Aqueous solubilization of hydrophobic supramolecular metal–organic nanocapsules†
Abstract
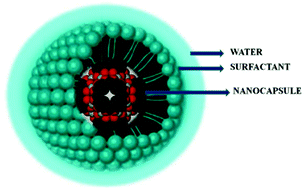
Micelles of metal-seamed C-propylpyrogallol[4]arene-based [(PgC3)nM4nL0–4n, L = ligand, M = metal] hexamers and dimers in water have been formulated using non-ionic surfactants and characterized using in situ scattering techniques. Polysorbitans (tween 20, tween 40, tween 80) are utilized to solubilize (PgC3)6Cu24 and (PgC3)6Ni24L24 hexamers, (PgC3)2Ni8L8, (PgC3)2Zn8L8, and (PgC3)2Co8L8 dimers in water. The dimensions of micelles of tweens⊂(PgC3)nM4nL0–4n dimers and hexamers are studied using dynamic light scattering (DLS; hydrodynamic radius/Rh) and small-angle neutron scattering (SANS). SANS data is fitted to a polydisperse core–shell–sphere model (static radius/RHS) and used to calculate the radius of gyration (Rg). An increase in radius of the tween 20 (C12)- and tween 40 (C16)-based micelles is observed with an increase in the radius of the nanocapsule (dimer vs. hexamer); however, given the longer alkyl tails of tween 80 (C18:1, cis-9), a progression in radius is not observed for tween 80-enclosed micelles due to the unsaturation (C9) and bent alkyl tail of the surfactant. The Rg : Rh ratios are close to 0.77 for the control (0.62), tween 20⊂dimeric (0.67) and tween 20⊂hexameric (0.68) micelles that demonstrates the sphericity of micelles in water. In contrast, the higher Rg/Rh ratio (0.76 to 0.93) of tween 80-based micelles is indicative of more elongated micellar species in aqueous solution.

 Please wait while we load your content...
Please wait while we load your content...