Synthesis and blue phosphorescent device performances of a new bipolar host material containing pyrazino[2,3-b]indole moiety†
Hong-Gu Jang,
Sang Kyu Jeon,
Jun Yeob Lee and
Seok-Ho Hwang*
Department of Polymer Science and Engineering, Dankook University, Yongin, Gyeonggi 448-701, Republic of Korea. E-mail: bach@dankook.ac.kr; Fax: +82-31-8005-3585; Tel: +82-31-8005-3588
First published on 16th October 2014
Abstract
A new bipolar host material (CzPhPz) consisting of 5H-pyrazino[2,3-b]indole unit was synthesized and its photophysical properties and device performances were investigated. It showed good electron transport properties with a high triplet energy of 2.83 eV. CzPhPz was effective as the host material for FIrpic-based blue PHOLED with a high quantum efficiency of 15.0%.
For more than ten years, the progress of device performances of phosphorescent organic light emitting devices (PHOLEDs) has attracted tremendous attention because of their high quantum efficiency as well as their potential applications in full-color flat-panel displays and solid-state lighting applications.1
In the research field for PHOLEDs, a host material plays a crucial role in determining the device performances (e.g., external quantum efficiency, current efficiency, and lifetime through charge balance and efficiency energy transfer). Thus, designing host materials with these features remains a challenge for material scientists. One of the most widely used building blocks for host materials in blue PHOLEDs is a carbazole moiety having excellent hole-transporting property and large triplet energy.2 However, the carbazole structure shows poor electron-accepting property. To surmount this constraint, many host materials possessing bipolar properties for PHOLEDs have been developed. To construct bipolarity in the carbazole-based host material designs, a large number of moieties that are capable of accepting electrons, such as pyridine, triazole, triazine, phenanthroline, oxadiazole, benzimidazole, phosphine oxide, and phosphine sulfide, were incorporated to behave as bipolar host materials.3
As another electron acceptor, pyridoindole having a high triplet energy and better electron-accepting properties than carbazole has been developed to be a building block for improving the poor electron-accepting properties of the carbazole-based host materials.4 Several research groups have used the pyridoindole derivatives as the host materials for blue PHOLEDs and white tandem PHOLEDs to enhance their quantum efficiency.5 The pyridoindole moiety could enhance the quantum efficiency of blue PHOLEDs because of balanced charge density by electron accepting character of the pyridoindole moiety. Therefore, the incorporation of six-membered heterocycle in the indole moiety was an effective approach for good charge balance in the emitting layer. However, only a few six-membered heterocycle-modified indole moieties have been reported, and most of them were pyridine-modified indole moieties. In particular, no six-membered heterocycles with two nitrogen atoms were combined with indole to develop the electron accepting moieties of bipolar host materials.
In this work, a heterocycle with two nitrogen atoms, 5H-pyrazino[2,3-b]indole, was designed and synthesized as the electron-acceptor unit of carbazole-based host material because of its high electron affinity, attributed to symmetrically unsaturated imine nitrogen (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) structures.6 Here, we demonstrate a new carbazole-based bipolar host material containing a pyrazino[2,3-b]indole unit, 5-(3-(9H-carbazol-9-yl)phenyl)-5H-pyrazino[2,3-b]indole (CzPhPz), for the purpose of improving device performances and photophysical properties.
N) structures.6 Here, we demonstrate a new carbazole-based bipolar host material containing a pyrazino[2,3-b]indole unit, 5-(3-(9H-carbazol-9-yl)phenyl)-5H-pyrazino[2,3-b]indole (CzPhPz), for the purpose of improving device performances and photophysical properties.
The design of 5H-pyrazino[2,3-b]indole is based on the concept that the introduction of two nitrogen atoms in the six-membered ring combined with a indole moiety would improve the electron accepting properties as compared to the well-known carbazole moiety. The two sp2 nitrogen atoms make 5H-pyrazino[2,3-b]indole electron deficient and allow it to easily accept electrons. Because 5H-pyrazino[2,3-b]indole moiety can possess electron accepting and transport properties, the conjugated connection of the electron deficient 5H-pyrazino[2,3-b]indole and electron rich 9-phenylcarbazole may produce bipolar host materials.
First, the synthesis of the strong electron-acceptor unit, 5H-pyrazino[2,3-b]indole (Scheme 1), began with commercially available 1-acetyl-1H-indol-3(2H)-one, which reacted under LDA/−78 °C/N-phenyl-bis(trifluoromethanesulfonimide) condition to obtain triflate 1 (77%), which was subsequently treated with ethylenediamine in the presence of Pd(PPh3)4 in DMF to produce the desired 5H-pyrazino[2,3-b]indole 2 (70%). This transformation was confirmed (1H and 13C NMR) by the appearance of peaks at 8.40, 8.48 ppm and 141.6, 141.8 ppm (protons and carbons of C2 and C3 position), respectively. The synthesis of 5-(3-(9H-carbazol-9-yl)phenyl)-5H-pyrazino[2,3-b]indole (CzPhPz; 4) began with commercially available 9H-carbazole, which was N-arylated with 1,3-dibromobenzene using the Ullmann coupling reaction to obtain 3 (47%), which was subsequently treated with 5H-pyrazino[2,3-b]indole to produce the desired host material, CzPhPz (33%). The synthesis of the expected host materials was confirmed by 1H and 13C NMR, mass data. (See Fig. S1 and S2 in ESI†).
To compare the electronic character of building blocks used in this study, the molecular simulation of the carbazole and pyrazino indole moieties was carried out. (See in Fig. S3 and Table S1 in ESI†) As shown in Fig. S3,† the LUMO distribution of pyrazino indole was more dispersed than that of the carbazole unit. This means that the LUMO of pyrazino indole is stabilized compared to that of the carbazole unit because of the electron-withdrawing character of symmetric unsaturated imine nitrogen (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) structures in the heterocycle. Furthermore, the increased intramolecular charge transfer (ICT) characters of the pyrazino indole unit make the energy of HOMO and LUMO stabilized and the energy gaps between HOMO and LUMO decrease, which red-shifts the photoluminescence spectrum (see in Fig. S4 in ESI†). It can be presumed from the molecular simulation results that the electron accepting properties of pyrazino indole moiety are better than those of carbazole moiety.
N) structures in the heterocycle. Furthermore, the increased intramolecular charge transfer (ICT) characters of the pyrazino indole unit make the energy of HOMO and LUMO stabilized and the energy gaps between HOMO and LUMO decrease, which red-shifts the photoluminescence spectrum (see in Fig. S4 in ESI†). It can be presumed from the molecular simulation results that the electron accepting properties of pyrazino indole moiety are better than those of carbazole moiety.
The thermal properties of CzPhPz were investigated by DSC under a nitrogen atmosphere. The glass transition temperature (Tg) of the host material was obtained from the second heating scan of the cooled glassy sample after the first heating scan up to their melting temperature (Tm). Its Tg and Tm were found to be 83 °C and 188 °C, respectively (see in Fig. S5 in ESI†). The Tg is higher than that of the N,N′-dicarbazolyl-3,5-benzene (55 °C)7 because of an enhanced intermolecular interaction between electron-rich carbazole and electron-deficient heteroaromatic moiety. It is even higher than the Tg of 9-(3-(9H-carbazol-9-yl)phenyl)-9H-pyrido[2,3-b]indole (75 °C) with pyridine instead of pyrazine.5c One additional nitrogen of pyrazine strengthens the intermolecular interaction and increases the Tg of the host material.
Fig. 1 shows the spatial distributions of the HOMO and LUMO of the CzPhPz that was calculated with the Gaussian 03 program at the B3LYP/6-31G(d) level, using the density function theory (DFT) for the geometry optimizations. Triplet energy was calculated as the energy difference between the first excited triplet state and the ground singlet state. The calculated HOMO energy of CzPhPz was estimated to be −5.45 eV and the LUMO energy was −1.65 eV. We also calculated the triplet energy level to be 2.94 eV using the same basis set, suggesting an effective triplet exciton confinement of phosphorescent blue emitters in CzPhPz. As shown in Fig. 1, the HOMO of CzPhPz was localized on the carbazole unit, while the LUMO of CzPhPz was dispersed over the pyrazino indole unit. The HOMO and LUMO of CzPhPz were separated because of the hole transport character of carbazole and electron transport character of the pyrazino indole unit.
Fig. S6† shows the UV/vis absorption and solution photoluminescence (PL) spectra of CzPhPz in various solvents at room temperature. The UV/vis absorption spectra in 1,3-dioxane, CH2Cl2, and DMSO solvents are almost irrespective of solvent polarity. This implies that the Franck–Condon excited-state is subject to a rather small dipolar change with respect to the ground state. However, the peaks in the solution PL spectra significantly moved to a longer wavelength with increasing solvent polarity. This phenomenon can be explained by a mechanism involving rapid photoinduced electron transfer between the carbazole donor and the pyrazino indole acceptor units resulting in a large change in the dipole moment of the excited state; a subsequent solvent relaxation process leads to a solvent polarity-dependent emission.8 By plotting the emission peak frequencies of CzPhPz in various organic solvents as a function of solvent polarities, we obtained an almost linear relationship together with a slope as steep as −4179 cm−1, which is consistent with our assignment of a charge-transfer emission (see Fig. S7 in ESI†).
The photophysical properties of CzPhPz were estimated using UV/vis and PL spectrometer. UV/vis absorption, solution PL and low temperature PL emission spectra of CzPhPz are shown in Fig. 2. The UV/vis absorption spectrum of CzPhPz shows strong absorption peaks corresponding to the π–π* absorption of the carbazole linked pyrazino indole backbone appearing below 300 nm. Moreover, the host material exhibits weak absorption bands between 310 nm and 370 nm assigned to the n–π* transition of the carbazole and pyrazino indole moiety.9 The triplet energy of CzPhPz was 2.83 eV, which could be calculated from the first phosphorescent emission peak of low temperature (77 K) PL spectrum at 438 nm. The triplet energy was sufficiently high to use the host material as a blue phosphorescent dopant. The triplet energy of bis[2-(4,6-difluorophenyl)pyridinato-C2,N](picolinato)iridium(III) (FIrpic) dopant is 2.62 eV,10 and effective energy transfer from synthesized host material to FIrpic dopant is expected.
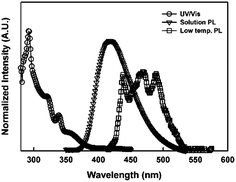 | ||
| Fig. 2 UV/vis, solution PL and low temperature PL spectra of CzPhPz (emission was excited at 340 nm). | ||
On the basis of the evaluated electrochemical oxidation onset potential, the HOMO level of CzPhPz was estimated to be −6.20 eV. Its LUMO level was −2.94 eV, calculated from the HOMO level and energy bandgap, determined from the UV/vis absorption threshold.
CzPhPz-based hole- and electron-only devices were fabricated to compare the hole and electron density in the host material. Fig. 3 shows the current density–voltage curves of hole- and electron-only devices bearing CzPhPz. The electron current density of CzPhPz was considerably higher than the hole current density, suggesting the electron transport character of the CzPhPz host material. The significant electron-deficiency of the pyrazine unit of 5H-pyrazino[2,3-b]indole moiety contributed to the electron transport character of CzPhPz. Although CzPhPz was developed as a bipolar host material, electron transport character was stronger than hole transport character.
The HOMO/LUMO and high triplet energy of CzPhPz enabled the fabrication of blue PHOLEDs using blue emitting FIrpic. The device performances of blue PHOLED with the CzPhPz host were investigated by changing the doping concentration of the blue-emitting FIrpic dopant from 5% to 30%. Fig. 4 shows the current density and luminance plots against the driving voltage of blue PHOLEDs with the CzPhPz. As shown in Fig. 4, the current density decreased with increasing doping concentration. This decreasing tendency is caused by the charge-trapping effect of the FIrpic dopant. Electrons are mostly trapped by FIrpic, and the current density was decreased because of the electron trapping at high doping concentration. The luminance showed a similar trend as that of the current density.
Quantum efficiency plots against luminance CzPhPz-based PHOLED devices are presented in Fig. 5. The quantum efficiency was calculated from the current density and luminance by assuming the Lambertian distribution of light emission. The quantum efficiency was increased with increasing doping concentration of host material, and the maximum quantum efficiency of 15.0% was obtained at 30% doping concentration. Moreover, the quantum efficiency at 1000 cd m−2 luminance was 11.2%. The high quantum efficiency of the highly doped device can be explained by the electron trapping of FIrpic dopant. In the single carrier device data, the CzPhPz host exhibited higher electron current density than hole current density. Therefore, holes and electrons balance in the emitting layer is improved by reducing the electron density in the emitting layer. As shown in the current density–voltage plots, the current density was decreased at high doping concentration because of the decrease of electron density caused by electron trapping by FIrpic dopant. The reduction of electron density improves charge balance and upgrades the quantum efficiency of the highly doped CzPhPy devices.
Blue emission spectra with the maximum emission peak at 473 nm were obtained from the blue PHOLEDs. (Fig. 6) Only FIrpic emission peak was detected with no other emission from CzPhPz, implying exciton confinement and complete energy transfer from CzPhPz to FIrpic dopant. The color coordinates of the CzPhPz devices are presented in Table S2.† Because of the intermolecular interaction between the dopant materials, the slight red-shift of the color coordinate occurred with increasing doping concentration.
In conclusion, CzPhPz with 5H-pyrazino[2,3-b]indole as an electron transport moiety was synthesized as the high triplet energy host material by coupling between 9-phenylcarbazole and 5H-pyrazino[2,3-b]indole. CzPhPz showed good electron transport properties and a high triplet energy of 2.83 eV. The application of CzPhPz compound as the host material for blue triplet emitter produced a blue PHOLED with a high quantum efficiency of 15.0%.
Acknowledgements
This research was supported by the research fund of Dankook University in 2013. (Grant no. 121652).Notes and references
- (a) M. A. Baldo, D. F. O'Brien, Y. You, A. Shoustikov, M. E. Thompson and S. R. Forrest, Nature, 1998, 395, 151 CrossRef CAS PubMed; (b) M. A. Baldo, S. Lemansky, P. E. Burrows, M. E. Thompson and S. R. Forrest, Appl. Phys. Lett., 1999, 76, 4 CrossRef PubMed; (c) A. Kohler, J. S. Wilson and R. H. Friend, Adv. Mater., 2002, 14, 701 CrossRef CAS; (d) S. R. Forrest, Org. Electron., 2003, 4, 45 CrossRef PubMed.
- H.-G. Jang, B.-S. Kim, J. Y. Lee and S.-H. Hwang, Dalton Trans., 2014, 7712 RSC.
- (a) S.-J. Su, C. Cai and J. Kido, Chem. Mater., 2011, 23, 274 CrossRef CAS; (b) M. K. Kim, J. Kwon, T.-H. Kwon and J.-I. Hong, New J. Chem., 2010, 34, 1317 RSC; (c) Z. Q. Gao, M. Luo, X. H. Sun, H. L. Tam, M. S. Wong, B. X. Mi, P. F. Xia, K. W. Cheah and C. H. Chen, Adv. Mater., 2009, 21, 688 CrossRef CAS; (d) Y. Tao, Q. Wang, L. Ao, C. Zhong, J. Qin, C. Yang and D. Ma, J. Mater. Chem., 2010, 20, 1759 RSC; (e) Z. Ge, T. Hayakawa, S. Ando, M. Ueda, T. Akiike, H. Miyamoto, T. Kajita and M. Kakimoto, Adv. Funct. Mater., 2008, 18, 584 CrossRef CAS; (f) S. O. Jeon, S. E. Jang, H. S. Son and J. Y. Lee, Adv. Mater., 2011, 23, 1436 CrossRef CAS PubMed.
- C. W. Lee and J. Y. Lee, Chem. Mater., 2014, 26, 1616 CrossRef CAS.
- (a) T. Motoyama, H. Sasabe, Y. Seino, J. Takamatsu and J. Kido, Chem. Lett., 2011, 40, 306 CrossRef CAS; (b) H. Fukagawa, N. Yokoyama, S. Irisa and S. Tokito, Adv. Mater., 2010, 22, 4774 CrossRef PubMed; (c) Y. Im and J. Y. Lee, Chem. Commun., 2013, 49, 5948 RSC; (d) S. J. Kim, Y. J. Kim, Y. H. Son, J. A. Hur, H. A. Um, J. Shin, M. Lee, J. Cho, J. K. Kim, S. Joo, J. H. Yang, G. S. Chae, K. Choi, J. H. Kwon and D. H. Choi, Chem. Commun., 2013, 49, 6788 RSC; (e) C. W. Lee, Y. Im, J. Seo and J. Y. Lee, Org. Electron., 2013, 14, 2687 CrossRef CAS PubMed.
- (a) T. Yamamoto, Z. H. Zhou, T. Kanbara, M. Shimura, K. Kizu, T. Maruyama, Y. Nakamura, T. Fukuda, B. L. Lee, N. Ooba, S. Tomaru, T. Kurihara, T. Kaino, K. Kubota and S. Sasaki, J. Am. Chem. Soc., 1996, 118, 10389 CrossRef CAS; (b) G. R. Newkome and W. W. Paudler, Comtemporary Heterocyclic Chemistry, John Wiley, New York, 1982 Search PubMed.
- S. J. Yeh, M. F. Wu, C. T. Chen, Y. H. Song, Y. Chi, M. H. Ho, S. F. Hsu and C. H. Chen, Adv. Mater., 2005, 17, 285 CrossRef CAS.
- (a) Y. Y. Chien, K. T. Wong, P. T. Chou and Y. M. Cheng, Chem. Commun., 2002, 2874 RSC; (b) K. T. Wong, S. Y. Ku, Y. M. Cheng, X. Y. Lin, Y. Y. Hung, S. C. Pu, P. T. Chou, G. H. Lee and S. M. Peng, J. Org. Chem., 2006, 71, 456 CrossRef CAS PubMed.
- W.-W. Zhang, W.-L. Mao, Y.-X. Hu, Z.-Q. Tian, Z.-L. Wang and Q.-J. Meng, J. Phys. Chem. A, 2009, 113, 9997 CrossRef CAS PubMed.
- C. Adachi, R. C. Kwong, P. Djurovich, V. Adamovich, M. A. Baldo, M. E. Thompson and S. R. Forrest, Appl. Phys. Lett., 2001, 79, 2082 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra10307f |
| This journal is © The Royal Society of Chemistry 2014 |