A relay ring-opening/double ring-closing metathesis strategy for the bicyclic macrolide-butenolide core structures†
Mahesh B. Halle and
Rodney A. Fernandes*
Department of Chemistry, Indian Institute of Technology Bombay, Powai, Mumbai 400 076, Maharashtra, India. E-mail: rfernand@chem.iitb.ac.in; Fax: +91-22-25767152; Tel: +91-22-25767174
First published on 12th November 2014
Abstract
A concise strategy has been developed for the synthesis of the bicyclic macrolide-butenolide core structures of various natural products with the macrolide ring size ranging from 12- to 16-membered. The bicyclic structure was easily assembled using the relay ring-opening/double ring-closing metathesis strategy. An efficient synthesis of (±)-desmethyl manshurolide has been achieved as an application of this strategy.
Introduction
The bicyclic macrolide-butenolide core structure 1 is present in many natural products (Fig. 1). The macrolide size may vary and there could be the presence of one or more double bonds. Selected examples are shown in Fig. 1 with the macrolide size varying from 12- to 14-membered (2a–e) being the most common. Gersemolide 2a (12-membered macrolide) is a pseudopterane, isolated from the soft coral Gersemia rubiformis.1 Manshurolide 2b, also a 12-membered macrolide is a sesquiterpene lactone isolated from the stems of Aristolochia manshuriensis.2 It has three trans-double bonds in the macrolide ring (the butenolide double bond is apparently trans- in the macrolide ring but geometrically cis- in the butenonide).Okilactomycins C 2c and D 2d have a 13-membered bicyclic macrolide-butenolide structure with an annulated cyclohexene and additional spiro functionality. These were isolated from Streptomyces scabrisporus.3 The cembranoid diterpenes, sarcophytonolides A-L were isolated from the soft coral of the genus Sarcophyton.4 These possess the 14-membered bicyclic macrolide-butenolide structure. The simpler member is sarcophytonolide L 2e. The bicyclic macrolide-butenolide structure is also present in many other natural products with various substituents and functionalities. Often there is the furan ring5 (or the oxidized diketo form) as another common feature in most of them (Fig. 1). These could be the furano-pseudopterane type 3a, furano-gersolene type 3b or the furano-cembrane type 3c (Fig. 1).5 The butenolide moiety gains it significance in medicinal chemistry as Michael acceptor to various biological nucleophiles.
With our continued interest in the synthesis of γ-lactones, butenolides and strained macrolides6 and considering the importance of these bicyclic structures we embarked on developing a strategy to rapidly construct both the rings (butenolide and macrolide) in an efficient manner based on a relay ring-opening/multiple ring-closing metathesis.7
Results and discussion
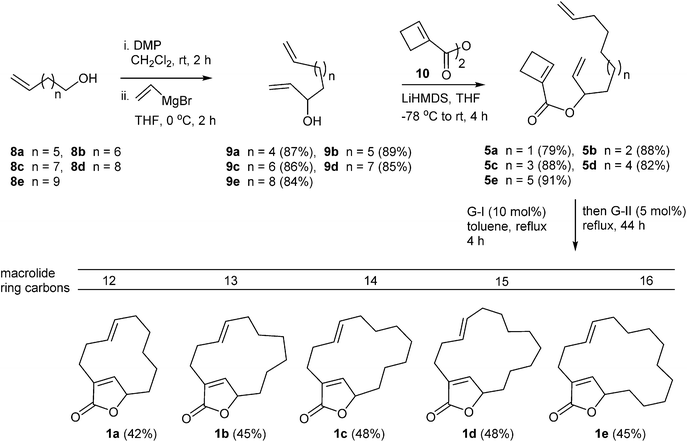
As shown in the retrosynthetic analysis (Scheme 1) the core structure 1 could be assembled through a double ring-closing metathesis of the tetraene 4. However this design would impose issues of regioselectivity in the ring-closure considering the competition of three available terminal mono-substituted double bonds. While the butenolide ring closure could occur with ease, the macrolide ring closure would be challenging. The latter would predominantly depend on ring size and would occur separately of the first ring-closure. We sought an alternative path to prevent competition by reducing the number of double bonds and hence the triene 5 seemed obvious choice. In this case during metathesis there would be carbene cascade which appeared promising for the challenging macrolide ring closure unlike the case of compound 4 where the two ring closures would occur separately. The substrate 5 can be considered as synthetic equivalent to compound 4. Compound 5 can easily be assembled through vinyl Grignard reaction on aldehyde 6 (of varying chain length) followed by esterification with cyclobutene carboxylic acid 7. An advantage of this strategy is the ready adaptability for chiral version where in the intermediate allyl alcohol 9 can be resolved through Sharpless kinetic resolution.8The forward synthesis towards the bicyclic core structure 1 with macrolide ring size varying from 12- to 16-membered is shown in Scheme 2. The vinyl Grignard addition to various aldehydes from the alcohols 8a–e gave the corresponding allyl alcohols 9a–e in 84–89% yields. The esterification of 9a with the cyclobutene carboxylic acid 7 using DCC gave the corresponding ester 5a in 38% yield. The reaction using Yamaguchi conditions9 delivered 5a in 51% yield. This was further improved with the use of the anhydride of 7, i.e. 10, and LiHMDS that delivered the ester 5a in good 79% yield. Similarly, other esters 5b–e were prepared in 82–91% yields. The relay ring-opening/double ring-closing metathesis (RO/DRCM) reaction on 5a–e was carried out using Grubbs catalysts (G-I and G-II) and Grubbs-Hoveyda catalysts (GH-I and GH-II) in toluene, benzene and hexane solvent. The best results were obtained through a sequential addition of first G-I catalyst (10 mol%) followed by addition of G-II catalyst (5 mol%) in toluene solvent providing the macrolide-butenolide bicyclic compounds 1a–e in 42–48% yields with E-selectivity for the macrolide double bond.10 We believe, G-I catalyst is known for ring-opening metathesis of strained small rings (in this case the cyclobutene ring). Thus the above catalysts combination worked well in this case. However, we did not isolate any of the intermediate compounds in this relay metathesis reaction which may require quenching with ethylene.
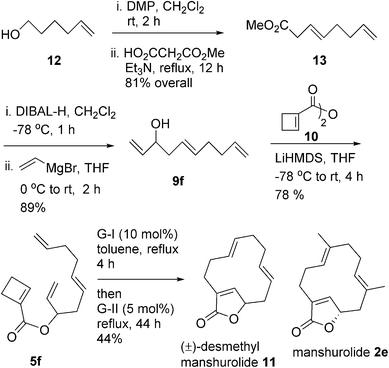
The strategy was extended towards the protecting group free synthesis of (±)-desmethyl manshurolide 11 as shown in Scheme 3. Commercially available 5-hexene-1-ol 12 was oxidized to aldehyde and the subsequent modified de-carboxylative deconjugative Knoevenagel condensation11 with half ester of malonic acid gave the ester 13 in 81% overall yield. DiBAL-H reduction of the ester group to aldehyde and subsequent vinyl Grignard reaction furnished the triene 9f (89% from 13). The esterification of 9f with the anhydride 10 using LiHMDS gave 5f in 78% yield. The subsequent relay RO/DRCM using G-I (10 mol%) and G-II (5 mol%) catalysts in toluene solvent delivered (±)-desmethyl manshurolide 11 in 44% yield.
Conclusions
In conclusion an efficient strategy for bicyclic macrolide-butenolide core structures of various natural products has been developed. The macrolide rings ranging from 12- to 16-membered have been synthesized. The efficiency of the relay ring-opening/double ring-closing metathesis strategy was further demonstrated by the protecting group free synthesis of (±)-desmethyl manshurolide. The strategy has potential towards the synthesis of bicyclic macrolide-butenolide natural products.Experimental section
General information
Flasks were oven- or flame-dried and cooled in a desiccator. Dry reactions were carried out under an atmosphere of Ar or N2. Solvents and reagents were purified by standard methods. Thin-layer chromatography was performed on EM250 Kieselgel 60 F254 silica gel plates. The spots were visualized by staining with KMnO4 or by UV lamp. 1H NMR and 13C NMR were recorded with Bruker AVANCE III 400 or 500 and 100 or 125 MHz, respectively, and chemical shifts are based on TMS peak at δ = 0.00 pm for proton NMR and CDCl3 peak at δ = 77.00 ppm (t) in carbon NMR. IR samples were prepared by evaporation from CHCl3 on CsBr plates. High-resolution mass spectra were obtained using positive electrospray ionization by TOF method.General procedure for synthesis of allyl alcohols 9a–e
To a solution of alcohol 8 (2.0 mmol) in CH2Cl2 (20 mL) was added Dess–Martin periodinate (DMP, 1.87 g, 4.40 mmol, 2.2 equiv.). The resulting mixture was stirred at room temperature for 2 h. It was then quenched with a solution of 10% aq. Na2S2O3 and sat. aq. NaHCO3 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 15 mL) and the solution extracted with CH2Cl2 (3 × 50 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was loaded on a short pad of silica gel and washed with petroleum ether/EtOAc (2
1, 15 mL) and the solution extracted with CH2Cl2 (3 × 50 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was loaded on a short pad of silica gel and washed with petroleum ether/EtOAc (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to give the aldehyde which was used directly for next step. Vinyl magnesium bromide (3.0 mL, 3.0 mmol, 1.5 equiv., 1 M solution in THF) was added to the aldehyde dissolved in THF (30 mL) at 0 °C. The reaction mixture was stirred for 2 h at 0 °C and warmed to room temperature. Saturated aq. NH4Cl (15 mL) was added and the mixture extracted with EtOAc (3 × 50 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (4
1) to give the aldehyde which was used directly for next step. Vinyl magnesium bromide (3.0 mL, 3.0 mmol, 1.5 equiv., 1 M solution in THF) was added to the aldehyde dissolved in THF (30 mL) at 0 °C. The reaction mixture was stirred for 2 h at 0 °C and warmed to room temperature. Saturated aq. NH4Cl (15 mL) was added and the mixture extracted with EtOAc (3 × 50 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give the alcohols 9a–e in 84–89% yield.
1) as eluent to give the alcohols 9a–e in 84–89% yield.
General procedure for synthesis of esters 5a–e
To a solution of allyl alcohol 9a–e (1.0 mmol) in dry THF (15 mL) at −78 °C was gradually added LiHMDS (0.75 mL, 1.5 mmol, 1.5 equiv., 2 M solution in THF) over a period of 5 min. After stirring for 15 min, a solution of anhydride 10 (0.267 g, 1.50 mmol, 1.5 equiv.) in dry THF (5 mL) was added and the reaction mixture stirred for 4 h at −78 °C. It was slowly warmed to room temperature and a solution of sat. aq. NH4Cl (5 mL) was added. The mixture was extracted with EtOAc (3 × 50 mL) and the combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give the esters 5a–e in 79–91% yield.
1) as eluent to give the esters 5a–e in 79–91% yield.
General procedure for synthesis of macrolide-butenolides 1a–e
To a solution of 5a–e (0.20 mmol) in dry and degassed toluene (150 mL) was added Grubbs-I catalyst (16.5 mg, 0.02 mmol, 10 mol%). The mixture was refluxed for 4 h under nitrogen atmosphere and then cooled and Grubbs-II catalyst (8.5 mg, 0.01 mmol, 5 mol%) was added. The mixture was further refluxed for 44 h under nitrogen atmosphere and then cooled and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give the macrolide-butenolides1a–e.
1) as eluent to give the macrolide-butenolides1a–e.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 15 mL) and the solution extracted with CH2Cl2 (3 × 50 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was loaded on a short pad of silica gel and washed with petroleum ether/EtOAc (2
1, 15 mL) and the solution extracted with CH2Cl2 (3 × 50 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was loaded on a short pad of silica gel and washed with petroleum ether/EtOAc (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to give the aldehyde which was used for the next step. To the aldehyde (0.582 g) was added mono methylmalonate (1.06 g, 9.0 mmol, 1.5 equiv.) followed by Et3N (1.2 mL, 12.0 mmol, 2.0 equiv.). The reaction mixture was refluxed for 12 h. It was then cooled and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (9
1) to give the aldehyde which was used for the next step. To the aldehyde (0.582 g) was added mono methylmalonate (1.06 g, 9.0 mmol, 1.5 equiv.) followed by Et3N (1.2 mL, 12.0 mmol, 2.0 equiv.). The reaction mixture was refluxed for 12 h. It was then cooled and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give ester 13 (0.748 g, 81%) as colorless oil (analysis showed it to contain <9% α,β-unsaturated isomer. This was accounted for yield calculation). IR (CHCl3): νmax 3070, 2926, 2849, 1741, 1641, 1437, 1256, 1199, 1164, 996, 971, 914, 667 cm−1; 1H NMR (400 MHz, CDCl3, ppm) δ 5.81–5.71 (m, 1H), 5.52–5.50 (m, 2H), 4.99–4.90 (m, 2H), 3.63 (s, 3H), 2.99 (d, J = 5.0 Hz, 2H), 2.10–2.02 (m, 4H); 13C NMR (100 MHz, CDCl3, ppm) δ 172.4, 137.9, 133.8, 121.9, 114.6, 51.6, 37.7, 33.2, 31.7; HRMS m/z calcd for [C9H14O2Na]+ 177.0863, found 177.0859.
1) as eluent to give ester 13 (0.748 g, 81%) as colorless oil (analysis showed it to contain <9% α,β-unsaturated isomer. This was accounted for yield calculation). IR (CHCl3): νmax 3070, 2926, 2849, 1741, 1641, 1437, 1256, 1199, 1164, 996, 971, 914, 667 cm−1; 1H NMR (400 MHz, CDCl3, ppm) δ 5.81–5.71 (m, 1H), 5.52–5.50 (m, 2H), 4.99–4.90 (m, 2H), 3.63 (s, 3H), 2.99 (d, J = 5.0 Hz, 2H), 2.10–2.02 (m, 4H); 13C NMR (100 MHz, CDCl3, ppm) δ 172.4, 137.9, 133.8, 121.9, 114.6, 51.6, 37.7, 33.2, 31.7; HRMS m/z calcd for [C9H14O2Na]+ 177.0863, found 177.0859.To a solution of above aldehyde in THF (15 mL) was added vinyl magnesium bromide (5.0 mL, 5.0 mmol, 1.51 equiv., 1 M solution in THF) at 0 °C. The reaction mixture was stirred for 2 h at 0 °C and then warmed to room temperature. Saturated aq. NH4Cl (10 mL) was added and the mixture extracted with EtOAc (3 × 20 mL). The combined organic layers were washed with brine, dried (Na2SO4) and concentrated. The residue was purified by silica gel flash column chromatography using petroleum ether/EtOAc (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give the allyl alcohol 9f (0.448 g, 89%) as colorless oil. IR (CHCl3): νmax 3444, 3077, 2926, 2855, 1641, 1618, 1577, 1481, 1467, 1378, 1252, 1176, 1127, 1070, 971, 914, 810, 694 cm−1; 1H NMR (400 MHz, CDCl3, ppm) δ 5.90–5.83 (m, 1H), 5.82–5.75 (m, 1H), 5.57–5.53 (m, 1H), 5.44–5.37 (m, 1H), 5.24 (dt, J = 17.2, 1.5 Hz, 1H), 5.13 (dt, J = 10.5, 1.4 Hz, 1H), 5.03–4.94 (m, 2H), 4.1 (brd, J = 1.6 Hz, 1H), 2.31–2.19 (m, 1H), 2.18–2.16 (m, 1H), 2.14–2.12 (m, 4H), 1.7 (brs, 1H); 13C NMR (125 MHz, CDCl3, ppm) δ 140.4, 138.3, 134.1, 125.6, 114.8, 114.6, 71.8, 40.5, 33.6, 32.0; HRMS m/z calcd for [C10H16ONa]+ 175.1093, found 175.1085.
1) as eluent to give the allyl alcohol 9f (0.448 g, 89%) as colorless oil. IR (CHCl3): νmax 3444, 3077, 2926, 2855, 1641, 1618, 1577, 1481, 1467, 1378, 1252, 1176, 1127, 1070, 971, 914, 810, 694 cm−1; 1H NMR (400 MHz, CDCl3, ppm) δ 5.90–5.83 (m, 1H), 5.82–5.75 (m, 1H), 5.57–5.53 (m, 1H), 5.44–5.37 (m, 1H), 5.24 (dt, J = 17.2, 1.5 Hz, 1H), 5.13 (dt, J = 10.5, 1.4 Hz, 1H), 5.03–4.94 (m, 2H), 4.1 (brd, J = 1.6 Hz, 1H), 2.31–2.19 (m, 1H), 2.18–2.16 (m, 1H), 2.14–2.12 (m, 4H), 1.7 (brs, 1H); 13C NMR (125 MHz, CDCl3, ppm) δ 140.4, 138.3, 134.1, 125.6, 114.8, 114.6, 71.8, 40.5, 33.6, 32.0; HRMS m/z calcd for [C10H16ONa]+ 175.1093, found 175.1085.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give the ester 5f (0.12 g, 78%) as colorless oil. IR (CHCl3): νmax 3075, 2929, 2857, 1731, 1641, 1464, 1252, 1180, 1046, 991, 917, 668 cm−1;1H NMR (500 MHz, CDCl3, ppm) δ 6.8 (s, 1H), 5.86–5.76 (m, 2H), 5.55–5.45 (m, 1H), 5.41–5.33 (m, 1H), 5.32–5.22 (m, 2H), 5.21–5.16 (m, 1H), 5.08–4.82 (m, 2H), 2.78–2.70 (m, 2H), 2.49–2.44 (m, 2H), 2.40–2.30 (m, 2H), 2.12–2.00 (m, 4H); 13C NMR (125 MHz, CDCl3, ppm) δ 161.5, 146.4, 138.9, 138.2, 136.1, 133.3, 124.8, 116.6, 114.6, 73.8, 37.6, 33.6, 32.0, 29.7, 27.0; HRMS m/z calcd for [C15H20O2Na]+ 255.1356, found 255.1361.
1) as eluent to give the ester 5f (0.12 g, 78%) as colorless oil. IR (CHCl3): νmax 3075, 2929, 2857, 1731, 1641, 1464, 1252, 1180, 1046, 991, 917, 668 cm−1;1H NMR (500 MHz, CDCl3, ppm) δ 6.8 (s, 1H), 5.86–5.76 (m, 2H), 5.55–5.45 (m, 1H), 5.41–5.33 (m, 1H), 5.32–5.22 (m, 2H), 5.21–5.16 (m, 1H), 5.08–4.82 (m, 2H), 2.78–2.70 (m, 2H), 2.49–2.44 (m, 2H), 2.40–2.30 (m, 2H), 2.12–2.00 (m, 4H); 13C NMR (125 MHz, CDCl3, ppm) δ 161.5, 146.4, 138.9, 138.2, 136.1, 133.3, 124.8, 116.6, 114.6, 73.8, 37.6, 33.6, 32.0, 29.7, 27.0; HRMS m/z calcd for [C15H20O2Na]+ 255.1356, found 255.1361.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to give the desmethyl manshurolide 11 (19.1 mg, 44%) as colorless oil. IR (CHCl3): νmax 2923, 2851, 1753, 1651, 1593, 1434, 1360, 1259, 1122, 1091, 1039, 1017, 957, 932, 869, 666 cm−1; 1H NMR (400 MHz, CDCl3, ppm) δ 6.75 (s, 1H), 5.18–5.11 (m, 1H), 5.06–5.02 (m, 2H), 5.00–4.98 (m, 2H), 2.80–2.75 (m, 1H), 2.56–2.52 (m, 1H), 2.36–2.18 (m, 6H), 1.88–1.79 (m, 2H); 13C NMR (100 MHz, CDCl3, ppm) δ 173.8, 150.5, 135.3, 133.7, 132.9, 129.8, 123.3, 80.1, 34.1, 31.8, 31.5, 29.7, 25.5; HRMS m/z calcd for [C13H16O2Na]+ 227.1043, found 227.1044.
1) as eluent to give the desmethyl manshurolide 11 (19.1 mg, 44%) as colorless oil. IR (CHCl3): νmax 2923, 2851, 1753, 1651, 1593, 1434, 1360, 1259, 1122, 1091, 1039, 1017, 957, 932, 869, 666 cm−1; 1H NMR (400 MHz, CDCl3, ppm) δ 6.75 (s, 1H), 5.18–5.11 (m, 1H), 5.06–5.02 (m, 2H), 5.00–4.98 (m, 2H), 2.80–2.75 (m, 1H), 2.56–2.52 (m, 1H), 2.36–2.18 (m, 6H), 1.88–1.79 (m, 2H); 13C NMR (100 MHz, CDCl3, ppm) δ 173.8, 150.5, 135.3, 133.7, 132.9, 129.8, 123.3, 80.1, 34.1, 31.8, 31.5, 29.7, 25.5; HRMS m/z calcd for [C13H16O2Na]+ 227.1043, found 227.1044.Acknowledgements
We thank the Board of Research in Nuclear Sciences (BRNS), Government of India (Basic Sciences, Grant no. 2013/37C/59/BRNS/2443) for financial support. M.B.H. thanks the Council of Scientific and Industrial Research (CSIR), New Delhi for senior research fellowship.Notes and references
- D. Williams and R. J. Andersen, J. Org. Chem., 1987, 52, 332–335 CrossRef CAS.
- G. Rücker, C. W. Ming, R. Mayer, G. Will and A. Güllmann, Phytochemistry, 1990, 29, 983–985 CrossRef.
- C. Zhang, J. G. Ondeyka, D. L. Zink, A. Basilio, F. Vicente, O. Salazar, O. Genilloud, K. Dorso, M. Motyl, K. Byrne and S. B. Singh, J. Antibiot., 2009, 62, 55–61 CrossRef CAS PubMed.
- (a) R. Jia, Y.-W. Guo, E. Mollo and G. Cimino, Helv. Chim. Acta, 2005, 88, 1028–1033 CrossRef CAS; (b) R. Jia, Y.-W. Guo, E. Mollo, M. Gavagnin and G. Cimino, J. Nat. Prod., 2006, 69, 819–822 CrossRef CAS PubMed; (c) X.-H. Yan, Z.-Y. Li and Y.-W. Guo, Helv. Chim. Acta, 2007, 90, 1574–1580 CrossRef CAS.
- P. A. Roethle and D. Trauner, Nat. Prod. Rep., 2008, 25, 298–317 RSC.
- (a) R. A. Fernandes and A. K. Chowdhury, J. Org. Chem., 2009, 74, 8826–8829 CrossRef CAS PubMed; (b) R. A. Fernandes, A. B. Ingle and V. P. Chavan, Tetrahedron: Asymmetry, 2009, 20, 2835–2844 CrossRef CAS PubMed; (c) R. A. Fernandes and A. B. Ingle, Synlett, 2010, 158–160 CrossRef CAS PubMed; (d) R. A. Fernandes and A. B. Ingle, Tetrahedron Lett., 2011, 52, 458–460 CrossRef CAS PubMed; (e) R. A. Fernandes and A. K. Chowdhury, Eur. J. Org. Chem., 2011, 1106–1112 CrossRef CAS; (f) R. A. Fernandes and P. Kattanguru, Tetrahedron: Asymmetry, 2011, 22, 1930–1935 CrossRef CAS PubMed; (g) R. A. Fernandes and P. Kattanguru, J. Org. Chem., 2012, 77, 9357–9360 CrossRef CAS PubMed; (h) R. A. Fernandes, V. P. Chavan, S. V. Mulay and A. Manchoju, J. Org. Chem., 2012, 77, 10455–10460 CrossRef CAS PubMed; (i) R. A. Fernandes and P. Kattanguru, Asian J. Org. Chem., 2013, 2, 74–84 CrossRef CAS; (j) R. A. Fernandes and V. P. Chavan, Chem. Commun., 2013, 49, 3354–3356 RSC; (k) R. A. Fernandes and M. B. Halle, Asian J. Org. Chem., 2013, 2, 593–599 CrossRef CAS; (l) R. A. Fernandes, P. H. Patil and A. K. Chowdhury, Eur. J. Org. Chem., 2013, 237–243 Search PubMed; (m) R. A. Fernandes, P. H. Patil and A. K. Chowdhury, Asian J. Org. Chem., 2014, 3, 58–62 CrossRef CAS; (n) R. A. Fernandes, P. Kattanguru and V. Bethi, RSC Adv., 2014, 4, 14507–14512 RSC.
- For selected examples of ring-opening/ring-closing metathesis see: (a) L. Yet, Chem. Rev., 2000, 100, 2963–3007 CrossRef CAS PubMed; (b) C. L. Chandler and A. J. Phillips, Org. Lett., 2005, 7, 3493–3495 CrossRef CAS PubMed; (c) M. W. B. Pfeiffer and A. J. Phillips, J. Am. Chem. Soc., 2005, 127, 5334–5335 CrossRef CAS PubMed; (d) A. C. Hart and A. J. Phillips, J. Am. Chem. Soc., 2006, 128, 1094–1095 CrossRef CAS PubMed; (e) N. Holub and S. Blechert, Chem.–Asian J., 2007, 2, 1064–1082 CrossRef CAS PubMed; (f) A. Song, K. A. Parker and N. S. Sampson, J. Am. Chem. Soc., 2009, 131, 3444–3445 CrossRef CAS PubMed; (g) C. K. Malik, R. N. Yadav, M. G. B. Drew and S. Ghosh, J. Org. Chem., 2009, 74, 1957–1963 CrossRef CAS PubMed; (h) H. D. Cooper and D. L. Wright, Molecules, 2013, 18, 2438–2448 CrossRef CAS PubMed; (i) K. Takao, R. Nanamiya, Y. Fukushima, A. Namba, K. Yoshida and K. Tadano, Org. Lett., 2013, 15, 5582–5585 CrossRef CAS PubMed.
- For Sharpless kinetic resolution see: (a) T. Katsuki and K. B. Sharpless, J. Am. Chem. Soc., 1980, 102, 5974–5976 CrossRef CAS; (b) V. S. Martin, S. S. Woodward, T. Katsuki, Y. Yamada, M. Ikeda and K. B. Sharpless, J. Am. Chem. Soc., 1981, 103, 6237–6247 CrossRef CAS . For few examples of kinetic resolution of allyl alcohols similar to 9 with additional isolated double bond see:; (c) J. M. Percy, R. Roig and K. Singh, Eur. J. Org. Chem., 2009, 1058–1071 CrossRef CAS; (d) X. Liao and X. Xu, Tetrahedron Lett., 2000, 41, 4641–4644 CrossRef CAS; (e) P. C. Bulman Page, C. M. Rayner and I. O. Sutherland, Tetrahedron Lett., 1986, 27, 3535–3538 CrossRef.
- For Yamaguchi esterification see: (a) J. Inanaga, K. Hirata, H. Saeki, T. Katsuki and M. Yamaguchi, Bull. Chem. Soc. Jpn., 1979, 52, 1989–1993 CrossRef CAS; (b) T. Nagamitsu, D. Takano, T. Fukuda, K. Otoguro, I. Kuwajima, Y. Harigaya and S. Omura, Org. Lett., 2004, 6, 1865–1867 CrossRef CAS PubMed; (c) I. Dhimitruka and J. StantaLucia Jr, Org. Lett., 2006, 8, 47–50 CrossRef CAS PubMed; (d) T. Nagamitsu, D. Takano, K. Marumoto, T. Fukuda, K. Furuya, K. Otoguro, K. Takeda, I. Kuwajima, Y. Harigaya and S. Omura, J. Org. Chem., 2007, 72, 2744–2756 CrossRef CAS PubMed.
- The (E)-selectivity was arrived at by comparison of similar ring closure and appearance of peaks (δ values) in the 1H NMR spectra which did not show any diastereomeric peaks. See: (a) A. Fürstner and C. Müller, Chem. Commun., 2005, 5583–5585 RSC; (b) M. K. Brown and A. H. Hoveyda, J. Am. Chem. Soc., 2008, 130, 12904–12906 CrossRef CAS PubMed; (c) Z. Cai, N. Yongpruksa and M. Harmata, Org. Lett., 2012, 14, 1661–1663 CrossRef CAS PubMed.
- For Knoevenagel condensation see: (a) H. Yamanaka, M. Yokoyama, T. Sakamoto, T. Shiraishi, M. Sagi and M. Mizugaki, Heterocycles, 1983, 20, 1541–1544 CrossRef CAS; (b) N. Ragoussis, Tetrahedron Lett., 1987, 28, 93–96 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Copies of 1H and 13C NMR spectra for all the compounds. See DOI: 10.1039/c4ra10937f |
| This journal is © The Royal Society of Chemistry 2014 |