Fabrication and characterization of nano-structured ZnS thin films as the buffer layers in solar cells
Alireza Goudarzia,
Azimeh Dorbeygi Namghib and
Chang-Sik Ha*c
aDepartment of Polymer Engineering, Golestan University, Gorgan 49188-88369, Iran
bDepartment of Chemistry, Payam-e Noor University of Sari, Sari, Iran
cDepartment of Polymer Science and Engineering, Pusan National University, Busan 609-735, Republic of Korea. E-mail: csha@pusan.ac.kr; Fax: +82-51-514-4331; Tel: +82-51-510-2407
First published on 4th November 2014
Abstract
High quality nanocrystalline ZnS thin films were deposited on glass substrates by a chemical bath deposition (CBD) method from an aqueous solution containing triethylamine, as a nontoxic complexing agent for zinc ions, at temperatures ranging from 50 to 90 °C. The effects of the deposition time and temperature on thickness as well as structural, morphological and optical properties of the ZnS thin films were examined by X-ray diffraction (XRD), field emission scanning electron microscopy (FE-SEM) and ultraviolet-visible (UV-vis) spectroscopy. Fourier transform infrared spectroscopy (FT-IR) studies were done to investigate the presence of organic and/or other compounds in films. UV-vis spectroscopy data revealed the obtained ZnS films to have an optical band gap ranging from 4.1 to 3.9 eV. XRD results showed that the prepared films were highly crystalline with a cubic zinc blende structure consisting of very small nanocrystallites, 2.0–2.5 nm in size, showing quantum confinement effects.
Introduction
Generally, CdS thin films produced by chemical bath deposition (CBD) are one of the most extensively applied materials as buffer layers in thin film solar cells based on CdTe, CuInS2, CuInSe2 and Cu(In,Ga)Se2(CIGS).1 On the other hand, the CdS buffer layer reduces the light absorption in the absorber layer owing to its narrow band gap, resulting in a decrease in conversion efficiency.2 In addition, Cd is a very toxic element and a large amount of it during chemical bath deposition process of the CdS thin films release in waste and can cause very serious problems for health and environment. Therefore, there is a need for the development of Cd-free buffer layers.Among the many compounds that can be used as the buffer layer, ZnS, a wide band gap semiconductor (3.6 eV) with much less toxicity, can be a suitable replacement for CdS buffer layer in solar cells. Bhattacharya et al. reported the high conversion efficiency of 18.5% for CIGS-based thin film solar cells using ZnS thin films as the buffer layer.2 In recent years, extensive research has been carried out on the deposition of ZnS thin films. Several physical and chemical methods have been used to prepare ZnS thin films and particles, such as evaporation,3 atomic layer deposition,4 sol gel,5 sputtering,6 spray pyrolysis,7 chemical vapor deposition,8 molecular beam epitaxy, physical vapor deposition,9 and CBD.10
Among these, CBD is used widely to prepare the semiconductor thin films because of its low cost and large area capability. In addition, CBD is a simple and convenient method because it is normally carried out at low temperatures under atmospheric pressure.11 Most studies of ZnS thin films fabricated by CBD are based on the use of ammonia and/or hydrazine as a complexing agent for zinc ions.10,12–23 Hydrazine and ammonia, however, are highly volatile, flammable, toxic, environmentally harmful, and carcinogenic materials.
A previous study reported the preparation of an ammonia and hydrazine free CBD-ZnS buffer layer using Na2EDTA as a non toxic complexing agent.24 Since then, several groups have examined CBD-ZnS thin films using less toxic complexing agents.2,25–27 Recently, S. W. Shin et al. reported an improvement in the rate of ZnS thin film growth using mixed complexing agents of Na2EDTA and hexamethylenetetramine (HMTA).2 Although the growth rate of the ZnS films prepared by S. W. Shin et al. was higher than that of previous works, the films exhibited very poor crystallinity with a hexagonal phase structure. The hexagonal structure of ZnS is stable at high temperatures but unstable at room temperature. F. Gode,28 reported the preparation of CBD-ZnS thin films using triethanolamine and tri-sodium citrate as less-toxic complexing agents in an ammonia solution.
S. W. Shin et al.25 prepared CBD-ZnS thin films using Na2EDTA and Na3-citrate as complexing agents in an ammonia solution, as an alkaline medium, at 80 °C and examined the effects of annealing on the ZnS films structure. In the fabrication of CIGS-based thin film solar cells, the CBD-buffer layers are normally annealed to improve the crystallinity of the buffer layer and adhesion to the absorber layer.29,30 Although S. W. Shin et al. used Na2EDTA and Na3-citrate as the less-toxic complexing agents, ammonia is still a toxic and harmful agent. In addition, the as-prepared ZnS buffer layers prepared by S. W. Shin et al. had an amorphous nature, but the crystallinity of the layers was improved by annealing at 500 °C.
In this study, well-crystallized, highly transparent and homogenous ZnS thin films were deposited at low temperatures using triethylamine as a non-toxic complexing agent by a simple CBD method. The crystallinity, transmission, band gap energy, and thickness of the ZnS thin films were improved considerably compared to other studies.
Table 1 lists the deposition conditions, transmission values, and crystalline phase structure of the prepared CBD-ZnS thin films using less toxic complexing agents than those reported in the literature and compared with this study.
| Zn source | Source | Complexing agents | pH | Transmission average in the visible region | Crystallinity & crystalline phase | Ref. |
|---|---|---|---|---|---|---|
| a TAA: thioacetamide, TU: thiourea and HMDA: hexamethylendiamine. The pH adjusted by the ammonia solution. | ||||||
| Zn (CH3COO)2 | TAA | Na2EDTA | 6 | 70% | Relatively poor-cubic | 24 |
| Zn (CH3COO)2 | TU | Na3-citrate | 10 | 65–75% | Mixture of ZnS & ZnO hexagonal | 27 |
| Zn (CH3COO)2 | TU | Na3-citrate & triethanolamine | 8.2–8.6 | Not reported | Hexagonal | 26 |
| Zn (CH3COO)2 | TAA | Na2EDTA | 4 | Not reported | Poor-hexagonal | 2 |
| Zn (CH3COO)2 | TAA | HMDA | 4 | 70–80% | Poor-hexagonal | 2 |
| Zn (CH3COO)2 | TAA | Na3-citrate & Na2EDTA | 10a | 70% | Amorphous or poor-hexagonal | 25 |
| Zn (CH3COO)2 | TAA | Triethylamine | 5.6 | More than 80% | Very well-cubic | This work |
Experimental
All chemicals used in the experiments were purchased from Merck Company, with the exception of thioacetamide, which was supplied by Sigma-Aldrich Company and used as received. The nanocrystalline ZnS thin films were grown on commercial microscope slide glass substrates using a triethylamine solution as a nontoxic complexing agent for zinc ions, along with zinc acetate and thioacetamide solutions as the zinc and sulfide ions source, respectively. Table 2 lists the concentrations in terms of mol L−1 and the molar ratios of the main species in the precursor solution. The precursor solutions were prepared as follows: 12 mL of a 1 M zinc acetate [Zn (CH3COO)2·2H2O] solution was mixed with 24 mL of a 0.5 M triethylamine [(C2H5)3N] solution. A few drops of a 1 M sulfuric acid solution were then added to remove the formed precipitate. In the next step, 30 mL of a 0.8 M thioacetamide [CH3CSNH2] solution was added. Finally, deionized water was added to the solution to make a total volume of 100 mL. At this step, the final pH of the as-prepared bath solution used in CBD was approximately 5.7 ± 0.1 and there was no pH adjustment of the bath solution. Film deposition was carried out at different temperatures ranging from 50 to 90 °C for durations ranging from 1 to 4 h.| Species | Zn2+ | TAA | Triethylamine |
|---|---|---|---|
| Conc.(mol L−1) | 0.12 | 0.24 | 0.12 |
| Molar ratio (relative to Zn2+) | 1 | 2 | 1 |
Prior to film deposition, the glass substrates were cleaned using detergent and deionized water. The substrates were dried for one hour in an oven at 80 °C. The cleaned substrates were immersed vertically into the reaction vessel, which was then placed in a thermostat bath at the desired temperature (50, 70, and 90 °C). The obtained ZnS films were washed with deionized water, dried in air at room temperature, and then characterized.
Characterization
The optical absorbance (A) spectra of films were obtained using a Shimadzu UV-visible spectrophotometer over the wavelength range, 300–800 nm. X-ray diffraction (XRD, Philips PW 1800) was performed to characterize the phase and structure of the ZnS films using CuKα radiation (40 kV and 40 mA) at angles ranging from 20° to 80° 2θ. The surface morphology of the ZnS films was observed by field emission scanning electron microscopy (FE-SEM; Hitachi S-4160) under an acceleration voltage of 15 KV. The band gap values were estimated from the UV-visible spectroscopic data. All measurements were carried out at room temperature. To investigate the presence of organic and/or other compounds in films, a Fourier transform infrared (FT-IR) spectrum was recorded using a Perkin Elmer-Spectrum RX-IFTIR spectrometer in the range of 300–4000 cm−1. The film thickness was determined using an α-step profilometer (Dektak,3 Veeco Inst.)Results and discussion
The mechanism of the CBD-ZnS thin films already has been reported.24,31 In CBD method, a complexing agent should be used to form a stable complex with metallic ions to prevent the fast reaction between Zn2+ and S2− ions. The fast reaction between S2− and Zn2+ ions lead to homogeneous precipitation along with aggregation of the particles instead of heterogenous reaction (film formation) as well as a weak adherence of the particles to the substrates. Therefore, using a good complexing agent for zinc ions is essential to control the film deposition and obtain films with high quality.In this study, triethylamine (C6H15N) was employed as a non-toxic complexing agent for Zn2+ ions and thioacetamide (CH3CSNH2) was used as S2− ions source. In the presence of triethylamine, the zinc complex [Zn(C6H15N)]2+ is formed by the following reaction:
| Zn2+ + C6H15N → [Zn (C6H15N)]2+ | (1) |
Due to the presence of triethylamine, the zinc ions exist mainly as [Zn (C6H15N)]2+ in the reaction solution and the required Zn2+ ions are released by decomposition of [Zn (C6H15N)]2+ complex, resulting in a slow release of the Zn2+ ions in the reaction solution as follows:
| [Zn (C6H15N)]2+ → Zn2+ + C6H15N | (2) |
Decomposition of thioacetamide to H2S and then hydrolyses of H2S in two steps produce the S2− ions necessary for the reaction with the released Zn2+ ions by the following chemical reactions:
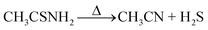 | (3) |
| H2S + H2O ⇄ HS− + H30+ | (4) |
| HS− + H2O ⇄ S2− + H3O+ | (5) |
The produced zinc and sulfide ions from reactions (2) and (5) migrate to the substrate surface and react to form the ZnS films. When the ionic product of Zn2+ and S2− exceeds the solubility product of ZnS (5.0 × 1024), the formation of ZnS can occur either in the solution (as the powder) or at the substrate surface (as the film) according to the following reaction:
| Zn2+ + S2− → ZnS | (6) |
Different parameters can affect the quality of the ZnS thin films prepared by CBD method. One of the most important parameters is the pH of the reaction solution. According to the reaction mechanism due to the presence of H3O+ ions in the equilibrium reactions of steps (4) and (5), variations in the pH of the bath solution can change S2− ions concentration in the reaction solution according to Le Chatelier's principle and affect the growth of the films. In previous studies of ZnS thin film preparation, the final pH of the bath was set using acidic or alkaline dilute solutions.2,25 In this study, however, after mixing the desired solutions, because of employing the suitable concentrations as well as molar ratios of the main species in the chemical bath solution (Table 2), final pH of the mixed solutions was spontaneously adjusted to be 5.7, and used directly for film deposition. The ZnS films deposited from this solution had very good quality in terms of adherence, particles compactness, thickness, crystallinity and transparency, and therefore the aforementioned procedure for the preparation of the bath solution could generate a favorite pH without adding any basic or acidic solutions for adjusting of the pH of the reaction solution. The omission of the pH adjusting process of the bath solution is another advantage of this study, particularly on an industrial scale.
Fig. 1 indicates the variation of thickness as a function of deposition time for the ZnS films prepared at 70 °C. As shown in Fig. 1, the rate of film growth is almost constant during 6 h deposition time. With increasing deposition time, the film thickness increases with an approximately growth rate of 23.5 nm h−1 and reaches to 150 nm after 6 h deposition time.
Fig. 2 shows the changes of the thickness with deposition temperature for the ZnS films prepared during 4 h deposition time. It can be seen that the growth rate of the prepared ZnS film at temperatures of 50, 70 and 90 °C is 9.5, 23.5 and 50 nm h−1, respectively. Increasing growth rate of the ZnS films with increasing the temperature can be related to increasing S2− ions concentration in the reaction solution due to the increase in the rate of thioacetamide decomposition (reaction (3)). According to the reaction (3), decomposition of thioacetamide is an endothermic reaction, and thus the increase in the temperature can increase the production rate of H2S, resulting in an increase in S2− ions concentration for reaction with Zn2+ to form ZnS.
 | ||
| Fig. 2 Variation of ZnS films thickness prepared at 4 hours deposition time as a function of deposition temperature. | ||
Fig. 3 shows XRD patterns of the ZnS thin films prepared at 50, 70, and 90 °C. The XRD peaks at 2θ = 29.2, 48.4, and 57.5°, were attributed to the (111), (220), and (311) lattice planes of the cubic zinc blend structure (JCPDS, no. 05-0566), respectively. No characteristic peaks for impurities, such as ZnO, Zn (OH)2, and/or other compounds, were detected. The observed strong XRD peaks suggest that the well-crystallized ZnS films with a polycrystalline structure had been obtained. Using the Debye Scherrer formula,24 and on the basis of the full width at half-maximum (FWHM) of the (111) peak, the nanocrystallite size of the prepared ZnS thin films was estimated to be 2.0, 2.2, and 2.5 nm at 50, 70, and 90 °C, respectively. As shown in Fig. 3, with increasing deposition temperature from 50 to 90 °C, the intensity of the peaks increased slightly and became narrower, which indicates an increase in nanocrystallite size and an improvement in the crystallinity of the fabricated ZnS films at upper temperatures.
Fig. 4 shows the effects of the deposition time on the surface morphology of the ZnS thin films prepared at 70 °C, which was recorded by field emission scanning electron microscopy (FE-SEM). As shown in Fig. 4, for all deposition times (2, 4 and 6 h), the film compactness was high, the surface uniformity was good, and the particles had a narrow size distribution. In addition, the film surface were free of cracks, pinholes or pits, indicating that homogeneous deposition had taken place during ZnS film formation.
 | ||
| Fig. 4 Effect of the deposition time on the surface morphology of the prepared ZnS films at 70 °C ((a) 2 h, (b) 4 h, and (c) 6 h). | ||
Fig. 5 presents FE-SEM images of the surface morphology of the ZnS films prepared at different temperatures for 4 hours deposition time. As shown in images (a–c) in Fig. 5, the particle size of the films increased with increasing deposition temperature from 50 to 90 °C. The observed difference in particles size, compactness and thickness of the ZnS films can be attributed to the difference in the growth rate of the prepared films at different temperatures (50, 70 and 90 °C) as indicated in Table 3. Change in deposition temperature can strongly affect kinetic energy of the ions (S2− and Zn2+) for reaction as well as deposition rate of the films. In this work, thioacetamide was used as the S2− ions source. Since the decomposition of thioacetamide is an endothermic reaction (reaction (3)), the increase in the temperature from 50 to 70 and 90 °C can increase the rate of thioacetamide decomposition as well as S2− ions concentration in the reaction solution. According to the reaction (6), increase or decrease in S2− ions concentration can directly affect the formation rate of ZnS and as a result on particles size, thickness and compactness of the films.
 | ||
| Fig. 5 Effect of the deposition temperature on the surface morphology of the prepared ZnS films during 4 h deposition at ((a) 50, (b) 70, and (c) 90 °C). | ||
| Deposition temp. (°C) | Thickness (nm) | Deposition rate (nm h−1) | Band energy (eV) | Particle size (nm) |
|---|---|---|---|---|
| 50 | 38 | 9.5 | 4.07 | 2.0 |
| 70 | 95 | 23.5 | 3.92 | 2.2 |
| 90 | 200 | 50 | 3.88 | 2.5 |
In Fig. 5, the ZnS film prepared at 70 °C has the best compactness, thus it seems that besides the suitable kinetic energy of ions (S2− and Zn2+), the produced S2− ions at this temperature have the most suitable concentration for the film deposition. This means that the deposition rate of 25 nm h−1 of the ZnS particles at 70 °C was neither too much to observe the particles aggregation and nor too low to reduce the film compactness or pine hole formation.
FE-SEM images (a–c) in Fig. 5 show that the mean size of the observable particles were approximately 30–50 nm for the prepared ZnS films during 4 h deposition at 50 (a) and 70 °C (b), while that for ZnS films deposited at 90 °C is ca. 100 nm, though it was hard to distinguish the size difference of (a) and (b). The mean nanocrystallite size of these films calculated from the XRD pattern (Fig. 5) using Debye Scherrer formula were, however, approximately 2.0, 2.2 and 2.5 nm, respectively.
This means that each particle observed in FE-SEM images (a–c) in Fig. 5 is polycrystalline. Fig. 6 presents a cross-sectional FE-SEM image of the ZnS thin film prepared at 90 °C for 4 h. The film thickness is approximately 200 nm, which was confirmed by thickness measurement of the film by an Alfa-step profilmeter.
 | ||
| Fig. 6 The cross sectional view FE-SEM image of the ZnS thin film prepared at 90 °C during 4 h with thickness of about 200 nm. | ||
Fig. 7a shows the optical transmission spectra of bare glass and the ZnS thin films deposited on the glass substrates at 50, 70 and 90 °C for 4 h. The obtained films were highly transparent (>85%) in the visible region. The high transmission values and sharp absorption edges of the films indicate a narrow particle size distribution as well as homogeneous deposition. This high quality assignment of the prepared films was confirmed by FE-SEM images.
Basically, thinner films have higher transmittance at shorter wavelengths, which is desirable for enhancing the short-circuit current and solar cell efficiency because an increasing light transmittance to the junction will generate more electron hole pairs. On the other hand, thicker films could avoid the damage caused by ZnO sputtering deposition in the solar cell fabrication process. Therefore, the preparation of ZnS buffer layers with high transmission and optimal thickness is essential for improving the overall efficiency of the cells.
As shown in Fig. 5a, the as-prepared ZnS thin films were highly transparent (more than 85–90%) in the visible range, which is suitable for increasing the short-circuit current and solar cell performance. In addition, as indicated in Fig. 2, the films have a noticeable thickness, which could avoid the damages caused by the ZnO sputtering deposition. For example, the as-prepared ZnS film with a thickness of 200 nm (Fig. 6 and 7a) had more than 85% transmission in the visible range. As shown in Fig. 7a, with increasing deposition temperature, the absorption edge of the films clearly shifts towards higher wavelengths (red shift). This red shift was assigned to a decrease in the band gap because of the increasing particle size of the films prepared at higher temperatures.24
The optical absorption coefficients (α) were estimated using the following equation: αhν = k(hν − Eg)n/2, where k is a constant, Eg is the optical band gap, and n is a constant with discrete values, such as 1, 3, 4 or more. For a direct and allowed transitions n = 1 whereas n = 3 in a direct but forbidden transition. For indirect and allowed transitions, n = 4 and n ≥ 6 in indirect and forbidden transitions. As shown in Fig. 7b, the plot of (αhν)2 against hν was linear at the absorption edge. This means that the transition modes in these films have a direct and allowed nature. The band gap, Eg, was estimated from an extrapolation of the straight-line portion of the (αhν)2 vs. hν curve to zero absorption coefficient value. Table 3 lists the band gap values and nanocrystallite sizes. These values were higher than the band gap of bulk ZnS (Eg, bulk = 3.6 eV). This difference can be attributed to the quantum confinement effect in the prepared ZnS nanocrystallite.32
In semiconductor nanoparticles, electronic and optical properties are influenced by quantum size confinement effect. This effect becomes significant, especially when the crystal size is comparable to or smaller than the Bohr exciton radius. The Bohr radius of ZnS is approximately 2.5 nm.33 In the prepared ZnS films, depending upon the deposition temperature, the nanocrystallite size, determined by Scherrer equation, ranged from 2.0 to 2.5 nm, and the absorbance edges varied from 4.07 to 3.88 eV. The observed blue shift in the absorption edge of these films compared with bulk ZnS (Eg, bulk = 3.6 eV) is attributed to the quantum confinement effect in ZnS nanocrystallites.34–36
As shown in Fig. 7b and Table 3, all the ZnS films have a large band gap, ranging from 3.9 to 4.1 eV, which is also desirable because the band gap of the buffer layer should be as large as possible to allow as many photons as possible to reach the p–n junction and increase the possibility of absorption.33 In addition, the increasing band gap can increase the short-circuit current of the cell to achieve thin film solar cells with higher conversion efficiency.
To investigate the presence of organic and/or other compounds such as Zn(OH)2 and ZnO as impurities in the prepared ZnS thin films, we obtained FT-IR spectrum in transmittance mode of a typical prepared ZnS film. The previous works reported the presence of isothiocyanate groups (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), Zn–O, Zn–OH bonds and adsorbed H2O in CBD-ZnS thin film.37–39 Zn–OH bending and stretching modes are observed at 648 and 1115 cm−1, respectively,40,41 Zn–O bonds in ZnO powder is appeared at 430 cm−1 with high intensity.42 Isothiocyanate groups (–N
S), Zn–O, Zn–OH bonds and adsorbed H2O in CBD-ZnS thin film.37–39 Zn–OH bending and stretching modes are observed at 648 and 1115 cm−1, respectively,40,41 Zn–O bonds in ZnO powder is appeared at 430 cm−1 with high intensity.42 Isothiocyanate groups (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S) exhibit a strong peak in the 2000–2273 cm−1 domain,43 whereas nanosized Zn–S particles show a strong peak at 315 cm−1.44,45 In Fig. 8, the FT-IR spectrum of one of the ZnS thin films prepared in this work, besides the Zn–S peak at 318 cm−1, no other peaks due to impurities are observed except for a very weak peak at 1564 cm−1 and a broad weak peak at 3000–3600 cm−1 that must be related to the stretching and bending modes of trace amounts of adsorbed water on the film.40
S) exhibit a strong peak in the 2000–2273 cm−1 domain,43 whereas nanosized Zn–S particles show a strong peak at 315 cm−1.44,45 In Fig. 8, the FT-IR spectrum of one of the ZnS thin films prepared in this work, besides the Zn–S peak at 318 cm−1, no other peaks due to impurities are observed except for a very weak peak at 1564 cm−1 and a broad weak peak at 3000–3600 cm−1 that must be related to the stretching and bending modes of trace amounts of adsorbed water on the film.40
Conclusion
High quality ZnS films in terms of transmission, crystallinity, structure, thickness, and particle compactness, which are desirable for buffer layers in solar cells, were fabricated using triethylamine as a nontoxic complexing agent for zinc ions using a simple chemical bath deposition method at low temperatures. The film thickness can be controlled from 38 to 200 nm by adjusting deposition time or temperature. The deposited ZnS films also had very strong adherence to the substrate. FE-SEM images indicated that the temperature and deposition time can be two important factors for controlling the film growth process. XRD data showed that the obtained films were well-crystallized and consisted of very small grains, ranging in size from 2.0 to 2.5 nm, and showing quantum size effects.Acknowledgements
We thank the National Research Foundation of Korea (NRF) Grant funded by the Ministry of Science, ICT & Future Planning, Korea; Pioneer Research Center Program (2010-0019308/2010-0019482); Brain Korea 21 Plus Program (21A2013800002).References
- S. D. Sartale, B. R. Sankapal, M. Lux-Steiner and A. Ennaoui, Thin Solid Films, 2005, 480, 168 CrossRef PubMed.
- S. W. Shin, S. R. Kang, K. V. Gurav, J. h. Yun, J. H. Moon, J. Y. Lee and J. H. Kim, Sol. Energy, 2011, 85, 2903 CrossRef CAS PubMed.
- H. Eilers, Mater. Lett., 2008, 62, 967 CrossRef CAS PubMed.
- Y. S. Kim and S. J. Yun, Appl. Surf. Sci., 2004, 229, 105 CrossRef CAS PubMed.
- Y. Kavanagh, M. J. Alam and D. C. Cameron, Thin Solid Films, 2004, 447, 85 CrossRef PubMed.
- S. Bonilla and E. A. Dalchiele, Thin Solid Films, 1991, 204, 397 CrossRef CAS.
- R. R. Chamberlin and J. S. Skarman, J. Electrochem. Soc., 1966, 113, 86 CrossRef CAS PubMed.
- H. Uda, S. lkegami and H. Sonomura, Jpn. J. Appl. Phys., 1990, 29, 30 CrossRef CAS.
- M. R. Khanlary and N. Ahmadi, Iran. J. Phys. Res., 2011, 10, 2011 Search PubMed.
- L. Wei-Long, Y. Chang-Siao, H. Shu-Huei, C. Wen-Jauh and F. Chi-Lon, Appl. Surf. Sci., 2013, 264, 213 CrossRef PubMed.
- G. Hodes, Chemical Solution Deposition of Semiconductor Films, Marcel Dekker Inc., New York, 2003 Search PubMed.
- T. Ben Nasr, N. Kamoun, M. Kanzari and R. Bennaceur, Thin Solid Films, 2006, 500, 4 CrossRef CAS PubMed.
- Z. Q. Bian, X. B. Xu, J. B. Chu, Z. Sun, Y. W. Chen and S. M. Huang, Surf. Rev. Lett., 2008, 15, 821 CrossRef CAS.
- S. M. Salim, A. H. Eid, A. M. Salem and H.M. Abou El-khair, Surf. Interface Anal., 2012, 44, 1214 CrossRef CAS.
- L. Zhou, N. Tang, S. Wu, X. Hu and Y. Xue, Phys. Procedia, 2011, 22(354), 16 Search PubMed.
- Z. Y. Zhong, E. S. Cho and S. J. Kwon, Mater. Chem. Phys., 2012, 135, 287 CrossRef CAS PubMed.
- S. Tec-Yam, J. Rojas, V. Rejón and A. I. Oliva, Mater. Chem. Phys., 2012, 136, 386 CrossRef CAS PubMed.
- M. Ladar, E. J. Popovici, I. Baldea, R. Grecu and E. Indrea, J. Alloys Compd., 2007, 434, 697 CrossRef PubMed.
- I. O. Oladeji and L. Chow, Thin Solid Films, 2005, 474, 77 CrossRef CAS PubMed.
- S. D. Sartale, B. R. Sankapal, M. Lux-Steiner and A. Ennaoui, Thin Solid Films, 2005, 480, 168 CrossRef PubMed.
- L. V. Makhova, I. Konovalov, R. Szargan, N. Aschkenov, M. Schubert and T. Chasse, Phys. Status Solidi, 2005, 2, 1206 CrossRef CAS.
- C. Hubert, N. Naghavi, B. Canava, A. Etcheberry and D. Lincot, Thin Solid Films, 2007, 515, 6032 CrossRef CAS PubMed.
- R. B. Kale, C. D. Lokhande, R. S. Mane and H. Sung-Hwan, Appl. Surf. Sci., 2006, 252, 5768 CrossRef CAS PubMed.
- A. Goudarzi, G. Motedayen Aval, R. Sahraei and H. Ahmadpoor, Thin Solid Films, 2008, 516, 4953 CrossRef CAS PubMed.
- W. Shin, S. R. Kang, J. H. Yun, A. V. Moholkar, J. Y. Lee and J. H. Kim, Sol. Energy Mater. Sol. Cells, 2011, 95, 856 CrossRef PubMed.
- M. Dhanam and B. Kavitha, Chalcogenide Lett., 2009, 6, 299 CAS.
- S. W. Shin, H. P. Oh, S. M. Parwar, J. H. Moon and J. H. Kim, J. Nanosci. Nanotechnol., 2010, 10, 3686 CrossRef CAS PubMed.
- F. Gode, Phys. B, 2011, 406, 1653 CrossRef CAS PubMed.
- L. Qi, G. Mao and J. Ao, Appl. Surf. Sci., 2008, 254, 5711 CrossRef CAS PubMed.
- I. Repinsl, B. Contreras, C. Egaas, J. Dehart, C. Scharf, B. Perkins and R. Noufi, Progress in Photovoltaics: Research and Applications, 2008, 16, 235 CrossRef.
- P. Roy, J. R. Ota and S. K. Srivastava, Thin Solid Films, 2006, 515, 1912 CrossRef CAS PubMed.
- R. Sahraei, A. Daneshfar, A. Goudarzi, S. Abbasi, M. H. Majles Ara and F. Rahimi, J. Mater. Sci.: Mater. Electron., 2013, 24, 260 CrossRef CAS.
- T. L. Chu and S. S. Chu, Solid-State Electron., 1995, 38, 533 CrossRef CAS.
- R. Rossetti, R. Hull, J. M. Gibson and L. E. Brus, J. Chem. Phys., 1985, 82, 552 CrossRef CAS PubMed.
- A. Goudarzi, G. Motedayen Aval, S. S. Park, M. C. Choi, R. Sahraei, M. Habib Ullah, A. Avanes and C. S. Ha, Chem. Mater., 2009, 21, 2375 CrossRef CAS.
- R. S. S. Saravanan, D. Pukazhselvan and C. K. Mahadevan, J. Alloys Compd., 2012, 517, 139 CrossRef CAS PubMed.
- T. Nakada, K. Furumi, M. Mizutani, Y. Hagiwara and A. Kunioka, Technical Digest of the 11th International PhotoVoltaic Science and Engineering Conference, Hokkaido, Japan, 20–24 September 1999 Search PubMed.
- B. Mokili, Y. Charreire, R. Cortes and D. Lincot, Thin Solid Films, 1996, 288, 21 CrossRef CAS.
- H. El Maliki, J. C. Bernede, S. Marsillac, J. Pinel, X. Castel and J. Pouzet, Appl. Surf. Sci., 2003, 205, 65 CrossRef CAS.
- F. Wegmuller, J. Colloid Interface Sci., 1987, 116, 312 CrossRef.
- J. W. Kauffman, R. H. Hauge and J. L. Margrave, J. Phys. Chem., 1985, 89, 3541 CrossRef CAS.
- J. R. Ferraro, Low-frequency vibrations of inorganic and coordination compounds, Plenum, New York, 1971 Search PubMed.
- R. Bayon, C. Maffiotte and J. Herrero, Thin Solid Films, 1999, 353, 100 CrossRef CAS.
- X. Jianfeng, M. Haitao and S. Yue, J. Vac. Sci. Technol., 1997, 15, 1465 CrossRef.
- A. Mandal, S. Mitra, A. Datta, S. Banerjee, S. Dhara and D. Chakravorty, J. Appl. Phys., 2012, 112, 074321 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2014 |




