A convenient synthesis of 2-substituted benzofurans from salicylaldehydes†
Swetha Reddya,
Srinivas Thadkapallya,
Mounika Mamidyalaa,
Jagadeesh Babu Nanubolub and
Rajeev S. Menon*a
aMedicinal Chemistry and Pharmacology Division, CSIR-Indian Institute of Chemical Technology, Hyderabad 500 007, India. E-mail: srajeevmenon@gmail.com; Fax: +91-40-27193189
bCentre for X-ray Crystallography, CSIR-Indian Institute of Chemical Technology, Hyderabad 500 007, India
First published on 24th December 2014
Abstract
Base-mediated cyclocondensation of 2-hydroxybenzaldehydes with 3-bromo-1-(arylsulfonyl)propenes and 4-bromocrotonates afforded (E)-2-(2-sulfonylvinyl)benzofurans and (E)-2-benzofuranyl-3-acrylates respectively.
Introduction
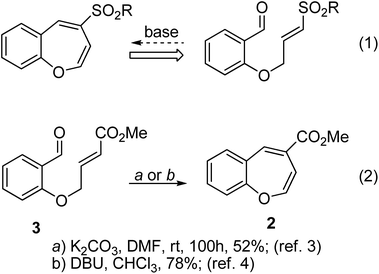
As part of a program in screening heterocycles for developing new anticancer therapeutics, we were interested in investigating the cytotoxicity profile of 4-sulfonyl-1-benzoxepine derivatives 1 (Fig. 1). The benzoxepine framework is found in a number of important natural products1 and 1-benzoxepine derivatives are known to exhibit biological activities such as estrogen regulator modulation.2A straightforward assembly of the 1-benzoxepine framework was envisaged as depicted in Scheme 1 (eqn (1)) in order to generate a library of derivatives for biological screening. This synthetic plan was adapted from the reports by Ciganek3 and Zeitler4 that described the formation of the corresponding carboxylate esters (eqn (2) and Scheme 1). For example, 4-methoxycarbonyl-1-benzoxepine 2 has been synthesized by base-mediated cyclization of the salicylaldehyde-derived ester 3.
 | ||
| Scheme 1 Planned synthesis of 1 (eqn (1)) and reported syntheses of its carboxy-analogue 2 (eqn (2)). | ||
Results and discussion
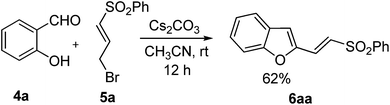
The attempted synthesis of 1 began with base-mediated O-alkylation of salicylaldehyde 4a with the previously described bromide 5a.5 This seemingly straightforward O-alkylation, however, afforded a mixture of products that were difficult to separate from each other by chromatography (the mixture of products arises presumably due to the base-catalyzed isomerization of the vinyl sulfone moiety into the corresponding allyl sulfone6 subsequent to the alkylation). Pleasingly, it was found that this mixture can be converted into a single product by exposing it again to base for a longer period of time. Further experiments revealed that the isolation of the O-alkylated intermediate was not necessary, as prolonged treatment of salicylaldehyde 4a and the bromosulfone 5a with 2 equivalents of cesium carbonate in acetonitrile for 12 h resulted in the formation of a single product. Spectroscopic analysis, however, indicated that the product is the benzofuran derivative 6aa (Scheme 2).In the 1H NMR spectrum of 6aa, the olefinic proton adjacent to the sulfonyl group resonated at δ 7.57 as a doublet with coupling constant of 15.0 Hz, confirming the presence of a trans double bond. The remaining olefinic hydrogen resonated along with four of the aromatic protons (δ 7.65–7.55). A singlet resonance corresponding to the benzofuran-3-H was discernible at δ 7.04 (1H). All other signals were in agreement with the assigned structure.
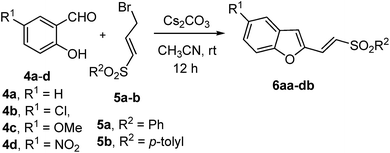
Natural and manmade molecules containing the benzofuran nucleus exhibit a wide range of biological activities.7 This includes, inter alia, antifungal,8a analgesic,8b antipsychotic8c and antimitotic8d activities. Therefore, the above-described annulation reaction that affords a benzofuran 6 endowed with a functionalizable handle at the 2-position appeared worth pursuing. Thus, a number of substituted salicylaldehydes 4a–d was treated with γ-bromovinyl sulfones 5a–b under the optimized reaction conditions. The results are summarized in Table 1.
The annulation reaction appears to be general affording 2-(2-sulfonylvinyl)benzofurans 6 in moderate to good yields. It is noteworthy that 2-functionalized benzofurans can be accessed in a one-pot operation from readily available starting materials, albeit in moderate yields. Out of the four substituted salicylaldehydes employed, 5-nitrosalicylaldehyde 4d failed to react under the optimized conditions, presumably due to the sluggish O-alkylation of the rather stable o-formyl-p-nitrophenolate intermediate.
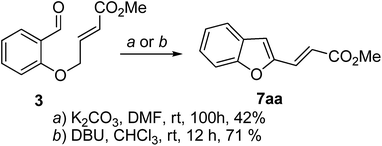
Thus it was evident that the approach3,4 that led to 4-methoxycarbonyl-1-benzoxepins (Scheme 1) was not suitable for the construction of the corresponding sulfonyl derivatives. This was surprising when the known propensity of E-vinyl sulfones to undergo base-mediated isomerisation to Z-allyl sulfones6 was taken into consideration. The unanticipated outcome of this reaction prompted us to investigate the previously described benzoxepine formation from salicylaldehyde and methyl-4-bromocrotonate. Thus, the ester 3, derived from salicylaldehyde when subjected to the cyclocondensation conditions as reported by Ciganek3 and Zeitler,4 afforded the benzofuran derivative 7aa (Scheme 3).
The product 7aa was characterized by spectroscopic analysis.9 In the 1H NMR, the olefinic protons were discernible as two mutually coupled doublets (J = 15.7 Hz) at δ 7.56 and 6.58. The 3-benzofuranyl hydrogen resonated at δ 6.94 as a singlet. It may be pointed out here that the spectroscopic data reported by Ciganek3 for the compound 2 matches with that obtained for 7aa in our laboratory. Most significantly, the presence of a mutually coupled pair of trans olefinic protons is inconsistent with the reported3 benzoxepine structure 2. Thus, in view of the new evidence, it may be concluded that the base-mediated cyclocondensation of 3 leads to the benzofuran derivative 7aa instead of the benzoxepine 2.10
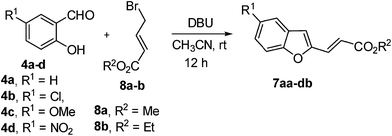
Optimization experiments revealed that the substituted benzofuran derivatives could be prepared in a one-pot operation by treating substituted salicylaldehydes 4a–d and 4-bromocrotonates 8a–b with 2 equivalents of 1,8-diazabicycloundec-7-ene (DBU) in acetonitrile at room temperature (Table 2).
As depicted in Table 2, the one-pot alkylation–annulation reaction can be extended to substituted salicylaldehydes. The benzofuran derivatives 7 are formed in moderate to good yields. It is important to note that the yields reported here correspond to the overall yield of two reactions, i.e., O-alkylation and cyclization.
Additionally, unambiguous evidence for the assigned structure (7) was obtained from single crystal X-ray analysis of a representative compound 7cb (Fig. 2).11
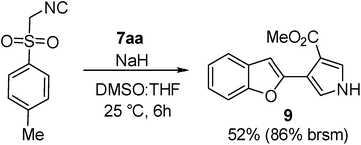
The α,β-unsaturated ester functionality offers a number of possibilities of further synthetic manipulations. For example, treatment of the benzofuran 7aa with van Leusen's reagent12 (tosylmethyl isocyanide) in presence of base afforded the 3,4-disubstituted pyrrole derivative 9 in 86% isolated yield (based on recovered stating material) (Scheme 4).
Conclusions
In conclusion, a convenient one-pot method for the synthesis of 2-alkenyl substituted benzofurans from 3-bromo-1-(arylsulfonyl)propenes or 4-bromocrotonates and salicylaldehydes was developed. It is noteworthy that both the salicylaldehydes and 4-bromocrotonates are commercially available and the benzofurans generated are amenable to further functionalization. The “pluripotent” phenylsulfonyl functional group13 has been termed as “arguably the most versatile functional group” and it is reasonable to assume that the benzofurans 6 possessing this functional group could also be employed in further transformations.Experimental
General information
All 1H NMR and 13C NMR spectra were recorded in CDCl3 solvent on Varian Bruker 300 MHz, a Varian Unity 400 MHz and Avance 500 MHz spectrometer at ambient temperature. IR spectra were recorded on Nicolet 380 FT-IR spectrophotometer. Mass spectra were obtained on a Finnegan Mat1020B, a micromass VG 70-70H or an Agilent technologies LC/MSD treapSL spectrometer operating at 70 eV using the direct inlet system and high resolution mass spectra (HRMS) were recorded on a QSTAR XL Hybrid MS/MS mass spectrometer. Melting points were recorded on an electrothermal apparatus and are uncorrected. All the reagents and solvents were used without further purification unless specified otherwise. Technical grade ethyl acetate and petroleum ether used for column chromatography were distilled prior to use. Column chromatography was carried out using silica gel (60–120 mesh and 100–200 mesh) packed in glass columns. All reactions were performed in oven-dried glassware with magnetic stirring. Salicylaldehydes (4a–d), methyl-4-bromocrotonates (8a, 85% technical grade) and ethyl-4-bromocrotonates (8b, 75% technical grade) used in the study were obtained from Aldrich and were used as received. 4-(2-Formylphenoxy)-but-2-enoate 3 was prepared by potassium carbonate-mediated alkylation of salicylaldehyde 4a with 8a.3 (E)-3-Bromo-1-(arylsulfonyl)propenes 5a–b were prepared as described by Gallagher and Grayson.14General procedure for the synthesis of 2-(β-sulfonylvinyl)benzofurans 6aa–cb
Cesium carbonate (326 mg, 1 mmol) was added to a solution of salicylaldehyde 4a–d (0.5 mmol) and 3-bromo-1-(arylsulfonyl)propene 5a–b (0.5 mmol) in anhydrous acetonitrile (5 mL). The resulting solution was stirred at ambient temperature for 12 h. The reaction mixture was then partitioned between dichloromethane and ice cold water, and aqueous phase was extracted with ethyl acetate. The combined organic phases were dried with anhydrous sodium sulfate and concentrated under reduced pressure. Column chromatography on silica gel using petroleum ether–ethyl acetate as eluent afforded analytically pure samples of 2-(2-sulfonylvinyl)benzofurans 6aa–cb.General procedure for the synthesis of 2-(β-alkoxycarbonylvinyl)benzofurans 7aa–db
DBU (152 mg, 1 mmol) was added to a solution of salicylaldehyde 4a–d (0.5 mmol) and the 4-bromocrotonate ester 8 (0.5 mmol; 0.07 mL for 8a and 0.09 mL for 8b) in anhydrous acetonitrile (5 mL). The resulting solution was stirred at ambient temperature for 12 h. The reaction mixture was then partitioned between dichloromethane and ice cold water, and aqueous phase was extracted with ethyl acetate. The combined organic phases were dried with anhydrous sodium sulfate and concentrated under reduced pressure. Column chromatography on silica gel using petroleum ether–ethyl acetate as eluent afforded analytically pure samples of 2-substituted benzofurans 7aa–db.Conversion of 7aa into the pyrrole derivative 9
A solution of 7aa (101 mg, 0.5 mmol) and tosylmethyl isocyanide (108 mg, 0.55 mmol) in anhydrous THF–DMSO (1 mL and 2 mL respectively) was added dropwise into a suspension of sodium hydride (60% dispersion in mineral oil, 42 mg, 1.1 mmol) in THF (2 mL). The mixture was stirred at room temperature for 6 h. The reaction mixture was then treated with water and extracted with ethyl acetate. The combined extracts were dried over anhydrous sodium sulfate, solvent was removed on a rotavapor and the residue obtained was chromatographed on silica gel using petroleum ether–ethyl acetate as eluent to afford the pyrrole derivative 9 (63 mg) and unreacted 7aa (40 mg).Acknowledgements
Financial support from Department of Science and Technology (DST), India in the form of a Ramanujan fellowship and a fast-track project to RSM is acknowledged. Financial support in part from the XIIth five year plan project “Affordable Cancer Therapeutics (ACT)” (CSC 0301) from the Council of Scientific and Industrial Research (CSIR), India is also acknowledged.Notes and references
- (a) S. Yamaguchi, N. Tsuchida, M. Miyazawa and Y. Hirai, J. Org. Chem., 2005, 70, 7505 CrossRef CAS PubMed; (b) S. Kim, Y.-W. Chin, B.-N. Su, S. L. Riswan, B. S. Kardono, J. J. Afriastini, H. Chai, N. R. Farnsworth, G. A. Cordell, S. M. Swanson and A. D. Kinghorn, J. Nat. Prod., 2006, 69, 1769 CrossRef CAS PubMed; (c) J. B. P. A. Wijnberg, A. van Veldhuizen, H. J. Swarts, J. C. Frankland and J. A. Field, Tetrahedron Lett., 1999, 40, 5767 CrossRef CAS; (d) F. A. Macias, J. M. G. Molinillo, R. M. Varela, A. Torres and F. R. Fronczek, J. Org. Chem., 1994, 59, 8261 CrossRef CAS; (e) S. K. Das, S. K. Dinda and G. Panda, Eur. J. Org. Chem., 2009, 204 CrossRef CAS.
- D. G. Lloyd, R. B. Hughes, D. M. Zisterer, D. C. Williams, C. Fattorusso, B. Catalanotti, G. Campaiani and M. J. Meegan, J. Med. Chem., 2004, 47, 5612 CrossRef CAS PubMed.
- E. Ciganek, Synlett, 1995, 1311 CAS.
- K. Zeitler and I. Mager, Adv. Synth. Catal., 2007, 349, 1851 CrossRef CAS.
- (a) C. Nájera, A. Pérez-Pinar and J. M. Sansano, Tetrahedron, 1991, 47, 6337 CrossRef; (b) J. Zindel and A. de Meijere, Synthesis, 1994, 190 CrossRef CAS.
- T. Hirata, Y. Sasada, T. Ohtani, T. Asada, H. Kinoshita, H. Senda and K. Inomata, Bull. Chem. Soc. Jpn., 1992, 65, 75 CrossRef CAS.
- (a) L. D. Luca, G. Nieddu, A. Porcheddu and G. Giacomelli, Curr. Med. Chem., 2009, 16, 1 CrossRef , and references cited therein; (b) M. Halabalaki, N. Aligiannis, Z. Papoutsi, S. Mitakou, P. Moutsatsou, C. Sekeris and A.-L. Skaltsounis, J. Nat. Prod., 2000, 63, 1672 CrossRef CAS PubMed.
- (a) M. W. Khan, M. J. Alam, M. A. Rashid and R. Chowdhury, Bioorg. Med. Chem., 2005, 13, 4796 CrossRef CAS PubMed; (b) P. R. Halfpenny, D. C. Horwell, J. Hughes, J. C. Hunter and D. C. Rees, J. Med. Chem., 1990, 33, 286 CrossRef CAS; (c) I. van Wijngaarden, C. G. Kruse, J. A. M. van der Heyden and M. T. M. Tulp, J. Med. Chem., 1988, 31, 1934 CrossRef CAS; (d) P. Wipf, J. T. Reeves, R. Balachandran, K. A. Giuliano, E. Hamel and B. W. Day, J. Am. Chem. Soc., 2000, 122, 9391 CrossRef CAS.
- S. Wei, X.-G. Wei, X. Su, J. You and Y. Ren, Chem.–Eur. J., 2011, 17, 5965 CrossRef CAS PubMed.
- Zeitler's report4 describes a schematic diagram without any experimental data for the assigned structure.
- ESI.†.
- H. Siderius, B. E. Hugenboom, D. van Leusen and A. M. van Leusen, Tetrahedron Lett., 1972, 13, 5337 CrossRef.
- A. El-Awa, M. N. Noshi, X. M. du Jourdin and P. L. Fuchs, Chem. Rev., 2009, 109, 2315 CrossRef CAS PubMed.
- E. T. Gallagher and D. H. Grayson, Org. Biomol. Chem., 2003, 1, 1374 CAS.
- S. Wei, X.-G. Wei, X. Su, J. You and Y. Ren, Chem.–Eur. J., 2011, 17, 5965 CrossRef CAS PubMed.
- N. Matsunaga, T. Kaku, F. Ito, T. Tanaka, T. Hara, H. Miki, M. Iwasaki, T. Aono, M. Yamaoka, M. Kusaka and A. Tasaka, Bioorg. Med. Chem., 2004, 12, 2251 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: 1H and 13C NMR spectra of all compounds and crystallographic data for compound 7cb. CCDC 1020845. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra14948c |
| This journal is © The Royal Society of Chemistry 2015 |