Effect of selective oxidation of bacterial cellulose on degradability in phosphate buffer solution and their affinity for epidermal cell attachment
Xiangning Shi,
Qiuyan Cui,
Yudong Zheng*,
Shuai Peng,
Guojie Wang and
Yajie Xie
School of Materials Science and Engineering, University of Science and Technology Beijing, Beijing 100083, P. R. China. E-mail: zhengyudong@mater.ustb.edu.cn; Fax: +86-010-62332336; Tel: +86-010-62330802
First published on 17th October 2014
Abstract
Bacterial cellulose (BC) has a very promising application in biomedical engineering due to its three dimensional nano-network and good biocompatibility. However, it is difficult for BC to degrade in vivo without cellulase, which has limited its potential application. In this work, oxidized bacterial cellulose (OBC) was prepared according to selective oxidation with NO2 gas. The structure and micromorphology of OBC were characterized by FTIR, XRD, and SEM. The results showed that the oxidation did not break the crystal structure or alter the crystallinity of BC. OBC still maintained the 3D nano-fibrils network, whereas the diameter of each fiber in the nano-fibrils network of OBC became wider. When immersed in PBS, OBC degraded gradually, and the mass loss rate and degradation rate of OBC were much higher than those of BC after degradation for 60 days. Degradation occurred from surface to inside and the oxidized part of the network favored the process. Results of cell adhesion and proliferation studies also revealed that OBC had excellent cellular affinity similar to BC.
1. Introduction
As a polymeric material with nanofiber-network structure, BC has various properties such as high water holding capacity, high crystallinity, high tensile strength, good biocompatibility as well as in situ moldability and low production cost,1–3 which has made it to be one of the most promising tissue engineering scaffold materials. At present, BC is widely used in wound covering, brain membrane, skin of tissue engineering and renovating artificial blood vessels and cornea.4–6 However, the degradability of BC in human body has become one of the key factors that constrain its potential application in tissue engineering, since there is no cellulase in body fluids.Oxidization of cellulose is an effective method to modify the chemical structure in order to improve its degradability in vivo for biomedical applications.7 Cellulose has already been oxidized by different oxidants such as TEMPO–NaClO–NaBr,8–10 NaIO4,11,12 NO2,13,14 and HNO3/H3PO4/NaNO2,15 through which the primary alcohol groups at C6 were selectively oxidized into carboxyl during the oxidation process. Oxidized cellulose can be used as Surgicel to stop bleeding.16 Moreover, the oral adsorbent, which is obtained from 2,3-dialdehyde cellulose compounds, can also function well in treating Chronic Renal Failure.17 Recently, 2,3-dialdehyde bacterial cellulose (DABC) oxidized by periodate was prepared,18 which made its degradation rate in PBS was faster than that of BC, but the crystallinity and mechanical strength were much lower. Nitrogen dioxide (NO2) is considered as a more suitable oxidant for cellulose due to its excellent selectivity19 among the oxidants as secondary reactions were almost avoided. We have used NO2 to oxidize bacterial cellulose in gaseous environment for different time and obtained OBC with carboxyl groups at C6.20 The oxidation did not affect the crystal structure of BC, but changed the morphology of its network with wider diameter. Results showed that the oxidation reaction was controllable for regulating the degree of degradation of BC. However, structural changes, aggregative state during degradation, and cellular affinity of OBC have not been studied yet. Moreover, the degradation rate of OBC according to glucose concentration in PBS was not detected either.
In this work, we focused on further discussing and analyzing the degradation of oxidized bacterial cellulose. The degradation performance in vitro, including changes in degradation rate, chemical and crystal structural and micromorphology changes of OBC and BC, were characterized and compared. Simultaneously, the degradability of OBC was analyzed and degradation mechanism was then proposed, and the cellular affinity of OBC was also evaluated and compared with that of BC.
2. Experiments
2.1 Preparation of OBC
Hainan Yida Food Co, Ltd was supplied by the BC membranes. As a pre-treatment, BC membranes were immersed in 10% NaOH solution for 30 min at 80 °C to remove the bacterial cell debris, and then thoroughly washed to neutral by de-ionized water. NO2 was produced by adding a suitable amount of Cu to superfluous HNO3 (65 wt%) solution, and NO2 reacted with purified BC (15 mm in diameter and 1 mm in thickness) for 3, 6, 9 and 12 days as per a previously used method.2.2 Degradability in vitro
The degradation of BC and OBC was tested in vitro in phosphate buffered saline solution (PBS, pH = 7.4) at 37 °C. The films of BC and OBC were cut into circular slices with a diameter of 15 mm, and then immersed in PBS in centrifuge tubes. The samples were taken out and washed by de-ionized water separately after degradation for 3, 6, 9, 12, 15, 18, 21, 24, and 30 days, and then freeze-dried and weighed. The original mass of each sample was designated as m0, whereas the mass after degradation was characterized as m1. Note that the mass loss rate is the ratio of reduced mass and original mass, which was calculated by the eqn (1).Ultraviolet spectrophotometer (Unico, UV-2100) was used to test the glucose content as degradation product. Its absorbance at 470 nm has a linear relationship with the concentration of the glucose solution. Degradation rate is the ratio of actual glucose mass and original mass, which was measured to analyze the degree of degradation of OBC. The concentration of glucose was noted as C and the volume of the degradation solution was denoted as V. Note that the degradation rates were calculated by eqn (2).21
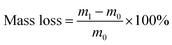 | (1) |
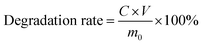 | (2) |
Special software Spss 13 was also used to statistically analyze the data of mass loss and concentration of glucose related to the degradation rate. The data between three groups were analyzed with variance. All data were represented by average ± standard deviation (![[x with combining macron]](https://www.rsc.org/images/entities/i_char_0078_0304.gif) ± sd).
± sd).
2.3 Cellular affinity test
Three groups of materials (named group a: blank control, group b: OBC, group c: BC) were cut into discs with a diameter of 15 mm and heat-sealed after packaging in plastic zip-lock bags. Samples were then sterilized under 18.40 kGy with the radiation of Co60. Experiments of cell cultivation were performed in compliance with the relevant laws and institutional guidelines and approved by No. 1 Hospital, which is affiliated to General Hospital of the Chinese People's Liberation Army.Newborn fetal rat was executed and immersed in 75% ethanol for 3 min. After that, subsequent operations were all under aseptic conditions. Torso skin was sheared off from the body of the rat. Then, the torso skin was cut into pieces of 0.5 cm2 and immersed in a mixture of 0.25% trypsin and 0.02% EDTA for 16 h at 4 °C in refrigerator. Then, epidermis was removed from the dermis and placed into a centrifugal tube containing Dulbecco's modified Eagle's medium (DMEM). Epidermal cells formed a cell suspension by blowing with a straw. The original number of the epidermal cells was counted microscopically and centrifugation treatment at the speed of 1000 rpm for 5 min was performed subsequently. DMEM solution containing 10% tire bovine serum was added to the suspension, adjusting the density of cells to 1 × 104 mL−1. The suspension was then inoculated in 24-hole training board coated with 1 mL mouse-tail collagen per hole in advance.
Three groups of materials were placed in holes after suspension's sedimentation for 1 h. They were then incubated in hatch box under the condition of 5% CO2 at 37 °C. Culture solution was renewed at the time interval of 4 and 7 days respectively. After 1, 4, 7, 10 days, cells cultivated with different materials were taken out and observed under an optical microscope, and then the cell numbers were microscopically counted and cell viability was tested through the MTT method.
2.4 Characterization
The microstructure of BC and OBC was observed by scanning electron microscopy (SEM, Apollo 300, and 10 kV). All of the samples were freeze-dried and coated with a thin layer of gold in a sputter coater in advance.The chemical structure of BC and OBC before and after degradation for different time intervals was tested by Fourier transform infrared spectroscopy (FTIR, NICOLET 750) with a range of frequency from 4000 cm−1 to 450 cm−1 and a resolution of 4 cm−1.
X-ray diffraction (XRD, D/MAX-RB, 20 kV, 40 mA) with Cu Kα radiation (λ = 0.154 nm) was used to examine the crystal structure of the samples, and the range of diffraction angles (2θ) was 10°–40°.
3. Results and discussion
3.1 Mass loss and degradation rate
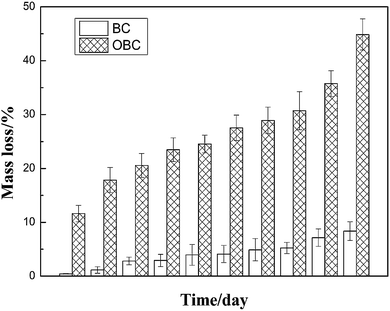
The mass loss rate and degradation rate are often used to assess the degradation of biomaterials in vitro.22 When immersed in PBS, both BC and OBC began to degrade from surface to inside, and degraded debris and fragments dropped from original films of BC and OBC, leading to the mass loss of original materials. The degradation product of cellulose, as well as for oxidized cellulose, is glucose; therefore, glucose content represents their biodegradability. The glucose concentration was determined by the absorbance at 470 nm. The glucose concentrations of different degradation stages were measured accurately, and the degradation rates were calculated according to eqn (2).Fig. 1 shows the mass loss of BC and OBC. Just as expected, the mass loss rate of OBC was much higher than that of BC obviously. For BC, the mass loss rate was less than 10% (wt) even after 60 days in PBS solution (Fig. 1), which was consistent with the results reported by Peng20 and Li.18 For OBC, however, it even rose up to 45% with increase in time. For DABC, as reported by Li,18 the mass loss rate was about 80% after 1000 h (about 41.5 days). Apparently, the mass loss rate of OBC in Fig. 1 was lower than that of 2,3-dialdehyde bacterial cellulose (DABC). This is mainly because oxidation by NO2 is much milder19 than that of periodate and degradation accompanied with oxidation was lower. Moreover, OBC with 45% mass loss after degradation in vitro for 60 days still maintained its initial shape and partial mechanical properties, indicating more or less that the oxidation occurring in the BC membrane was uniform and selective one. Furthermore, the high crystalline degree and less crystal defects of OBC may also help to interpret its slow degradation speed in comparison with electrospun plant cellulose oxidized by NO2.23
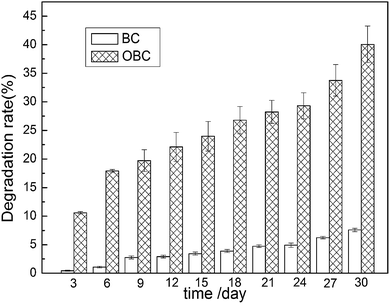
The existence of glucose in PBS solution suggests that the C–O–C bond became weaker and cracked after degradation, which contributed to the production of small molecular glucose as well as the degradation of BC and OBC. The degradation rate of BC and OBC (Fig. 2) maintained the same trend with mass loss rate as time passed. The degradation rate of BC was less than 8% after 60 days, whereas for OBC, it came up to 40% (Fig. 2), indicating that degradation of OBC in PBS solution was greatly sped up and OBC was easier to degrade in PBS solution with its characteristic carboxyl groups at C6 because of selective oxidization.
Another significant phenomenon is that during the early stages of degradation, the mass loss rate was always higher than the degradation rate for BC as well as for OBC. This is because mass loss was calculated according to the mass before and after degradation, whereas degradation rate was determined through the glucose concentration in PBS. When materials began to degrade, small fragments such as fiber bundles left the materials, which causes mass loss. However, these bundles do not decompose into glucose completely at once, leading to a relatively lower degradation rate than mass loss rate. Overall, there was no big difference between mass loss and degradation rate, since most of the fiber bundles finally got degraded into glucose after falling off. Therefore, we are sure that BC and OBC have degraded in PBS solution and OBC possess a higher degradation rate.
3.2 Structural changes of BC and OBC after degradation
The hydroxyl groups at the position C6 of cellulose could convert to carboxyl groups when selectively oxidized by NO2. Fig. 3 shows the chemical structure of OBC together with BC before and after degradation for different time intervals.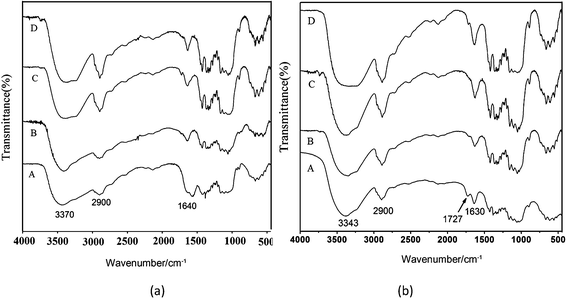 | ||
| Fig. 3 FTIR spectra of (a) BC and (b) OBC degraded in PBS for (A) 0 day, (B) 12 days, (C) 30 days, (D) 60 days. | ||
For original BC, there are three absorption peaks at 3370, 2900, and 1060 cm−1, corresponding to the stretching vibrations of the group ν(–OH), ν(–CH2) and ν(C–O), respectively (Fig. 3a, curve A).24,25 Compared with FTIR curve of original BC, new absorption peaks appeared at 1727 cm−1, which can be assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of carboxyl groups after oxidation, indicating the existence of the oxidation product, i.e. carboxyl cellulose. Moreover, the intensity of the absorption peaks near 1320–1210 cm−1 associated with the C–O groups decreased, further proving the formation of OBC (Fig. 3b, curve A). After degradation in PBS for different time length, the peaks of the hydroxyl groups of BC were obviously strengthened (Fig. 3a, curve B, C and D; Fig. 3b, curve B, C and D), whereas the typically characteristic peak of OBC appeared at 1727 cm−1, corresponding to the disappearance of C
O stretching vibration of carboxyl groups after oxidation, indicating the existence of the oxidation product, i.e. carboxyl cellulose. Moreover, the intensity of the absorption peaks near 1320–1210 cm−1 associated with the C–O groups decreased, further proving the formation of OBC (Fig. 3b, curve A). After degradation in PBS for different time length, the peaks of the hydroxyl groups of BC were obviously strengthened (Fig. 3a, curve B, C and D; Fig. 3b, curve B, C and D), whereas the typically characteristic peak of OBC appeared at 1727 cm−1, corresponding to the disappearance of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration (Fig. 4b curve D). This indicates that more hydroxyl groups were formed due to the breaking of C–O–C bond and the degradation occurred at the position of C6.
O stretching vibration (Fig. 4b curve D). This indicates that more hydroxyl groups were formed due to the breaking of C–O–C bond and the degradation occurred at the position of C6.
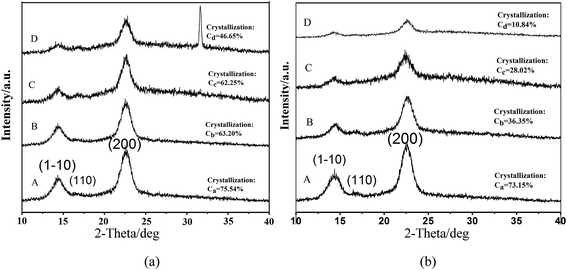 | ||
| Fig. 4 XRD patterns of (a) BC and (b) OBC immersed in PBS for (A) 0 day, (B) 12 days, (C) 30 days, (D) 60 days. | ||
3.3 Crystal structure comparison of BC and OBC after degradation
Fig. 4 shows the XRD patterns of BC and OBC degraded for different times. For original BC (Fig. 4a curve A), three peaks were located at diffraction angles of 14.6°, 16.5° and 22.5°, corresponding to the primary diffraction of the crystal plane (1−10), (110) and (200), which shows the structure of a well-defined cellulose I crystal.26–29 The diffraction peaks of OBC according to Fig. 4b curve A are almost the same as those of original BC, indicating that OBC still retained the crystal structure of cellulose I and the oxidation had little impact on the crystal structure as well as their crystallinity.30When immersed in PBS solution for different time intervals, the crystallinity of both BC and OBC decreased, and the crystallinity of BC decreased from 75.54% to 46.65% as the time interval was extended from 0 day to 60 days (Fig. 4a), whereas for OBC, it ranged from 73.15% to 10.84% (Fig. 4b). This is because with the presence of carboxyl groups and higher activity, a long-term degradation of OBC enabled water molecules to permeate into inner crystalline regions, leading to a loose arrangement of molecular chains and expanded amorphous regions.31,32 At the same time, degradation in amorphous regions was under way. All these above helped OBC drop crystallinity. Moreover, the intensity of the diffraction peaks mentioned above weakened as time passed, especially for OBC. The peaks at crystal plane (1−10) of OBC nearly disappeared after immersion for 30 days and 60 days (Fig. 4b curve D). Obviously, the crystallinity of OBC decreased more sharply compared with BC, indicating that OBC was much easier to degrade in PBS solution because of the active action of carboxyl groups at C6 position. Note that a similar phenomenon was also reported by Li18 and Calvini.33
3.4 Surface morphology
Fig. 5 shows the SEM images of BC and OBC before and after degradation for 60 days. BC has a dense 3-dimensional network structure of the nanofibrils, which was almost retained after oxidation. Moreover, the nanofibrils of OBC appeared to aggregate or fuse into bundles gradually after oxidization for different time (Fig. 5b–d). In addition, a few fibrils began to be fragmentized by the oxidation process (Fig. 5d); i.e., oxidation was accompanied by a light degree of deterioration visible on oxidized fibers, which was similar to the phenomenon described by Kim15 and Kumar.34After degradation in PBS solution for 60 days, BC maintained the original nano-fibrous networks with a few fibrils ruptured (Fig. 5e), while for OBC oxidized for 3 days, more fibrils ruptured and its network became loose (Fig. 5f). The 3D nano-fibrous network of OBC oxidized for 6 days partly disappeared, and many fibrils fracture and fell into pieces (Fig. 5g) because of the accelerated degradation of OBC. More OBC fibers eroded and the nano-fibrous network almost collapsed after degradation for 60 days for OBC oxidized for 12 days (Fig. 5h), which was inconsistent with its high mass loss rate and degradation rate of above 40%. Obviously, the oxidization of BC by NO2 sped up the degradation of OBC, which could gradually destruct nano-fibrous network of OBC at a desirable degradation rate by controlling the time.
3.5 Discussion and analysis on degradation mechanism
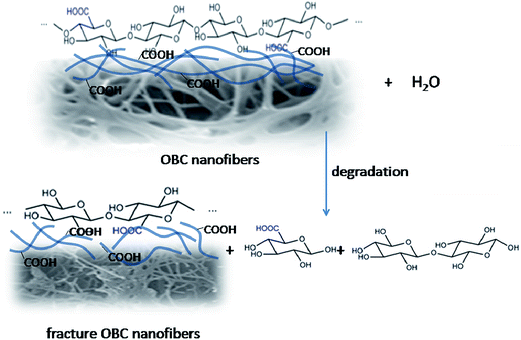
It is well known that BC degrades very slowly without cellulase; however, oxidization can help to improve its degradability, which has been proved by the results above. When immersed in PBS, both BC and OBC would swell from surface to inside, and the crystallinity and the binding force of hydrogen bonds within and between molecular chains would decline,35 leading to the fracture of hydrogen bonds at the position of C2, C3 and C6, which could induce the formation of new hydrogen bonds with water molecule. For both BC and OBC, swelling first began from amorphous regions and the surface of crystalline regions during degradation; i.e., the degradation occurred from amorphous regions and surface of crystalline regions, and then moved to the inner crystalline regions with increase in time. The breakage of the structure progressed from crystalline regions to amorphous ones, which leads to the decrease in crystallinity and causes the wideness and excursion of hydroxyl groups peaks (Fig. 3). This process contributed to the further swelling and degradation of cellulose macromolecular chain as a long-term swelling enabled water molecules to permeate into the inner regions and turned it to be a loose arrangement of molecular chains with expanded amorphous regions.36 Then, the C–O–C bond between the monomers was broken and degraded parts dropped from the surface, leading to mass loss. With the degradation of the surface parts, the original inner parts turned out to be surface and expose to PBS to repeat the process. This regular pattern of degradation is in line with that of 2,3-dialdehyde bacterial cellulose reported by Kim12 and Calvini.33What made OBC degrade faster than BC is that after oxidation, carboxyl groups of OBC at C6 possess higher energy as well as better activity. Moreover, the carboxyl groups are easier to rotate around C5–C6 bond than that of the hydroxyl group, which makes the bond between monomers of OBC more liable to fracture than BC. The degradation process of OBC is shown in Fig. 6. Molecule chains of OBC first break into short chains with different degrees of polymerization, and then these shorter chains finally get degraded into glucose. This phenomenon was close to the reported value for the periodate oxidation modified on BC.20,33 Moreover, OBC with better hydrophilicity because of the carboxyl group at C6 enables OBC to absorb more water molecules, which will also contribute to its hydrolysis. Furthermore, as mentioned in 3.3, the crystallinity of BC was 75.54%, whereas the crystallinity of OBC was 73.15%. With the presence of carboxyl groups and higher activity, OBC enabled water molecules to permeate into inner crystalline regions and led to a loose arrangement of molecular chains and expanded amorphous regions, which caused a faster degradation. Carboxyl groups of OBC, better hydrophilicity and lower crystallinity helped to drop crystallinity down. The disappearance of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O peaks (Fig. 4b) proved that the molecular chains break into terminal groups at the position of carboxyl group when they were degraded in PBS solution, which lead to the reduction as well as complete vanishing of the peaks of the carboxyl groups.
O peaks (Fig. 4b) proved that the molecular chains break into terminal groups at the position of carboxyl group when they were degraded in PBS solution, which lead to the reduction as well as complete vanishing of the peaks of the carboxyl groups.
Crystalline region contributes greatly to the strength of cellulose37 and mechanical properties of the cellulose often deteriorate by secondary oxidation process.25 Therefore, it is necessary to investigate the mechanical properties and other physical properties of OBC oxidized by NO2, which is an ongoing study.
3.6 Cells cultivation
Direct culture method was adopted to evaluate and compare the cellular affinity of OBC with BC. The epidermal cells of fetal rat were co-cultivated with three groups of materials (a: blank control, b: OBC and c: BC) for different time. After seeding epidermal cells on these samples, the cell proliferations status and cytomorphology after cultivation for 1, 4, 7, and 10 days are shown in Fig. 8.Obviously, non-transparent small cells with an orbicular shape observed under optical microscope were epidermal cells while others were fibroblast ones. On the whole, the amount of the cells rose continuously as time increased. After cultivation for 1 day, a small number of cells were distributed in the DMEM solution (Fig. 7). The cell number of group a was more than those of group b and group c. After 4 days, more colonies of epidermal cells in three groups appeared together with several fibroblast cells, and the cells were gathered and overlapped mutually. Note that the cells on the edge grew outside with a form of single layer, contributing to the extension of colonies (Fig. 7). Orbicular epidermal cells and spindly fibroblasts proliferated rapidly after 7 days. Epidermal cells aggregated into small areas, and at the same time, there were no significant differences among the groups. After 10 days, fibroblast cells proliferated abundantly, especially for group a, in which fibroblast cells almost filled the whole area. For group b and group c, more epidermal cells grew and their numbers increased apparently (Fig. 7). The total number of epidermal cells and fibroblast ones in each group became almost the same finally, suggesting that BC and OBC both support cell proliferation.
3.7 Cellular affinity evaluation
Fig. 8a shows a specific number of epidermal cells during proliferation when cultivated in vitro. Similar to the results observed in Fig. 8, the number epidermal cells grew slowly during early days and then grew fast. The number of epidermal cells cultivated with three groups of materials all slightly rose after one-day proliferation and cells of group a is a little more than that of b and c. After 4 days, the number of epidermal cells in all three groups increased slightly, and the number of epidermal cells in group a is still the most. When it comes to 7 days, epidermal cells proliferated faster than before and the differences among the three groups diminished (P < 0.05). When cultivated for 10 days, the distinction among the cells numbers of three groups almost disappeared (P < 0.05). On the whole, cell growth of the three groups share consistent trend without great differences, suggesting that BC and OBC have little impact on cell proliferation and they both support cell proliferation with good cellular affinity.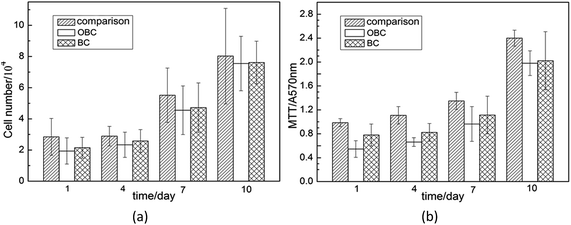 | ||
| Fig. 8 Cellular affinity of OBC compared with blank control and BC (a) epidermal cell numbers during the proliferation, (b) MTT of cells cultivated with different materials. | ||
The MTT data describe the relative viability of cells, including epidermal cells as well as fibroblast cells growing on the surface of different materials. The data are comparable since the same numbers of cells were added to the samples. The consequence of MTT colorimetric estimation is shown in Fig. 8b. MTT absorbance value of group a kept the most all along the cultivation. MTT values of cells cultivated for 4 days were almost the same with that of cells cultivated for 1 day. When 7 days passed, the data increased a little, whereas for 10 days, the MTT values rose up to a high level due to the proliferation of both epidermal cells and fibroblast cells. These materials hardly affect proliferation of epidermal cells. As absorbance result of MTT colorimetric estimation is in line with cell growth number to same extent, we can arrive at the conclusion that OBC and BC support cell proliferation with good biocompatibility, which extends their potential application in vivo as implanted materials.
4. Conclusions
OBC oxidized by NO2 in aphotic condition maintains the original 3D nano-fibrous network crystal structure of BC with relatively wider diameter and it is prone to degrade in PBS solution more easily according to mass loss and degradation rate compared with BC. For OBC, the mass loss rate went up to 45% after 60 days while it was only approximately 10% for BC. FTIR spectra of OBC immersed in PBS for different time intervals also suggest that the degradation procedure of OBC first occurs from amorphous regions and surface of crystalline regions, and then moves to the inner crystalline regions. Carboxyl groups with higher energy and better activity are easier to rotate around bond of C5–C6 than hydroxyl group, making the bond between monomers of OBC more liable to break than BC. Note that oxidation has little effect on the crystal structure of bacterial cellulose. After degradation in PBS solution, crystallinity of OBC decreased sharply, indicating that OBC is much easier to degrade in PBS solution and further proved the degradation mechanism. According to SEM micrographs, OBC oxidized for different time degraded at varying degrees with the fibrils fragmentized more or less. When cultivated with epidermal cells of fetal rat, OBC showed a good cellular affinity, contributing to a wider application in tissue engineering. Consequently, the selective oxidation by nitrogen dioxide has been proven to be a practical method for the modification of BC's degradability without weakening its biocompatibility and crystal structure.Acknowledgements
This study is financially supported by National Natural Science Foundation of China Project (Grant no. 51273021, 51473019).References
- A. Svensson, E. Nicklasson, T. Harrah, B. Panilaitis, D. Kaplan, M. Brittberg and P. Gatenholm, Biomaterials, 2005, 26, 419–431 CrossRef CAS PubMed.
- K. Watanabe, Y. Eto, S. Takano, S. Nakamori, H. Shibai and S. Yamanaka, Cytotechnology, 1993, 13, 107–114 CrossRef CAS.
- G. Helenius, H. Bäckdahl, A. Bodin, U. Nannmark, P. Gatenholm and B. Risberg, J. Biomed. Mater. Res., Part A, 2006, 76, 431–438 CrossRef PubMed.
- W. Czaja, A. Krystynowicz, S. Bielecki and R. M. Brown Jr, Biomaterials, 2006, 27, 145–151 CrossRef CAS PubMed.
- H. Fink, J. Hong, K. Drotz, B. Risberg, J. Sanchez and A. Sellborn, J. Biomed. Mater. Res., Part A, 2011, 97, 52–58 CrossRef PubMed.
- D. Klemm, D. Schumann, U. Udhardt and S. Marsch, Prog. Polym. Sci., 2001, 26, 1561–1603 CrossRef CAS.
- R. L. Stilwell, M. G. Marks, L. Saferstein and D. M. Wiseman, Handb. Biodegrad. Polym., 1998, 7, 291–306 Search PubMed.
- T. Saito, S. Kimura, Y. Nishiyama and A. Isogai, Biomacromolecules, 2007, 8, 2485–2491 CrossRef CAS PubMed.
- A. Isogai, T. Saito and H. Fukuzumi, Nanoscale, 2011, 3, 71–85 RSC.
- Y. Kato, J. Kaminaga, R. Matsuo and A. Isogai, Carbohydr. Polym., 2004, 58, 421–426 CrossRef CAS PubMed.
- A. Varma and M. Kulkarni, Polym. Degrad. Stab., 2002, 77, 25–27 CrossRef CAS.
- U. J. Kim, S. Kuga, M. Wada, T. Okano and T. Kondo, Biomacromolecules, 2000, 1, 488–492 CrossRef CAS.
- S. Coseri, G. Nistor, L. Fras, S. Strnad, V. Harabagiu and B. Simionescu, Biomacromolecules, 2009, 10, 2294–2299 CrossRef CAS PubMed.
- L. Saferstein and G. Serafica, US Pat., 7645874 B2, 2010 Search PubMed.
- C.-W. Kim, D.-S. Kim, S. Y. Kang, M. Marquez and Y. L. Joo, Polymer, 2006, 47, 5097–5107 CrossRef CAS PubMed.
- M. C. Henry, D. B. Tashjian, H. Kasowski, C. Duncan and R. L. Moss, J. Pediatr. Surg., 2005, 40, 9–11 CrossRef PubMed.
- M. Yagi, S. Kato, T. Nishitoba, H. Sato, N. Kobayashi, N. Iinuma and N. Nagano, Nephron, 1998, 78, 433–439 CrossRef CAS PubMed.
- J. Li, Y. Wan, L. Li, H. Liang and J. Wang, Mater. Sci. Eng., C, 2009, 29, 1635–1642 CrossRef CAS PubMed.
- S. Camy, S. Montanari, A. Rattaz, M. Vignon and J.-S. Condoret, J. Supercrit. Fluids, 2009, 51, 188–196 CrossRef CAS PubMed.
- S. Peng, Y. Zheng, J. Wu, Y. Wu, Y. Ma, W. Song and T. Xi, Polym. Bull., 2012, 68, 415–423 CrossRef CAS.
- Y. Chen, T. Xi, Y. Zheng, T. Guo, J. Hou, Y. Wan and C. Gao, J. Bioact. Compat. Polym., 2009, 24, 137–145 CrossRef CAS PubMed.
- A. Oyane, H. M. Kim, T. Furuya, T. Kokubo, T. Miyazaki and T. Nakamura, J. Biomed. Mater. Res., Part A, 2003, 65, 188–195 CrossRef PubMed.
- M. S. Khil, H. Y. Kim, Y. S. Kang, H. J. Bang, D. R. Lee and J. K. Doo, Macromol. Res., 2005, 13, 62–67 CrossRef CAS.
- Y. Maréchal and H. Chanzy, J. Mol. Struct., 2000, 523, 183–196 CrossRef.
- M. Kačuráková, A. C. Smith, M. J. Gidley and R. H. Wilson, Carbohydr. Res., 2002, 337, 1145–1153 CrossRef.
- V. Klechkovskaya, Y. G. Baklagina, N. Stepina, A. Khripunov, P. Buffat, E. Suvorova, I. Zanaveskina, A. Tkachenko and S. Gladchenko, Crystallogr. Rep., 2003, 48, 755–762 CrossRef CAS.
- C. Rambo, D. Recouvreux, C. Carminatti, A. Pitlovanciv, R. Antônio and L. Porto, Mater. Sci. Eng., C, 2008, 28, 549–554 CrossRef CAS PubMed.
- L. Yu, J. Lin, F. Tian, X. Li, F. Bian and J. Wang, J. Mater. Chem. A, 2014, 2, 6402–6411 CAS.
- J. Lin, L. Yu, F. Tian, N. Zhao, X. Li, F. Bian and J. Wang, Carbohydr. Polym., 2014, 109, 35–43 CrossRef CAS PubMed.
- V. Chavan, B. Sarwade and A. Varma, Carbohydr. Polym., 2002, 50, 41–45 CrossRef CAS.
- M. H. Lee, H. S. Park, K. J. Yoon and P. J. Hauser, Text. Res. J., 2004, 74, 146–154 CrossRef CAS PubMed.
- L. Hong, Y. Wang, S. Jia, Y. Huang, C. Gao and Y. Wan, Mater. Lett., 2006, 60, 1710–1713 CrossRef CAS PubMed.
- P. Calvini, A. Gorassini, G. Luciano and E. Franceschi, Vib. Spectrosc., 2006, 40, 177–183 CrossRef CAS PubMed.
- V. Kumar and Y. Tianrun, Carbohydr. Polym., 2002, 48, 403–412 CrossRef CAS.
- W. Mormann and U. Michel, Carbohydr. Polym., 2002, 50, 349–353 CrossRef CAS.
- H. Yamamoto and F. Horii, Macromolecules, 1993, 26, 1313–1317 CrossRef CAS.
- F. Goelzer, P. Faria-Tischer, J. Vitorino, M.-R. Sierakowski and C. Tischer, Mater. Sci. Eng., C, 2009, 29, 546–551 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |