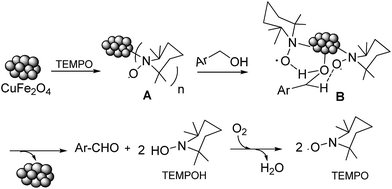
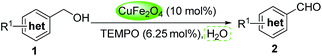
Magnetic copper ferrite nanoparticles/TEMPO catalyzed selective oxidation of activated alcohols to aldehydes under ligand- and base-free conditions in water†
Xiao Zhua,
Daoshan Yang*a,
Wei Weia,
Min Jiangb,
Lulu Lia,
Xiangbing Zhua,
Jinmao Youa and
Hua Wang*a
aThe Key Laboratory of Life-Organic Analysis, Key Laboratory of Pharmaceutical Intermediates and Analysis of Natural Medicine, School of Chemistry and Chemical Engineering, Qufu Normal University, Qufu 273165, Shandong, China. E-mail: yangdaoshan@tsinghua.org.cn; huawang_qfnu@126.com
bKey Laboratory of Bioorganic Phosphorus Chemistry, Chemical Biology (Ministry of Education), Department of Chemistry, Tsinghua University, Beijing 100084, People's Republic of China
First published on 20th November 2014
Abstract
A novel, effective and sustainable strategy for the synthesis of aldehydes has been developed using inexpensive, readily available, oxygen-stable and recyclable CuFe2O4 nanoparticles as the catalyst. The corresponding substituted aldehydes were obtained in moderate to good yields by aerobic oxidation of aromatic alcohols in water under dioxygen atmosphere. Importantly, a ligand or a base was not necessary. The catalyst was completely recoverable with an external magnet and could be reused six times without significant loss of catalytic activity.
Introduction
Direct conversion of primary alcohols to the corresponding aldehydes is still a pivotal reaction in organic synthesis.1 In traditional oxidation processes, the oxidation of alcohols is accomplished by use of stoichiometric amounts of inorganic oxidants such as CrO3, KMnO4, SeO2, hypervalent iodine compounds and toxic heavy metal salts.2 Unfortunately, these oxidants might lead to the formation of quantities of unfriendly waste in the environment such as non-recoverable toxic by-products, and large volumes of solid waste. In recent years, transition metals such as Pd,3 Au,4 Cu,5 etc., or their corresponding metal complexes have been widely applied in the aerobic alcohol oxidation. As a pioneering work, Nakazawa and co-workers recently reported a first iron-catalyzed method for aerobic oxidation of alcohols to aldehydes.6 Non-metal catalytic systems for this oxidation transformation have also been attract much attention,7 In 2014, Jahn and co-workers reported that TEMPO/t-BuONO system can catalyze primary and secondary alcohols to the corresponding aldehydes and ketones, respectively.8 However, in some cases, most of these methods might suffer from some drawbacks such as the rarity and price transition metal catalyst, ligand together with strong base conditions, metal contamination in the end products and undesirable stoichiometric oxidants, which should still impede their applications on a large scale. Therefore, a more green and effective process is needed.In view of the principles of green chemistry, the proposal of reaction conditions, media and catalysts, is still challenging in the current chemistry.9 Since catalysis has a tremendous impact on “green chemistry”, optimization of existing chemical transformations together with the development of practical, environmentally friendly processes depend greatly on improvement of catalyst performance.10 Recently, heterogeneous catalysts have attracted much attention in organic transformations due to their interesting reactivity as well as for economic and environmental reasons. Particularly, a large number of recyclable supported catalytic systems have been developed.11 For example, Li and co-workers reported an efficient palladium on graphene catalyst for the selective oxidation of aromatic alcohols to corresponding carbonyl compounds.11f Very recently, Garcia's group developed an elegant work for the oxidative transformation catalyzed by metal–organic frameworks with the assistance of TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy).12 Excellent as these works are, the small size of catalyst particles might often make their separation and recycling difficult, and the efficiency of the recovered catalyst might be somewhat reduced as a result of a filtration step.
Recently, magnetic nanoparticles (MNPs) have emerged as ideal catalysts or supports, and have been extensively used in organic transformations because of their easy preparation, large surface area ratio, high dispersion property in organic solvents and efficient separation by using an external magnet and without the need for filtration step.13 Among all the magnetic nanoparticles, copper(II) ferrite (CuFe2O4) nanoparticles have emerged as a powerful and excellent catalyst for many organic transformations.14 Furthermore, from the viewpoint of green chemistry, replacement of conventional hazardous organic solvents by safe and green reaction media has always been a thread in the sustainable chemistry. There is no doubt that, water can be considered as an attractive solvent, because it is easily available, non-toxic and environmentally friendly in the world. Besides that, water is also known to enhance the reaction rates and to affect the selectivity of a lot of organic transformations.15 In addition, for economical and environmental reasons, there is an increasing demand for the use of dioxygen as an ideal oxidant for many oxidation reactions due to its abundance, low cost and nontoxicity. In continuation of our endeavors to develop CuFe2O4-catalyzed organic transformations,16 we herein report a green, practical and efficient CuFe2O4-catalyzed aerobic oxidation of aromatic alcohols to aldehydes by using environmentally benign water as the solvent under dioxygen atmosphere. Notably, when we were performing these studies, Lipshutz and his coworkers reported an elegant example of the direct oxidation of activated alcohols to aldehydes or ketones using CuBr/TEMPO as the catalyst using water as the solvent.17
Results and discussion
The copper(II) ferrite nanoparticles were prepared according to the literature procedure,18 and were characterized by X-ray diffraction (Fig. 1). The energy-dispersive X-ray spectroscopy shows that the spheres are composed of Cu, Fe and O, as expected (Fig. 1, ESI†). Moreover, the diffraction patterns of all the peaks are in agreement with the standard XRD pattern (JCPDS34-0425), so are the ones after three usages. Also, as can be seen from the SEM and TEM images, the CuFe2O4 nanoparticles could remain well in a similar state, even after third cycles (Fig. 2 and 3).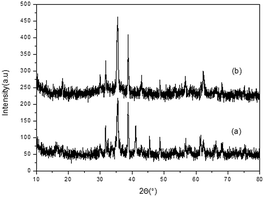 | ||
| Fig. 1 XRD spectrum of native CuFe2O4 catalyst. (b) XRD spectrum of reused CuFe2O4 catalyst after 3rd cycle. | ||
 | ||
| Fig. 2 (a) SEM images of the fresh CuFe2O4 nanoparticles. (b) SEM image of the CuFe2O4 nanoparticles after 3rd cycle. | ||
 | ||
| Fig. 3 (a) TEM image of the fresh CuFe2O4 nanoparticles. (b) TEM image of the CuFe2O4 nanoparticles after 3rd cycle. | ||
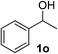
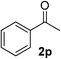
Initially, phenylmethanol (1a) was selected as the model substrate to optimize the reaction conditions, including the choice of solvent, temperature and the amount of TEMPO under a dioxygen atmosphere (Table 1). Firstly, a range of solvents such as H2O, toluene, DMF, DMSO and THF were investigated in the presence of 0.1 equiv. of CuFe2O4 nanoparticles and 6.25 mol% TEMPO (2,2,6,6-tetramethylpiperidine 1-oxyl) (relative to amount of 1a) under dioxygen atmosphere (entries 1–5), H2O and THF gave the highest yield among the solvents tested (entry 1 and 5), and we chose the most green and environmentally friendly water as the solvent. The reason might be the optimum oxygen solubility of water. Besides that, the hydrogen bonding interaction between water and alcohols which could active the alcohols can not be excluded. We attempted different temperature (compare entries 1, 6–8), and 100 °C was optimal. The yield was not significantly improved when the reaction time was prolonged to 48 h, at 30 °C, 60 °C and 90 °C respectively. The reaction yield decreased when the amount of TEMPO was lowered (entries 1, 9–10). Control experiments confirmed that the desired benzaldehyde was not formed in the absence of the catalyst (entries 11), and it gave a moderate yield under air atmosphere. Considering the importance of bases and ligands in the traditional Cu/TEMPO oxidative systems, we added 2.0 mmol KOH in the standard reaction system, however, the reaction efficiency was not obviously enhanced (entry 13). The reason might be the large surface area ratio of CuFe2O4, and make it easy to coordinate with alcohol and TEMPO without bases and ligands (see Scheme 1, plausible mechanism). As a result, the optimized reaction conditions were identified as using CuFe2O4 nanoparticles (10 mol%), TEMPO (1.25 mol%), H2O (2 mL) at 100 °C under dioxygen atmosphere.
| Entry | Solvent | Temp. (°C) | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: benzyl alcohol (1a; 2 mmol), catalyst (0.1 mmol), TEMPO (0.125 mmol), H2O (2 mL), under O2 atmosphere.b Isolated yield.c TEMPO (0.025 mmol).d TEMPO (0.05 mmol).e No catalyst.f Not detected.g The reaction time prolonged to 48 h.h Under air atmosphere.i 2.0 mmol KOH was added. | |||
| 1 | H2O | 100 | 95 |
| 2 | Toluene | 100 | 40 |
| 3 | DMSO | 100 | —f |
| 4 | DMF | 100 | —f |
| 5 | THF | 100 | 92 |
| 6 | H2O | 30 | —f,g |
| 7 | H2O | 60 | 75, 76g |
| 8 | H2O | 90 | 83, 88g |
| 9 | H2O | 100c | 75 |
| 10 | H2O | 100d | 80 |
| 11 | H2O | 100e | —f |
| 12 | H2O | 100 | 64h |
| 13 | H2O | 100 | 87i |
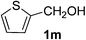
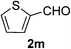
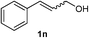
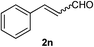
As shown in Table 2, the scope of substrates was investigated, and all the examined substrates provided the corresponding aldehydes in moderate to good yields. For substituted aromatic alcohols, the substrates containing electron-withdrawing groups exhibited higher reactivity than the others. For example, a low yield of the oxidative product (49%) was obtained from (4-nitrophenyl)methanol (Table 2, entry 4). Thiophen-2-ylmethanol (1m) was a good substrate in this transformation, and afforded corresponding aldehyde in 62% yield. In addition, 3-phenylprop-2-en-1-ol (1n) was also tolerated in this transformation, thus generating 2n in 85% yield (Table 2, entry 14). Although primary alcohols showed high reactivity, unfortunately, secondary benzylic alcohols were poor substrates (Table 2, entry 15). The cascade reactions could tolerate some functional groups such as methyl, nitro, ether, C–F bond, C–Cl bond and C–Br bond, which could be used for further transformations at the substituted positions.
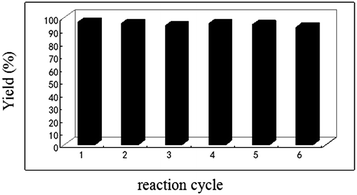
We also studied the recyclability of the catalyst. For this, we investigated the CuFe2O4-catalyzed oxidation of phenylmethanol (1a) to benzaldehyde (2a) under the optimized conditions. After completion of the reaction, the reaction mixture was cooled to room temperature, and the catalyst was magnetically separated from the reaction mixture (Fig. 4), washed ethanol/acetone and dried at 100 °C for 2 h and then used directly for further catalytic reactions. The catalyst showed no significant loss of activity after six cycles as illustrated in Fig. 5. This illustrate a promising approach from sustainable and practical chemistry viewpoints. Then, we investigated the leaching of metal from the CuFe2O4 nanoparticles. After completion of the reaction, the filtrate was tested by the AES (Atomic Emission Spectroscopy) technique, it was found that the leaching of Cu and Fe in three consecutive cycles was ≤0.5 ppm.
 | ||
| Fig. 4 CuFe2O4 after oxidation reaction dispersed (a); magnetic separation of the catalyst at ambient temperature with a Nd–Fe–B based magnet (b). | ||
The control experiments reveal that TEMPO and CuFe2O4 are indispensable parts of this efficacious catalytic system (entries 1 and 9–11, Table 1). Although the mechanism for the present oxidation pathway is not yet clear, according to the previous report a proposal mechanism would be herein presented (Scheme 1). First, TEMPO coordinate to the CuFe2O4 nanoparticles to lead to complex A. Subsequently, species A in combination with alcohols lead to the formation of the species B (in CuFe2O4, Cu is present as Cu(II) in the crystal lattice, so alcohols and TEMPO might be coordinate to Cu(II), which is consistent with the previous reports5b,19a), which was considered as the intermediate species in the present catalytic system. Intramolecular transfer of the α and β-hydrogen would afford aldehydes, CuFe2O4, and TEMPOH. TEMPOH was oxidized by dioxygen to TEMPO and water.19b Further investigations on the more detailed mechanism are ongoing.
Experimental section
General experimental procedures
All reagents and solvents were obtained from commercial suppliers and used without further purification. Flash chromatography was performed on silica gel (200–300 mesh). 1H and 13C NMR data were recorded at 400 and 100 MHz on a BRUKER 400 spectrometer. Chemical shifts (δ) are expressed in parts per million (ppm) coupling constants (J) are in Hz. Proton and carbon magnetic resonance spectra (1H NMR and 13C NMR) were recorded using tetramethylsilane (TMS) in the solvent of CDCl3 as the internal standard (1H NMR: TMS at 0.00 ppm, CDCl3 at 7.28 ppm; 13C NMR: CDCl3 at 77.0 ppm).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 20
1 to 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to afford the target product (2a–2n).
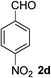
1) to afford the target product (2a–2n).Benzaldehyde (2a)20. Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 95% (201 mg from 216 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.0 (s, 1H), 7.87 (d, 2H, J = 8.0 Hz), 7.66 (t, 1H, J = 8.0 Hz), 7.51 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 192.5, 136.4, 134.5, 129.7, 129.0.
1). Yield 95% (201 mg from 216 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.0 (s, 1H), 7.87 (d, 2H, J = 8.0 Hz), 7.66 (t, 1H, J = 8.0 Hz), 7.51 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 192.5, 136.4, 134.5, 129.7, 129.0.
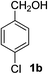
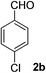
4-Chlorobenzaldehyde (2b)20. Eluent petroleum ether/ethyl acetate (20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 79% (221 mg from 284 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.99 (s, 1H), 7.83 (d, 2H, J = 8.0 Hz), 7.52 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.8, 140.9, 134.7, 130.9, 129.5.
1). Yield 79% (221 mg from 284 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.99 (s, 1H), 7.83 (d, 2H, J = 8.0 Hz), 7.52 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.8, 140.9, 134.7, 130.9, 129.5.
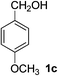
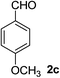
4-Methoxybenzaldehyde (2c)20. Eluent petroleum ether/ethyl acetate (25
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 86% (234 mg from 276 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.89 (s, 1H), 7.84 (d, 2H, J = 8.0 Hz), 7.01 (d, 2H, J = 8.0 Hz), 2.89 (s, 3H). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.8, 164.6, 132.0, 130.0, 114.4, 55.6.
1). Yield 86% (234 mg from 276 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.89 (s, 1H), 7.84 (d, 2H, J = 8.0 Hz), 7.01 (d, 2H, J = 8.0 Hz), 2.89 (s, 3H). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.8, 164.6, 132.0, 130.0, 114.4, 55.6.
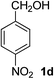
4-Nitrobenzaldehyde (2d)20. Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 49% (148 mg from 306 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.18 (s, 1H), 8.42 (d, 2H, J = 8.0 Hz), 8.10 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.3, 151.2, 140.1, 130.5, 124.3.
1). Yield 49% (148 mg from 306 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.18 (s, 1H), 8.42 (d, 2H, J = 8.0 Hz), 8.10 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.3, 151.2, 140.1, 130.5, 124.3.
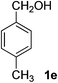
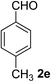
4-Methylbenzaldehyde (2e)20. Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 87% (209 mg from 244 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.96 (s, 1H), 7.82 (d, 2H, J = 8.0 Hz), 7.43 (d, 2H, J = 8.0 Hz), 2.42 (s, 3H). 13C NMR (CDCl3, 200 MHz, ppm) δ 193.1, 145.7, 134.5, 130.2, 127.7, 21.9.
1). Yield 87% (209 mg from 244 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.96 (s, 1H), 7.82 (d, 2H, J = 8.0 Hz), 7.43 (d, 2H, J = 8.0 Hz), 2.42 (s, 3H). 13C NMR (CDCl3, 200 MHz, ppm) δ 193.1, 145.7, 134.5, 130.2, 127.7, 21.9.
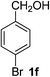
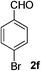
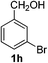
4-Bromobenzaldehyde (2f)20. Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 84% (309 mg from 372 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.97 (s, 1H), 7.54 (d, 2H, J = 8.0 Hz), 7.68 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.0, 135.1, 132.4, 131.0, 129.7.
1). Yield 84% (309 mg from 372 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.97 (s, 1H), 7.54 (d, 2H, J = 8.0 Hz), 7.68 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.0, 135.1, 132.4, 131.0, 129.7.
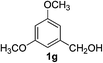
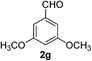
3,5-Dimethoxybenzaldehyde (2g)17. Eluent petroleum ether/ethyl acetate (20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 94% (315 mg from 336 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.88 (s, 1H), 6.99 (s, 2H), 6.68 (s, 1H), 3.83 (s, 6H). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.8, 161.2, 138.4, 107.1, 107.0, 55.6.
1). Yield 94% (315 mg from 336 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.88 (s, 1H), 6.99 (s, 2H), 6.68 (s, 1H), 3.83 (s, 6H). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.8, 161.2, 138.4, 107.1, 107.0, 55.6.
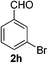
3-Bromobenzaldehyde (2h)21. Eluent petroleum ether/ethyl acetate (25
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 80% (293 mg from 370 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.99 (s, 1H), 8.04 (s, 1H), 7.84 (d, 1H, J = 8.0 Hz), 7.77 (d, 1H, J = 8.0 Hz), 7.45 (t, 1H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.7, 138.0, 137.3, 132.4, 130.6, 128.4, 123.4.
1). Yield 80% (293 mg from 370 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.99 (s, 1H), 8.04 (s, 1H), 7.84 (d, 1H, J = 8.0 Hz), 7.77 (d, 1H, J = 8.0 Hz), 7.45 (t, 1H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 190.7, 138.0, 137.3, 132.4, 130.6, 128.4, 123.4.
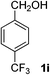
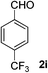
4-(Trifluoromethyl)benzaldehyde (2i). Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 66% (230 mg from 352 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.13 (s, 1H), 8.04 (d, 2H, J = 8.0 Hz), 7.84 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.1, 138.7, 135.8 (d, J = 26.1 Hz), 131.1, 129.9, 126.1 (d, J = 4.1 Hz).
1). Yield 66% (230 mg from 352 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.13 (s, 1H), 8.04 (d, 2H, J = 8.0 Hz), 7.84 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.1, 138.7, 135.8 (d, J = 26.1 Hz), 131.1, 129.9, 126.1 (d, J = 4.1 Hz).
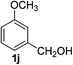
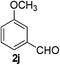
3-Methoxybenzaldehyde (2j)20. Eluent petroleum ether/ethyl acetate (20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 96% (261 mg from 276 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.98 (s, 1H), 7.47–7.45 (m, 2H), 7.40 (s, 1H), 7.20–7.18 (m, 1H), 3.87 (s, 3H). 13C NMR (CDCl3, 200 MHz, ppm) δ 192.1, 160.2, 137.9, 130.1, 123.5, 121.5, 112.2, 55.5.
1). Yield 96% (261 mg from 276 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.98 (s, 1H), 7.47–7.45 (m, 2H), 7.40 (s, 1H), 7.20–7.18 (m, 1H), 3.87 (s, 3H). 13C NMR (CDCl3, 200 MHz, ppm) δ 192.1, 160.2, 137.9, 130.1, 123.5, 121.5, 112.2, 55.5.
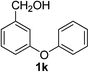
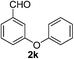
3-Phenoxybenzaldehyde (2k). Eluent petroleum ether/ethyl acetate (20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 72% (285 mg from 400 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.98 (s, 1H), 7.64–7.61 (m, 2H), 7.54–7.49 (m, 2H), 7.42–7.38 (m, 2H), 7.33–7.29 (m, 1H), 7.20 (t, 1H, J = 8.0 Hz), 7.07 (t, 1H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.6, 158.4, 156.2, 138.1, 130.5, 130.1, 124.7, 124.6, 124.2, 119.5, 118.2.
1). Yield 72% (285 mg from 400 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.98 (s, 1H), 7.64–7.61 (m, 2H), 7.54–7.49 (m, 2H), 7.42–7.38 (m, 2H), 7.33–7.29 (m, 1H), 7.20 (t, 1H, J = 8.0 Hz), 7.07 (t, 1H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 191.6, 158.4, 156.2, 138.1, 130.5, 130.1, 124.7, 124.6, 124.2, 119.5, 118.2.
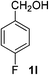
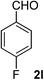
4-Fluorobenzaldehyde (2l). Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 79% (196 mg from 252 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.97 (s, 1H), 7.91 (d, 2H, J = 8.0 Hz), 7.21 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 167.5 (d, J = 10.1 Hz), 132.9, 132.6 (d, J = 26.1 Hz), 116.3 (d, J = 3.1 Hz).
1). Yield 79% (196 mg from 252 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.97 (s, 1H), 7.91 (d, 2H, J = 8.0 Hz), 7.21 (d, 2H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 167.5 (d, J = 10.1 Hz), 132.9, 132.6 (d, J = 26.1 Hz), 116.3 (d, J = 3.1 Hz).
Thiophene-2-carbaldehyde (2m). Eluent petroleum ether/ethyl acetate (20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 62% (139 mg from 228 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.01 (s, 1H), 7.87–7.83 (m, 2H), 7.29 (t, 1H, J = 4.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 183.2, 144.2, 136.2, 135.5, 128.6.
1). Yield 62% (139 mg from 228 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 10.01 (s, 1H), 7.87–7.83 (m, 2H), 7.29 (t, 1H, J = 4.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 183.2, 144.2, 136.2, 135.5, 128.6.
Cinnamaldehyde (2n)17. Eluent petroleum ether/ethyl acetate (30
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Yield 85% (224 mg from 268 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.73 (s, 1H), 9.71 (s, 1H), 7.59–7.57 (m, 4H), 7.47–7.44 (m, 6H), 6.75 (d, 1H, J = 8.0 Hz); 6.70 (d, 1H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 193.7, 152.8, 134.0, 131.3, 129.1, 128.6, 128.5.
1). Yield 85% (224 mg from 268 mg starting material). 1H NMR (CDCl3, 400 MHz, ppm) δ 9.73 (s, 1H), 9.71 (s, 1H), 7.59–7.57 (m, 4H), 7.47–7.44 (m, 6H), 6.75 (d, 1H, J = 8.0 Hz); 6.70 (d, 1H, J = 8.0 Hz). 13C NMR (CDCl3, 200 MHz, ppm) δ 193.7, 152.8, 134.0, 131.3, 129.1, 128.6, 128.5.
Conclusions
In conclusion, we have developed a novel, green and practical magnetic copper ferrite nanoparticle-catalyzed method for aerobic oxidation of primary alcohol to aldehydes. The method is of the following advantages: (a) oxygen-stable and recyclable copper ferrite nanoparticle as the catalyst; (b) environmentally friendly water as the solvent; (c) without addition of any base, ligand or additive; (d) easy workup procedure; (e) outstanding tolerance of functional groups. All these results meet the requirements of green and sustainable chemistry, so the present method will attract much attention in academic and industrial fields. Further studies to catalyze other chemical reaction with copper(II) ferrite magnetic nanoparticles are ongoing.Acknowledgements
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (nos 21302110, 21302109 and 21375075), the Taishan Scholar Foundation of Shandong Province, the Natural Science Foundation of Shandong Province (ZR2013BQ017), the Project of Shandong Province Higher Educational Science and Technology Program (J13LD14), and the Scientific Research Foundation of Qufu Normal University (BSQD 2012021).Notes and references
- R. A. Sheldon, I. W. C. E. Arends and A. Dijksman, Catal. Today, 2000, 57, 157–166 CrossRef.
- (a) M. Hudlicky, Oxidation in Organic Chemistry, ACS Monograph Series, American Chemical Society, Washington, DC, 1990 Search PubMed; (b) J. March, Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, John Wiley & Sons, New York, 4th edn, 1992 Search PubMed.
- For selected examples, see: (a) G. J. ten Brink, I. W. C. E. Arends and R. A. Sheldon, Science., 2000, 287, 1636–1639 CrossRef; (b) M. S. Sigman and D. R. Jensen, Acc. Chem. Res., 2006, 39, 221–229 CrossRef PubMed; (c) C. Liu, S. Tang and A. Lei, Chem. Commun., 2013, 49, 1324–1326 RSC; (d) G. F. Zhang, Y. Wang, X. Wen, C. R. Ding and Y. Li, Chem. Commun., 2012, 48, 2979–2981 RSC.
- For selected examples, see: (a) B. T. Guan, D. Xing, G. X. Cai, X. B. Wan, N. Yu, Z. Fang, L. P. Yang and Z. J. Shi, J. Am. Chem. Soc., 2005, 127, 18004–18005 CrossRef PubMed; (b) H. Miyamura, R. Matsubara, Y. Miyazaki and S. Kobayashi, Angew. Chem., Int. Ed., 2007, 46, 4151–4154 CrossRef PubMed; (c) B. Karimi and F. K. Esfahani, Adv. Synth. Catal., 2012, 354, 1319–1326 CrossRef CAS.
- For selected examples, see: (a) Z. Lu, T. Ladrak, O. Roubeau, J. van der Toorn, S. J. Teat, C. Massera, P. Gamez and J. Reedijk, Dalton Trans., 2009, 3559–3570 RSC; (b) J. M. Hoover, B. L. Ryland and S. S. Stahl, J. Am. Chem. Soc., 2013, 135, 2357–2367 CrossRef CAS PubMed; (c) J. Yu, J. Xu and M. Lu, Appl. Organomet. Chem., 2013, 27, 606–610 CrossRef CAS; (d) P. Karthikeyan, S. A. Aswar, P. N. Muskawar, P. R. Bhagat and S. S. Kumar, Catal. Commun., 2012, 26, 189–193 CrossRef CAS PubMed.
- M. Kamitani, M. Ito, M. Itazaki and H. Nakazawa, Chem. Commun., 2014, 50, 7941–7944 RSC.
- (a) Z. Rong, H. Pan, H. Yan and Y. Zhao, Org. Lett., 2014, 16, 208–211 CrossRef CAS PubMed; (b) R. Liu, X. Liang, C. Dong and X. Q. Hu, J. Am. Chem. Soc., 2004, 126, 4112–4113 CrossRef CAS PubMed; (c) L. Wang, J. Li, H. Yang, Y. Lv and S. Gao, J. Org. Chem., 2012, 77, 790–794 CrossRef CAS PubMed.
- M. Holan and U. Jahn, Org. Lett., 2014, 16, 58–61 CrossRef CAS PubMed.
- (a) R. B. Nasir Baig and R. S. Varma, Chem. Commun., 2013, 49, 752–770 RSC; (b) M. B. Gawande, P. S. Brancoa and R. S. Varma, Chem. Soc. Rev., 2013, 42, 3371–3393 RSC; (c) D. Wang and D. Astruc, Chem. Rev., 2014, 114, 6949–6985 CrossRef CAS PubMed and references therein.
- Y. Wang, X. Wang and M. Antonietti, Angew. Chem., Int. Ed., 2012, 51, 68–89 CrossRef CAS PubMed.
- For selected examples, see: (a) T. Mallat and A. Baiker, Chem. Rev., 2004, 104, 3037–3058 CrossRef CAS PubMed; (b) R. A. Sheldon, I. W. C. E. Arends, G. J. Ten Brink and A. Dijksman, Acc. Chem. Res., 2002, 35, 774–781 CrossRef CAS PubMed; (c) K. Mori, T. Hara, T. Mizugaki, K. Ebitani and K. Kaneda, J. Am. Chem. Soc., 2004, 126, 10657–10666 CrossRef CAS PubMed; (d) J. Chen, Q. H. Zhang, Y. Wang and H. L. Wan, Adv. Synth. Catal., 2008, 350, 453–464 CrossRef CAS; (e) H. Wang, S. X. Deng, Z. R. Shen, J. G. Wang, D. T. Ding and T. H. Chen, Green Chem., 2009, 11, 1499–1502 RSC; (f) G. Wu, X. Wang, N. Guan and L. Li, Appl. Catal., B, 2013, 136–137, 177–185 CrossRef CAS PubMed.
- A. Dhakshinamoorthy, M. Alvaro and H. Garcia, ACS Catal., 2011, 1, 48–53 CrossRef CAS.
- (a) L. M. Rossi, N. J. S. Costa, F. P. Silva and R. Wojcieszak, Green Chem., 2014, 16, 2906–2933 RSC; (b) M. B. Gawande, S. N. Shelke, R. Zboril and R. S. Varma, Acc. Chem. Res., 2014, 47, 1338–1348 CrossRef CAS PubMed; (c) M. B. Gawande and P. S. Branco, Green Chem., 2011, 13, 3355–3359 RSC; (d) M. B. Gawande, V. D. B. Bonifácio, R. Luque, P. S. Brancoa and R. S. Varma, Chem. Soc. Rev., 2013, 42, 5522–5551 RSC; (e) R. Hudson, Y. Feng, R. S. Varma and A. Moores, Green Chem., 2014, 16, 4493–4505 RSC.
- For selected examples, see: (a) S. Yang, C. Wu, H. Zhou, Y. Yang, Y. Zhao, C. Wang, W. Yang and J. Xu, Adv. Synth. Catal., 2013, 355, 53–58 CrossRef CAS; (b) A. Dandia, A. K. Jain and S. Sharma, RSC Adv., 2013, 3, 2924–2934 RSC; (c) S. M. Baghbanian and M. Farhang, RSC Adv., 2014, 4, 11624–11633 RSC; (d) D. Kundu, N. Mukherjee and B. C. Ranu, RSC Adv., 2013, 3, 117–125 RSC; (e) R. Zhang, J. Liu, S. Wang, J. Niu, C. Xia and W. Sun, ChemCatChem, 2011, 3, 146–149 CrossRef CAS; (f) R. Hudson, S. Ishikawa, C.-J. Li and A. Moores, Synlett, 2013, 24, 1637–1642 CrossRef CAS PubMed; (g) P. R. Naveen and B. S. Arulananda, Catal. Commun., 2012, 29, 118–121 CrossRef PubMed; (h) D. Kundu, T. Chatterjee and B. C. Ranu, Adv. Synth. Catal., 2013, 355, 2285–2296 CrossRef CAS; (i) A. S. Kumar, M. Reddy, A. M. Knorn, O. Reiser and B. Sreedhar, Eur. J. Org. Chem., 2013, 4674–4680 CrossRef CAS.
- A. Chanda and V. V. Fokin, Chem. Rev., 2009, 109, 725–748 CrossRef CAS PubMed.
- (a) D. Yang, B. An, W. Wei, M. Jiang, J. You and H. Wang, Tetrahedron, 2014, 70, 3630–3634 CrossRef CAS PubMed; (b) D. Yang, X. Zhu, W. Wei, N. Sun, L. Yuan, M. Jiang, J. You and H. Wang, RSC Adv., 2014, 4, 17832–17839 RSC; (c) D. Yang, X. Zhu, W. Wei, M. Jiang, N. Zhang, D. Ren, J. You and H. Wang, Synlett, 2014, 25, 729–735 CrossRef CAS PubMed.
- B. H. Lipshutz, M. Hageman, J. C. Fennewald, R. Linstadt, E. Slack and K. Voigtritter, Chem. Commun., 2014, 50, 11378–11381 RSC.
- H. Jiao, G. Jiao and J. Wang, Synth. React. Inorg. Met.-Org. Chem., 2013, 43, 131–134 CrossRef CAS.
- (a) G. Zhang, X. Han, Y. Luan, Y. Wang, X. Wen and C. Ding, Chem. Commun., 2013, 49, 7908–7910 RSC; (b) A. Dijksman, I. W. C. E. Arends and R. A. Sheldon, Org. Biomol. Chem., 2003, 1, 3232–3236 RSC.
- Z. Hu and F. M. Kerton, Appl. Catal., A, 2012, 413–414, 332–339 CrossRef CAS PubMed.
- D. Liu, H. Zhou, X. Gu, X. Shen and P. Li, Chin. J. Chem., 2014, 32, 117–122 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details. See DOI: 10.1039/c4ra14152k |
| This journal is © The Royal Society of Chemistry 2014 |