn could be controlled by the content of ZnO and the delivery of Zn ions.50 Previous studies had revealed that the pharmacological performance of Zn-containing implants could be greatly influenced by the pattern of Zn release.1 In general, a slow and sustained release of the Zn-incorporated implant material significantly enhanced osseointegration.49 In this study, the ZrO2-NT structure balances antibiosis and osteogenesis successfully by inhibiting the growth of S. aureus and E. coli clearly and increasing the osseointegration in vitro by stimulating MC3T3-E1 cell adhesion and proliferation. It has been corroborated from several aspects that the unique nanotubular structure showed profound effects on the differentiation of MC3T3-E1 cells in vitro. The likely mechanism might be ascribed to the regulation of ALP activity and the expression of osteogenesis-related gene expression (Col-I, OCN, OPN and BMP-2). The novel drug-loaded nanotubular material was verified to improve the cellular activity on the surface, which is important for the bone–implant contact in vivo.
5. Conclusion
In this study we reported direct evidence that a unique bifunctional effect for cellular activities and antibiosis, at least in vitro, could be obtained by means of carefully controlling the Zn content and release. The content of incorporated Zn can be easily controlled by the pH value of the HCl solution used in the preparation of the nanostructures. All Zn-incorporated nanoarrays showed good antibacterial properties against both E. coli and S. aureus, as directly reflected by high antibacterial rates and apparent inhibition zone. Analysis of the biocompatibility confirmed that different hybrid nanoarrays could result in promotion of the adhesion and spreading of MC3T3-E1 cells to variable degrees. ZrO2-NT could balance well the antibiosis and osteogenesis, at least in vitro, as proved by its good antibacterial effect as well as the up-regulated MTT and ALP activities, along with the increase of the osteogenesis-related gene expression of OCN, OPN and BMP-2 mRNA. The novel bone implant materials with better antibacterial properties can promote osteogenesis, which has promising and useful applications in biomedical devices and antibacterial control systems.Acknowledgements
This work was supported by the National Natural Science Foundation of China (no. 50802032 and 21103059), the Key Project of Natural Science Foundation of Hubei Province (no. 2011CDA092), the Key Scientific Project of Wuhan City (no. 2013011801010598), the Scientific Project of AQSIQ (no. 2013IK093) and self-determined research funds of CCNU from the colleges’ basic research and operation of MOE (no. CCNU13A05007).Notes and references
- H. Hu, W. Zhang, Y. Qiao, X. Jiang, X. Liu and C. Ding, Acta Biomater., 2012, 8, 904 CrossRef CAS PubMed.
- N. Høiby, T. Bjarnsholt, M. Givskov, S. Molin and O. Ciofu, Int. J. Antimicrob. Agents, 2010, 35, 322 CrossRef PubMed.
- O. Rzhepishevska, S. Hakobyan, R. Ruhal, J. Gautrot, D. Barbero and M. Ramstedt, Biomater. Sci., 2013, 1, 589 RSC.
- K. Varaprasad, G. S. M. Reddy, J. Jayaramudu, R. Sadiku, K. Ramam and S. S. Ray, Biomater. Sci., 2014, 2, 257 RSC.
- M. M. Fernandes, A. Francesko, J. Torrent-Burgués and T. Tzanov, React. Funct. Polym., 2013, 73, 1384 CrossRef CAS PubMed.
- D.-W. Lee, Y.-P. Yun, K. Park and S. E. Kim, Bone, 2012, 50, 974 CrossRef CAS PubMed.
- A. Hoppe, V. Mouriño and A. R. Boccaccini, Biomater. Sci., 2013, 1, 254 RSC.
- W. Jiang, H. Mashayekhi and B. Xing, Environ. Pollut., 2009, 157, 1619 CrossRef CAS PubMed.
- L. C. Ann, S. Mahmud, S. K. M. Bakhori, A. Sirelkhatim, D. Mohamad, H. Hasan, A. Seeni and R. A. Rahman, Ceram. Int., 2014, 40, 2993 CrossRef CAS PubMed.
- K. R. Raghupathi, R. T. Koodali and A. C. Manna, Langmuir, 2011, 27, 4020 CrossRef CAS PubMed.
- A. Azam, A. S. Ahmed, M. Oves, M. S. Khan, S. S. Habib and A. Memic, Int. J. Nanomed., 2012, 7, 6003 CrossRef CAS PubMed.
- T. N. Phan, T. Buckner, J. Sheng, J. Baldeck and R. Marquis, Oral Microbiol. Immunol., 2004, 19, 31 CrossRef CAS.
- F. Velard, D. Laurent-Maquin, J. Braux, C. Guillaume, S. Bouthors, E. Jallot, J.-M. Nedelec, A. Belaaouaj and P. Laquerriere, Biomaterials, 2010, 31, 2001 CrossRef CAS PubMed.
- M. Roy, G. A. Fielding, A. Bandyopadhyay and S. Bose, Biomater. Sci., 2013, 1, 74 RSC.
- K. Huo, X. Zhang, H. Wang, L. Zhao, X. Liu and P. K. Chu, Biomaterials, 2013, 34, 3467 CrossRef CAS PubMed.
- I.-S. Kwun, Y.-E. Cho, R.-A. R. Lomeda, H.-I. Shin, J.-Y. Choi, Y.-H. Kang and J. H. Beattie, Bone, 2010, 46, 732 CrossRef CAS PubMed.
- K. Yusa, O. Yamamoto, M. Fukuda, S. Koyota, Y. Koizumi and T. Sugiyama, Biochem. Biophys. Res. Commun., 2011, 412, 273 CrossRef CAS PubMed.
- N. Iitsuka, M. Hie and I. Tsukamoto, Eur. J. Pharmacol., 2013, 714, 41 CrossRef CAS PubMed.
- A. Gao, R. Hang, X. Huang, L. Zhao, X. Zhang, L. Wang, B. Tang, S. Ma and P. K. Chu, Biomaterials, 2014, 35, 4223 CrossRef CAS PubMed.
- Q. Kang, Q. Z. Lu, S. H. Liu, L. X. Yang, L. F. Wen, S. L. Luo and Q. Y. Cai, Biomaterials, 2010, 31, 3317 CrossRef CAS PubMed.
- M. S. Aw, M. Kurian and D. Losic, Biomater. Sci., 2014, 2, 10 RSC.
- B. S. Necula, J. P. T. M. van Leeuwen, L. E. Fratila-Apachitei, S. A. J. Zaat, I. Apachitei and J. Duszczyk, Acta Biomater., 2012, 8, 4191 CrossRef CAS PubMed.
- F. Y. Zhou, B. L. Wang, K. J. Qiu, L. Li, J. P. Lin, H. F. Li and Y. F. Zheng, J. Biomed. Mater. Res., Part B, 2013, 101B, 237 CrossRef CAS PubMed.
- H.-L. Huang, Y.-Y. Chang, Y.-C. Chen, C.-H. Lai and M. Y. C. Chen, Thin Solid Films, 2013, 549, 108 CrossRef CAS PubMed.
- Z. Lu, Z. Zhu, J. Liu, W. Hu and C. Ming Li, Nanotechnology, 2014, 25, 215102 CrossRef PubMed.
- S. Bauer, J. Park, J. Faltenbacher, S. Berger, K. von der Mark and P. Schmuki, Integr. Biol., 2009, 1, 525 RSC.
- T. D. Zaveri, N. V. Dolgova, B. H. Chu, J. Lee, J. Wong, T. P. Lele, F. Ren and B. G. Keselowsky, Biomaterials, 2010, 31, 2999 CrossRef CAS PubMed.
- J. Lellouche, A. Friedman, J. P. Lellouche, A. Gedanken and E. Banin, Nanomed. Nanotechnol., 2012, 8, 702 CrossRef CAS PubMed.
- Y. Wang, S. Zhang, X. Zeng, L. L. Ma, W. Weng, W. Yan and M. Qian, Acta Biomater., 2007, 3, 191 CrossRef CAS PubMed.
- W.-q. Yu, X.-q. Jiang, F.-q. Zhang and L. Xu, J. Biomed. Mater. Res., Part A, 2010, 94A, 1012 CAS.
- J. Lee, B. S. Kang, B. Hicks, T. F. Chancellor Jr., B. H. Chu, H. T. Wang, B. G. Keselowsky, F. Ren and T. P. Lele, Biomaterials, 2008, 29, 3743 CrossRef CAS PubMed.
- W. Song, J. Zhang, J. Guo, J. Zhang, F. Ding, L. Li and Z. Sun, Toxicol. Lett., 2010, 199, 389 CrossRef CAS PubMed.
- S. E. Bae, J. Choi, Y. K. Joung, K. Park and D. K. Han, J. Controlled Release, 2012, 160, 676 CrossRef CAS PubMed.
- Y. Hu, K. Cai, Z. Luo, Y. Zhang, L. Li, M. Lai, Y. Hou, Y. Huang, J. Li, X. Ding, B. Zhang and K. L. Paul Sung, Biomaterials, 2012, 33, 3515 CrossRef CAS PubMed.
- K. C. Popat, M. Eltgroth, T. J. LaTempa, C. A. Grimes and T. A. Desai, Biomaterials, 2007, 28, 4880 CrossRef CAS PubMed.
- H. Li, Q. Cui, B. Feng, J. Wang, X. Lu and J. Weng, Appl. Surf. Sci., 2013, 284, 179 CrossRef CAS PubMed.
- M.-Y. Lan, S.-L. Lee, H.-H. Huang, P.-F. Chen, C.-P. Liu and S.-W. Lee, Ceram. Int., 2014, 40, 4745 CrossRef CAS PubMed.
- G. Wang, X. Liu, H. Zreiqat and C. Ding, Colloids Surf., B, 2011, 86, 267 CrossRef CAS PubMed.
- C. J. Frandsen, K. S. Brammer, K. Noh, L. S. Connelly, S. Oh, L.-H. Chen and S. Jin, Mater. Sci. Eng., C, 2011, 31, 1716 CrossRef CAS PubMed.
- H. Zreiqat, Y. Ramaswamy, C. Wu, A. Paschalidis, Z. Lu, B. James, O. Birke, M. McDonald, D. Little and C. R. Dunstan, Biomaterials, 2010, 31, 3175 CrossRef CAS PubMed.
- S. Nair, A. Sasidharan, V. V. Divya Rani, D. Menon, S. Nair, K. Manzoor and S. Raina, J. Mater. Sci. Mater. Med., 2009, 20, 235 CrossRef PubMed.
- G. Applerot, A. Lipovsky, R. Dror, N. Perkas, Y. Nitzan, R. Lubart and A. Gedanken, Adv. Funct. Mater., 2009, 19, 842 CrossRef CAS.
- J. T. Seil and T. J. Webster, Acta Biomater., 2011, 7, 2579 CrossRef CAS PubMed.
- G. Subbiahdoss, B. Pidhatika, G. Coullerez, M. Charnley, R. Kuijer, H. C. van der Mei, M. Textor and H. J. Busscher, Eur. Cell. Mater., 2010, 19, 205 CAS.
- Y. Hu, K. Cai, Z. Luo, D. Xu, D. Xie, Y. Huang, W. Yang and P. Liu, Acta Biomater., 2012, 8, 439 CrossRef CAS PubMed.
- E. Saino, S. Grandi, E. Quartarone, V. Maliardi, D. Galli, N. Bloise, L. Fassina, M. G. C. De Angelis, P. Mustarelli and M. Imbriani, Eur. Cell. Mater., 2011, 21, 59 CAS.
- A. Hoppe, N. S. Güldal and A. R. Boccaccini, Biomaterials, 2011, 32, 2757 CrossRef CAS PubMed.
- G. Jin, H. Cao, Y. Qiao, F. Meng, H. Zhu and X. Liu, Colloids Surf., B, 2014, 117, 158 CrossRef CAS PubMed.
- S. Miao, K. Cheng, W. Weng, P. Du, G. Shen, G. Han, W. Yan and S. Zhang, Acta Biomater., 2008, 4, 441 CrossRef CAS PubMed.
- S. Miao, N. Lin, K. Cheng, D. Yang, X. Huang, G. Han, W. Weng and Z. Ye, J. Am. Chem. Soc., 2011, 94, 255 CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2015 |