Active phase of highly active Co3O4 catalyst for synthetic natural gas production
Abstract
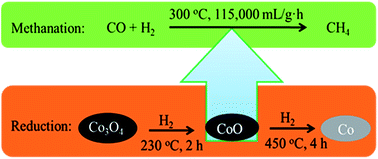
Co3O4 catalyst was studied with respect to methanation in synthetic natural gas (SNG) production. Nanosized Co3O4 particles were prepared using a facile precipitation method. The different chemical valence states of Co species were obtained by adopting various reduction processes prior to methanation. The physicochemical properties of the obtained catalysts were characterized by N2-physisorption, TPR, TEM, XRD, XPS and TG technologies. The catalytic activities for CO methanation were investigated over catalysts with different reduction treatments. The catalyst subjected to a mild reduction process with the appearance of CoO species immediately exhibited 100% conversion of CO with a high space velocity of 11 500 mL g−1 h−1 at 300 °C, 3 MPa. The catalyst without reduction of Co3O4 achieved the same high activity after 3 h exposure to syngas. When Co oxides were fully reduced to metallic Co, they showed no activity for methanation. Combining the results of characterization with evaluation of catalytic performance, it can be concluded that CoO is the active phase for CO methanation. The reduction treatment can improve the stability of the catalyst.

 Please wait while we load your content...
Please wait while we load your content...