A new approach to pyridines through the reactions of methyl ketones with 1,2,4-triazines†
Shu-Wen Wang,
Wei-Si Guo,
Li-Rong Wen* and
Ming Li*
State Key Laboratory Base of Eco-Chemical Engineering, College of Chemistry and Molecular Engineering, Qingdao University of Science and Technology, Qingdao 266042, P. R. China. E-mail: wenlirong@qust.edu.cn; liming928@qust.edu.cn
First published on 29th October 2014
Abstract
A new route to prepare pyridine derivatives based on inverse electron demand Diels–Alder/retro-Diels–Alder reactions of ketones with 1,2,4-triazines is reported. It is the first time using methyl ketones directly as a dienophile to react with 1,2,4-triazines without enamine intermediates, which is complementary to the classical Boger reaction.
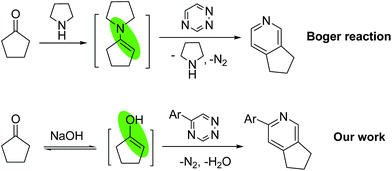
Among the nitrogen heterocycles, pyridine is one of the most prevalent skeletons found in natural products, pharmaceuticals, agrochemicals, and functional materials.1 As a consequence, pyridine synthesis has been a hot research field for a long time. Besides the transition metal-catalyzed methods to construct pyridine derivatives,2 the traditional condensations are still the most commonly used methods because of reliability and simplicity.3 Among the known synthetic methods, the inverse electron demand Diels–Alder/retro-Diels–Alder reactions of 1,2,4-triazines with electron-rich enamines across C3/C6 of the 1,2,4-triazine nucleus to yield pyridines, developed by Boger4 and Taylor,5 are considered to be a powerful synthetic approach. However, the widely used classical methodologies have the same limitations, which required generating an enamine intermediate and long reaction time (Scheme 1).
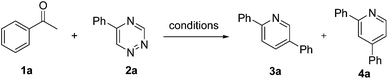
We speculate that the enols tautomerized from ketones under basic conditions could be used directly as dienophiles in the inverse electron demand Diels–Alder reactions. Thus, the reaction of acetophenone 1a and 5-phenyl-1,2,4-triazine 2a was investigated in the presence of NaOH in refluxing ethanol. To our delight, the expected pyridine products 2,5-diphenylpyridine 3a and 2,4-dipenylpyridine 4a were formed. To the best of our knowledge, the reactions of acetophenones and cyclic ketones as potential dienophiles with 1,2,4-triazines directly to synthesize the pyridine derivatives in the presence of NaOH have not been studied. In continuation of our research interests in exploring useful heterocyclic compounds,6 we report herein a convenient protocol to synthesize the pyridine derivatives through the reactions of methyl ketones with 1,2,4-triazines directly without formation of enamine intermediate (Scheme 1).
The optimization of the reaction conditions was carried out with acetophenone 1a and 5-phenyl-1,2,4-triazine 2a as the model substrates (Table 1). Initially, 2,5-diphenylpyridine 3a and 2,4-dipenylpyridine 4a were obtained in a low total yield of 30% in EtOH at 40 °C with NaOH (1.0 equiv.) as the base (Table 1, entry 1). Delightedly, the total yield increased with the raise of temperature, and achieved 75% yield at reflux temperature (Table 1, entry 2). The utility of other solvents, such as CHCl3, THF, CH3CN, and DMF was all unsatisfactory (Table 1, entries 3–6). Next, other bases were tested, and the results revealed that K2CO3 gave lower yield than NaOH (Table 1, entry 7). In the presence of Et3N or DABCO, the reactions were sluggish, and no desired products were obtained (Table 1, entries 8 and 9). EtONa and t-BuONa provided the similar yields with NaOH (Table 1, entries 10 and 11). Finally, the equivalent of the base was also tested (Table 1, entries 12–14). The control experiment suggested that without any base the reaction did not take place even in refluxing ethanol (Table 1, entry 15). In addition, regarding methyl ketones could also tautomerize to the enols under acid conditions, some acids such as HOAc and HCl were employed to catalyze this reaction. However, the reaction did not occur under HOAc conditions; whereas 1,2,4-triazine hydrochloride was formed exclusively in the presence of HCl. Therefore, the optimum reaction condition was using NaOH (1.5 equiv.) as the base in refluxing ethanol (Table 1, entry 13). In particular, with the optimum conditions the obtained two products were easy to be separated by flash chromatography. Interestingly, when the reaction of 1a with 2a was performed in the traditional Boger's conditions (pyrrolidine as catalyst in CHCl3 at 45 °C), the reaction did not proceed.
| Entry | Base (equiv.) | Solvent | Temp (°C) | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|
| a 1a (1.0 mmol) and 2a (1.0 mmol) was used.b Total yield of 3a and 4a.c No base.d No reaction. | |||||
| 1 | NaOH (1.0) | EtOH | 40 | 12 | 30 |
| 2 | NaOH (1.0) | EtOH | Reflux | 4 | 75 |
| 3 | NaOH (1.0) | CHCl3 | Reflux | 4 | Trace |
| 4 | NaOH (1.0) | THF | Reflux | 4 | Trace |
| 5 | NaOH (1.0) | CH3CN | Reflux | 4 | 65 |
| 6 | NaOH (1.0) | DMF | 100 | 4 | 45 |
| 7 | K2CO3 (1.0) | EtOH | Reflux | 4 | 42 |
| 8 | Et3N (1.0) | EtOH | Reflux | 4 | NRd |
| 9 | DABCO (1.0) | EtOH | Reflux | 4 | NRd |
| 10 | EtONa (1.0) | EtOH | Reflux | 4 | 73 |
| 11 | t-BuONa (1.0) | EtOH | Reflux | 4 | 74 |
| 12 | NaOH (0.5) | EtOH | Reflux | 4 | 15 |
| 13 | NaOH (1.5) | EtOH | Reflux | 1.5 | 85 |
| 14 | NaOH (2.0) | EtOH | Reflux | 1.5 | 85 |
| 15 | —c | EtOH | Reflux | 12 | NRd |
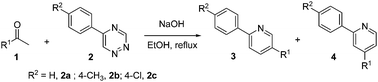
To test the generality of this reaction, various methyl ketones 1a–1i and 5-aryl-1,2,4-triazines 2a–2c were examined under the optimized conditions. As shown in Table 2, the substituents on the aromatic group of triazines had no influence on the reactions (compare entries 1 with 9 and 15; entries 4 with 12 and 16). And acetophenones bearing either electron-withdrawing or electron-donating groups at 2-, 3- or 4-position of the benzene ring, in all cases, provided smoothly the desired target molecules 3 and 4 in good yields. When 1-(4-nitrophenyl)ethanone was employed to react with 2a under the same conditions, however, the reaction did not occur, and 1-(4-nitrophenyl)ethanone could be quantitatively recovered.
| Entry | 1/R1 | 2 | Time (h) | 3 (Yieldb %) | 4 (Yieldb %) |
|---|---|---|---|---|---|
| a Reaction conditions: methyl ketones 1 (1.0 mmol), 5-aryl-1,2,4-triazines 2 (1.0 mmol), NaOH (1.5 mmol), in 5 mL of EtOH at refluxing temperature.b Isolated yields by column chromatography. | |||||
| 1 | 1a C6H5 | 2a | 1.5 | 3a (25) | 4a (60) |
| 2 | 1b 4-FC6H4 | 2a | 2 | 3b (20) | 4b (59) |
| 3 | 1c 4-ClC6H4 | 2a | 2 | 3c (20) | 4c (61) |
| 4 | 1d 4-BrC6H4 | 2a | 2.5 | 3d (21) | 4d (62) |
| 5 | 1e 4-CH3C6H4 | 2a | 2.5 | 3e (35) | 4e (53) |
| 6 | 1f 4-CH3OC6H4 | 2a | 3 | 3f (35) | 4f (52) |
| 7 | 1g 3-ClC6H4 | 2a | 2 | 3g (19) | 4g (56) |
| 8 | 1h 2,4-Cl2C6H3 | 2a | 2.5 | 3h (18) | 4h (57) |
| 9 | 1a C6H5 | 2b | 2 | 3i (25) | 4i (58) |
| 10 | 1b 4-FC6H4 | 2b | 2.5 | 3j (18) | 4j (57) |
| 11 | 1c 4-ClC6H4 | 2b | 2.5 | 3k (19) | 4k (57) |
| 12 | 1d 4-BrC6H4 | 2b | 3 | 3l (20) | 4l (58) |
| 13 | 1e 4-CH3C6H4 | 2b | 3.5 | 3m (34) | 4m (52) |
| 14 | 1f 4-CH3OC6H4 | 2b | 4 | 3n (35) | 4n (52) |
| 15 | 1a C6H5 | 2c | 2 | 3o (25) | 4o (59) |
| 16 | 1d 4-BrC6H4 | 2c | 2.5 | 3p (20) | 4p (61) |
| 17 | 1f 4-CH3OC6H4 | 2c | 3 | 3q (34) | 4q (52) |
| 18 | 1i CH3 | 2a | 4.5 | 3r (42) | 4r (43) |
Additionally, all of experimental results showed the major products were the 2,4-disubstituted-pyridines 4 rather than the 2,5-disubstituted-pyridines 3, showing the same regioselectivity with previous reports.7
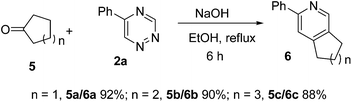
Encouraged by the above results, the substrate scope was extended to cyclic ketones (Scheme 2). To our pleasure, cyclopentanone 5a, cyclohexanone 5b, and cycloheptanone 5c were successfully employed to produce single pyridine isomers in excellent yields.
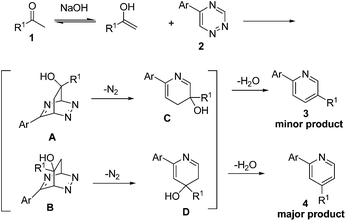
Based on the above experimental results, a plausible reaction mechanism is presented in Scheme 3. Initially, the methyl ketones tautomerized to the enols under the basic conditions. Then, as dienophiles, the enols reacted directly with the 1,2,4-triazines 2 through the inverse electron demand Diels–Alder reaction to give intermediates A and B. Subsequently, intermediates C and D were formed by retro-Diels–Alder process with a loss of nitrogen molecule. Finally, the target products 3 and 4 were obtained by elimination–aromatization of the intermediate C and D.
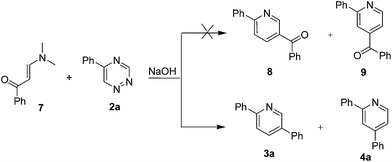
Considering that enaminones contain the enamine motif, we attempted to use 3-(dimethylamino)-1-phenylprop-2-en-1-one 7 as a dienophile to react with 1,2,4-triazine 2a directly (CHCl3, 45 °C), expecting to obtain products 8/9 (Scheme 4). Unfortunately, none of the desired products 8/9 were observed. However, when this reaction was conducted under above optimum conditions, the decarbonylated products 3a/4a were obtained instead of the desired products 8/9. The control experiments revealed an unprecedented phenomenon that enaminone 7 could easily converted to the corresponding acetophenone 1a under the reaction conditions. To our knowledge, this retro-aldol type conversion under basic conditions has never been reported.
Conclusions
In summary, we have successfully developed a simple and efficient synthetic protocol for pyridine derivatives by inverse electron demand Diels–Alder/retro-Diels–Alder reactions using methyl ketones and 1,2,4-triazines. The striking feature of the reaction is using methyl ketones as a potential dienophile without formation of enamine intermediate. Undoubtedly, the novel method is complementary to Boger reaction and revealed a convenient and green way to access pyridine derivatives in an atom-economic manner. We hope this approach may be of value to others seeking novel synthetic fragments with unique properties for medicinal and materials chemistry.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21372137 and 21072110) and the Natural Science Foundation of Shandong Province (ZR2012BM003).Notes and references
- For reviews, see: (a) G. D. Henry, Tetrahedron, 2004, 60, 6043 CrossRef CAS PubMed; (b) G. Jones, in Comprehensive Heterocyclic Chemistry II, ed. A. R.Katritzky, C. W.Rees, and E. F. V.Scriven, Pergamon Press, Oxford, U.K., 1996, vol. 5, pp. 167–243 Search PubMed; (c) P. A. Keller, in Comprehensive Heterocyclic Chemistry III, ed. A. R.Katritzky, C. A.Ramsden, E. F. V.Scriven and R. J. K.Taylor, Elsevier, Oxford, U.K., 2008, vol. 7, pp. 217–308 Search PubMed.
- (a) M. Movassaghi and M. D. Hill, J. Am. Chem. Soc., 2006, 128, 4592 CrossRef CAS PubMed; (b) V. S. Thoi and C. J. Chang, Chem. Commun., 2011, 47, 6578 RSC; (c) M. Ohashi, I. Takeda, M. Ikawa and S. Ogoshi, J. Am. Chem. Soc., 2011, 133, 18018 CrossRef CAS PubMed; (d) M. Z. Chen and G. C. Micalizio, J. Am. Chem. Soc., 2012, 134, 1352 CrossRef CAS PubMed; (e) N. S. Y. Loy, A. Singh, X. Xu and C. M. Park, Angew. Chem., Int. Ed., 2013, 52, 2212 CrossRef CAS PubMed; (f) Y. Satoh and Y. Obora, J. Org. Chem., 2013, 78, 7771 CrossRef CAS PubMed; (g) J. M. Neely and T. Rovis, J. Am. Chem. Soc., 2013, 135, 66 CrossRef CAS PubMed; (h) Y. Wei and N. Yoshikai, J. Am. Chem. Soc., 2013, 135, 3756 CrossRef CAS PubMed; (i) C.-H. Lei, D.-X. Wang, L. Zhao, J. Zhu and M.-X. Wang, J. Am. Chem. Soc., 2013, 135, 4708 CrossRef CAS PubMed; (j) V. Richard, M. Ipouck, D. S. Mérel, S. Gaillard, R. J. Whitby, B. Witulski and J. Renaud, Chem. Commun., 2014, 50, 593 RSC.
- (a) M. J. Schnermann and D. L. Boger, J. Am. Chem. Soc., 2005, 127, 15704 CrossRef CAS PubMed; (b) M. Ermd, M. heuschmann and H. Zipse, Helv. Chim. Acta, 2005, 88, 1491 CrossRef; (c) M. Movassaghi, M. D. Hill and O. K. Ahmad, J. Am. Chem. Soc., 2007, 129, 10096 CrossRef CAS PubMed; (d) J. Lu, J. A. Keith, W. Shen, M. Schurmann, H. Preut, T. Jacob and H. Arndt, J. Am. Chem. Soc., 2008, 130, 13219 CrossRef CAS PubMed; (e) J. Hu, Q. Zhang, H. Yuan and Q. Liu, J. Org. Chem., 2008, 73, 2442 CrossRef CAS PubMed; (f) Z. C. Chen, J. Zhu, H. Xie, Y. Wu and Y. Gong, Org. Lett., 2010, 12, 4376 CrossRef CAS PubMed; (g) Z. Chen, D. Hong and Y. Wang, J. Org. Chem., 2009, 74, 903 CrossRef CAS PubMed; (h) J. Li, P. He and C. Yu, Tetrahedron, 2012, 68, 4138 CrossRef CAS PubMed.
- (a) D. L. Boger and J. S. Panek, J. Org. Chem., 1981, 46, 2179 CrossRef CAS; (b) D. L. Boger, J. S. Panek and M. M. Meier, J. Org. Chem., 1982, 47, 897 CrossRef; (c) D. L. Boger, Tetrahedron, 1983, 39, 2869 CrossRef CAS; (d) D. L. Boger and J. S. Panek, J. Am. Chem. Soc., 1985, 107, 5745 CrossRef CAS.
- (a) E. C. Taylor and J. E. Macor, J. Org. Chem., 1989, 54, 1249 CrossRef CAS; (b) E. C. Taylor and J. E. Macor, J. Org. Chem., 1989, 54, 4984 CrossRef CAS; (c) S. A. Raw and J. K. Taylor, J. Am. Chem. Soc., 2004, 126, 12260 CrossRef CAS PubMed; (d) Y. F. Sainz, S. A. Raw and R. J. K. Taylor, J. Org. Chem., 2005, 70, 10086 CrossRef CAS PubMed; (e) N. Catozzi, M. G. Edwards, S. A. Raw, P. Wasnaire and R. J. K. Taylor, J. Org. Chem., 2009, 74, 8343 CrossRef CAS PubMed.
- (a) M. Li, H. Cao, Y. Wang, X.-L. Lv and L.-R. Wen, Org. Lett., 2012, 14, 3470 CrossRef CAS PubMed; (b) L.-R. Wen, Z.-R. Li, M. Li and H. Cao, Green Chem., 2012, 14, 707 RSC; (c) L.-R. Wen, T. He, M.-C. Lan and M. Li, J. Org. Chem., 2013, 78, 10617 CrossRef CAS PubMed; (d) L.-R. Wen, Q.-C. Sun, H.-L. Zhang and M. Li, Org. Biomol. Chem., 2013, 11, 781 RSC; (e) M. Li, X.-L. Lv, L.-R. Wen and Z.-Q. Hu, Org. Lett., 2013, 15, 1262 CrossRef CAS PubMed; (f) L.-R. Wen, L.-B. Men, T. He, G.-J. Ji and M. Li, Chem.–Eur. J., 2014, 20, 5028 CrossRef CAS PubMed.
- L. Boger, Chem. Rev., 1986, 86, 781 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedures, full spectroscopic data for all new compounds, and crystal data for 4d (CIF). CCDC 955651. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra11294f |
| This journal is © The Royal Society of Chemistry 2014 |