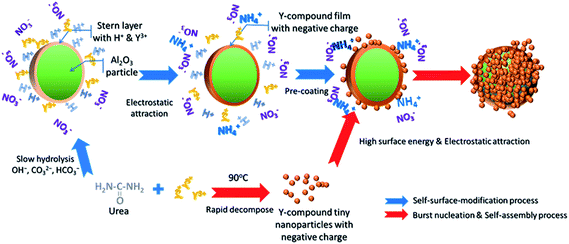
Al2O3/yttrium compound core–shell structure formation with burst nucleation: a process driven by electrostatic attraction and high surface energy†
Daidong Guo‡
a,
Lili Zhao‡a,
Yuanhua Sang*a,
Hong Liu*a,
Shaohong Liub and
Xudong Sunb
aState Key Laboratory of Crystal Materials, Shandong University, 27 Shandanan Road, Jinan, 250100, China. E-mail: sangyh@sdu.edu.cn; hongliu@sdu.edu.cn
bSchool of Materials and Metallurgy, Northeastern University, Shenyang, 110004, China
First published on 13th October 2014
Abstract
A partial wet chemical route has been developed, in which aluminium oxide (Al2O3) nanoparticles in a Y(NO3)3 solution are induced to form a core–shell-structured yttrium aluminum garnet (YAG) precursor based on a burst nucleation synthesis. By simulating the reaction conditions with urea and ammonium bicarbonate as precipitants, a two-step mechanism involving different dynamic processes is proposed to describe the core–shell structure formation. During the majority of the process, the electrostatic attraction between the opposite surface charges of Al2O3 and nanoparticles of the Y-compound is regarded as the single driving force. However, the high surface energy of Y-compound nanoparticles originating from the burst nucleation process plays a key role in completing the formation of the core–shell structure. The precursor obtained can be transformed into pure YAG nanoparticles, which retain the morphology of the Al2O3 template, and these are of sufficiently high quality for the preparation of transparent ceramics. An understanding of the mechanism makes this a novel method for the synthesis of the nanostructured core–shell binary oxide precursor.
Introduction
Binary oxides have attracted considerable attention because of their extensive potential applications. For example, LaMnO3 is employed as an electrolyte in solid oxide fuel cells,1 and Y3Fe5O12 is used in laser alignment and microwave devices, and is also an important fundamental component of circulators, phase shifters, and modulators.2 Transparent magnesium aluminate (MgAl2O4) ceramic is an excellent optical material in the ultraviolet, visible and infrared bands and is used as bow cap and window materials.3 Materials with the correct nanoparticle morphology are essential for high performance. The control of morphology in the synthesis of the complex oxides has therefore become a hot topic.4,5 As one of the most attractive materials involved, the formation of yttrium aluminum garnet (YAG) nanoparticles has been extensively studied in order to achieve a well-controlled morphology, including sol–gel methods,6,7 homogeneous precipitation,8,9 co-precipitation methods,10,11 microwave irradiation12,13 and solvothermal methods.14,15In practice, control of the morphology of the yttrium–aluminium (Y–Al) precursor is difficult because of the hydrolytic character of Al3+ ions, which induces the formation of a flocculated sediment during co-precipitation.16 For example, the YAG precursor nanoparticles with a gradient distribution of Y and Al elements are obtained by adding ammonium bicarbonate (NH4HCO3) solution to the Y–Al solution in droplet form, whereas a hollow morphology is produced by adding the Y–Al solution into the NH4HCO3 solution.17 In addition, a number of other factors can affect the morphology and composition of the YAG precursor during synthesis, including the nature of the precipitant, the reactant concentration and temperature, the reaction rate, and even the ambient humidity.17–20 Thus, it is difficult to produce YAG nanopowders with a uniform size distribution, less aggregation and a precise chemical composition. In contrast, individual spherical nanosize particles of yttrium(III) oxide (Y2O3) of uniform size distribution are easy to synthesize using a homogeneous precipitation process.21,22 In addition, monodisperse Al2O3 nanopowders of high quality are commercially available. For control of the morphology of the YAG precursor with good dispersion, a core–shell structure, with Al2O3 nanoparticles providing the core and the Y-compound the shell, seems to be a promising approach.
The usual approaches for producing a core–shell structure include chemical synthesis,23 chemical vapor deposition,24 laser-induced assembly,25 and electrostatic attraction.26 In this case, the synthesis of spherical YAG nanoparticles was examined by assembling a layer of a Y-compound on Al2O3 nanoparticles to form an Al2O3/Y-compound core–shell nanostructure, and then producing YAG nanoparticles using a micro-solid state reaction.27
The concept of our route involved the self-assembly of the Y-compound on the surface of Al2O3 nanoparticles based on a burst nucleation synthesis process, and the advantages of this route will be discussed. Based on our initial understanding of the basic concept of the partial wet chemical route, and inspired by an appreciation of its extensive applications, the mechanism of formation of the core–shell YAG precursor will be discussed. The fundamentals of the self-assembled core–shell precursors have been studied by dividing the reaction process into two stages, firstly the pre-coating of the Y-compound on the Al2O3 nanoparticle surface, driven by electrostatic attraction, and secondly the assemblage of the tiny Y-compound nanoparticles formed in the burst nucleation process onto the pre-coated Al2O3 nanoparticle surface, driven principally by its high surface energy. Based on a full understanding of the formation of the YAG core–shell nanostructure precursor, this partial wet chemical route may be applied to the synthesis of other core–shell nanostructure binary oxides.
Experimental
A typical experimental procedure for the synthesis of the YAG core–shell precursor by the partial wet route is as follows.27Yttrium nitrate (Y(NO3)3·6H2O, 99.99%, Alfa Aesar, Ward Hill, MA), and α-Al2O3 nanopowders (100−200 nm, 99.99%; Taimei Chemicals, Tokyo) were well dispersed in deionized water by ultrasonication, forming a stoichiometric (Y![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al = 3
Al = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) suspension. The concentration of Y3+ was maintained at 0.015 mol L−1 throughout. Urea (Specpure; Sinopharm Chemical Reagent Co Ltd, China) was applied as precipitant at a 20
5) suspension. The concentration of Y3+ was maintained at 0.015 mol L−1 throughout. Urea (Specpure; Sinopharm Chemical Reagent Co Ltd, China) was applied as precipitant at a 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of urea
1 ratio of urea![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Y3+. After homogenizing at room temperature, the mixed liquid was heated at 90 °C for 3 h with stirring. Surface modified Al2O3 with polyacrylic acid (PAA) or ammonium polyacrylate (PAA-NH4) as surfactants were used in place of pure Al2O3 nanoparticles to study the effect of surface modification of Al2O3 on the core–shell structure formation. The concentration of surfactants was 100 ppm throughout the reaction system.
Y3+. After homogenizing at room temperature, the mixed liquid was heated at 90 °C for 3 h with stirring. Surface modified Al2O3 with polyacrylic acid (PAA) or ammonium polyacrylate (PAA-NH4) as surfactants were used in place of pure Al2O3 nanoparticles to study the effect of surface modification of Al2O3 on the core–shell structure formation. The concentration of surfactants was 100 ppm throughout the reaction system.
An experiment involving individual Al2O3 particles was also performed in the absence of the Y3+/urea dispersion. To maintain a slow reaction rate during precipitation, a NH4HCO3 solution (0.2 M, Specpure; Sinopharm) was used as precipitant by adding it to the above suspension in droplet form. The precursors obtained in this manner were collected by filtration, washed with deionized water to remove by-products and ethanol to inhibit hard agglomeration. After drying in an oven at 80 °C for 24 h the powders were ready for characterization. Some samples were reserved during the reaction process to determine the intermediate state of the core–shell structure formation. For ceramic sintering, the YAG-precursor powders were sieved and then calcined at 1250 °C for 2 h. The calcined powders were uniaxially pressed at 50 MPa into pellets, followed by cool isostatic pressure at 200 MPa, and then sintered at 1780 °C for 4 h in a 4 × 10−3 Pa vacuum in a tungsten element furnace (EK712-T; Southwest Thermal Technology, Santa Rosa, CA).
The pH of the suspension was monitored using a pH meter (PHS-3D, Leica, China) with an accuracy of 0.02 units. A 0.1 mg mL−1 suspension of powder in deionized water was prepared for the measurement of zeta potential (ZetaPALS, Brookhaven Instruments Corporation, USA). Fourier transform infrared (FTIR) analysis of the YAG precursor was conducted using an infrared (IR) spectrometer (NEXUS 670; Thermo Nicolet Company) over the range 4000–400 cm−1. The core–shell structure of the precursor materials was investigated using a high-resolution transmission electron microscope (HRTEM, JEM-2100F, Jeol, Japan), and the X-ray powder diffraction (XRD) patterns of the powders were recorded on a Bruker D8 Advance powder X-ray diffractometer with Cu Kα (λ = 0.15406 nm). The morphology of the powder and the synthesized samples were characterized using field emission scanning electron microscopy (S-4800, Hitachi, Japan).
The distribution of the elements in the powder was studied using energy-dispersive X-ray spectrometer (EDS) attached to the HRTEM. The residual Y3+ in the filtration solution was checked using an inductively coupled plasma emission spectrometer (ICP-AES, Iris Intrepid II XSP, Thermo Electron Corporation, USA) to confirm the stoichiometry of the YAG precursor.
Results and discussion
Fig. 1 shows the scanning electron microscopy (SEM) and transmission electron microscopy (TEM) images of the α-Al2O3, Al2O3/Y-compound precursor, the calcined YAG particles synthesized by the partial wet chemical process using urea as precipitant, and also the EDS mapping results of the precursor.Fig. 1(a) shows that the Al2O3 nanoparticles had a smooth surface and homogeneous size distribution around 130 nm. Following the precipitation process the YAG precursor (Fig. 1(b)) had a rough surface and a size of 250 nm. Using ICP-AES, the residual Y ions in the filtration solution were found to be very small, indicating complete precipitation of the Y ions during the reaction (ESI, Table S1†). After calcination at 1250 °C for 2 h, pure YAG nanoparticles were obtained and were identified using XRD (ESI Fig. S1†). The powder (Fig. 1(c)) exhibited a similar morphology to that of the YAG precursor, with a size of 170 nm, but with a highly regular surface, superior even to that of pure Al2O3. The TEM image of pure Al2O3 confirmed the good dispersibility of the Al2O3 nanoparticles, and the inset shows a lattice image of pure α-Al2O3, confirming its high degree of crystallinity (Fig. 1(d)). This implied that the Al2O3 nanoparticles were suitable for use as templates.
The core–shell structure of the YAG precursor may be deduced from the TEM image and the EDS mapping results (Fig. 1(e) and (f)). The precursor particles obtained exhibited a clear interface between core and shell, which corresponded to the different compositions of their structure. In addition, the lattice structure of the Y-compound precursor shell (inset in Fig. 1(e)) shows its polycrystalline character. It is notable that the shell structure was easily affected by electron exposure (ESI, Fig. S2†). In comparison with the morphology of pure α-Al2O3, the shell composition was different from the composition of the center. This indicated that it should be possible to form the Al2O3/Y-compound core–shell structure with an Al2O3 core and a Y-compound shell.
The EDS mapping results give direct evidence of the core–shell structure formed by Al2O3 and the Y-compound. It was clear that the Al component occupied the core, with the Y component surrounding it. Blocked by the Y-compound shell, the XRD intensity of the Al2O3, consistently decreased with increase in the Y-compound shell (ESI, Fig. S1†). It is notable that the core–shell structure in the Y-compound shell restricted the neck sintering between neighboring YAG particles at a given temperature because of the high melting points of Y-rich compounds, leading to good dispersibility.27 Highly transparent ceramics (ESI, Fig. S3†) were obtained by sintering at 1780 °C in a vacuum of 4 × 10−3 Pa, demonstrating the high quality of the YAG powders and confirming that the YAG-precursor powders with good sintering properties could be synthesized using a partial wet chemical route.
The process of assembling Y-compound nanoparticles on the surface of Al2O3 nanoparticles is important to help understanding the mechanism of formation of Al2O3/Y-compound core–shell nanostructures. As reported previously, Y-compound nuclei are formed abruptly in a Y(NO3)3-urea solution at temperatures above 83 °C, known as a burst nucleation process.28 Normally, the Y-compound nuclei can self-assemble to form uniform spherical nanoparticles.
To study the mechanism of formation of the Al2O3/Y-compound core–shell structure in the Al2O–Y(NO3)3–urea system, the evolution of chemical bands under a variety of conditions was investigated using FTIR. As shown in Fig. 2, the FTIR spectra of the α-Al2O3 (curve (a)), the mid-product obtained from the suspension at a reaction temperature of 80 °C (pH 2.6, curve (b)) and the precursor particle obtained after holding at 90 °C for 3 h (pH 6.8, curve (c)) were different. Absorption bands in the range 800−400 cm−1, attributed mainly to metal–oxygen lattice vibration, were identified in the FTIR spectra of the pure Al2O3 (curve (a)). The main peaks at 450, 600, 640 and 735 cm−1 were assigned to characteristic Al–O vibrations.29 The band at 1640 cm−1 corresponded to water adsorption. After the system had been heated to 80 °C, the FTIR spectra of the Al2O3 mid-product collected (curve b) showed no obvious differences to that of pure Al2O3. However, the enlarged spectra in the region 800−1300 cm−1 showed some peaks around 1086 cm−1, becoming stronger than those of the pure Al2O3 (inset of Fig. 2). These peaks could be assigned to C–O bond stretching vibration.30 This implied that the Al2O3 surface had been modified by some kind of carbonate precipitant or precipitate, and was consistent with a separate study of Al2O3 modified with NH4HCO3 (ESI, Fig. S4†).
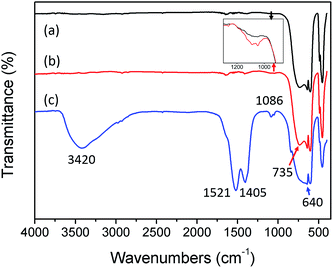 | ||
| Fig. 2 FTIR spectra of the pure Al2O3 (curve (a)), mid-product (curve (b)), YAG precursor (curve (c)), in the Al2O3–Y(NO3)3–urea system. | ||
As shown by the FTIR spectra of the YAG precursor obtained after reaction at 90 °C for 3 h (curve (c)), some peaks became much stronger and other new peaks appeared. The broad peak centered at 3420 cm−1 corresponded to the stretching mode of the hydroxyl groups (–OH).31 The peaks at 850 cm−1 and at ∼1085, ∼1405 and ∼1530 cm−1 originated from C–O bond bending and stretching vibration, respectively.32 On examination of the bands within the range 400–800 cm−1, they were mainly attributed to metal–oxygen lattice vibration, it was notable that the peak between 735 and 640 cm−1 had significantly increased. Assuming the peak at ∼730 cm−1 to be because of the characteristic Al–O vibration, and the well-known fact that heavy atom substitution reduces the metal–oxygen vibration frequency, observed as a red-shift in IR absorption, the peak between 735 and 640 cm−1 could be assigned to Y–O vibration. These peaks therefore corresponded to the formation of the Y-compound.
Based on the above results, the formation of the core–shell structure illustrated in Fig. 3 was confirmed. As urea hydrolysis began, the precipitant groups OH−, CO32− and HCO3− were generated and modified the surface of the template particle (α-Al2O3 core). These groups could be attributed to the slow decomposition of urea below 83 °C. A layer of these groups would attract Y ions on to the surface of the α-Al2O3 template in the reaction system.
As reported previously, Y(OH)CO3·xH2O is formed by hydrolysis of the urea in the suspension.28,33 However, restricted by the competition of crystal nucleation and growth related to the concentration of Y(OH)CO3·xH2O, the Y ions on the Al2O3 surface can readily form the precipitate. This represents the precoating of the Al2O3 nanoparticles with the Y-compound. Once the urea decomposes rapidly above 83 °C, the concentration of the negative ion groups (HCO3−, OH−, etc.) will increase sharply and these will react quickly with Y3+ to form Y-compound nuclei – the burst nucleation process – and the Y-compound nuclei then assemble on the surface of Al2O3 nanoparticles and form a shell. This means that the core–shell structure formation process consists of two stages, precoating of Y-compound on the surface of Al2O3 nanoparticles, and then formation of the shell structure following the burst nucleation process. To confirm and clarify the individual stages, specific experiments were designed accordingly.
An Al2O3 surface-modified with PAA or PAA-NH4 was prepared to check the effect of organic surfactant on the formation of the Al2O3/Y-compound core–shell structures. As shown in Fig. 4, the SEM images showed that the precursors obtained with either PAA (Fig. 4(a)) or PAA-NH4 (Fig. 4(b)) comprised large particles within the range 100–200 nm and smaller particles of 10–50 nm. Because the Al2O3 nanoparticles used as templates were larger in size than 100 nm, particles smaller than 50 nm could be identified as the newly formed Y-compounds. This indicated that the morphology of the YAG precursor obtained with PAA or with PAA-NH4 had not led to the formation of the core–shell structure.
 | ||
| Fig. 4 SEM images of the YAG precursors obtained in the Al2O3–Y(NO3)3–urea system with (a) PAA and (b) PAA-NH4, respectively, as surfactants. | ||
Considering the success of core–shell structure formation without PAA or PAA-NH4, the surface charge modification and the self-assembly process should be different with those of the addition of organic surfactant. The evolution of the surface charge properties of samples could provide information to help understanding the formation process. The surface charge properties of the α-Al2O3 particle in an Al2O3–Y(NO3)3–urea suspension, the Al2O3/Y-compound particle obtained at pH 2.6, the Al2O3/Y-compound particle obtained at pH 6.8, the individual Y-compound itself, as well as the α-Al2O3 particle with PAA and with PAA-NH4, were each analyzed in terms of zeta potential (Table 1). The zeta potential of the original α-Al2O3 powder was 46.1 ± 1.9 mV. When Al2O3 nanoparticles were dispersed in a urea solution and the solution heated below 83 °C, the zeta potential dropped to about 33 mV, which was caused by the adsorption of the negative ions (HCO3−, OH−, etc.), as demonstrated in the previous section. The zeta potential of Y-compound particles, measured by the homogeneous precipitation method, was about −28.5 mV, indicating that the Al2O3 and Y-compound nanoparticles possessed opposite charges. This was able to supply the initial driving force for assembling the core–shell structure. With increase in shell thickness, the effect of the electrostatic attraction became weaker and could no longer support the assembly process. The high surface energy of the tiny Y-compounds which originated from the burst nucleation process would then provide the entire driving force for the formation of the core–shell structure.
The low zeta potential value of the YAG precursor (−5.7 mV) was assumed to be because of the compensation of the opposite charges from the two different nanoparticles. However, after surface modification with PAA and PAA-NH4, Al2O3 nanoparticles showed high negative zeta potential values of −44.5 ± 0.6 mV and −46.8 ± 0.7 mV, respectively. Electrostatic repulsion between these Al2O3 nanoparticles and Y-compounds in the suspension accounted for the failure of the core–shell structure formation. As illustrated in Fig. 5, the main effect of the surface modification with PAA or PAA-NH4 was prevention of the assembly process.
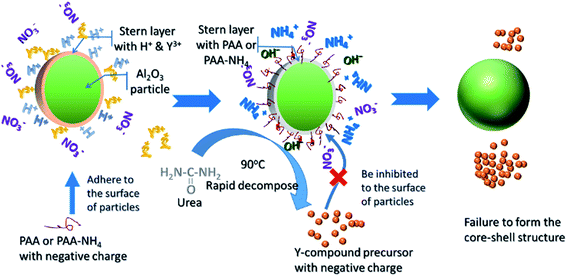 | ||
| Fig. 5 Sketch of the reaction process in the Al2O3–Y(NO3)3–urea system, with either PAA or PAA-NH4 as surfactant. | ||
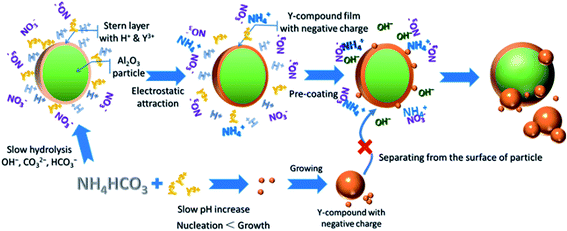
To clarify the proposed mechanism of the core–shell structure formation, an experiment was designed using NH4HCO3 as precipitant, in order to simulate a similar reaction process but with a slower reaction rate. Dropwise addition of NH4HCO3 to the Al2O3–Y(NO3)3 suspension was performed to achieve a slow evolution of pH (ESI, Fig. S5†), and the morphology of the samples obtained is shown in Fig. 6. The Al2O3–Y(NO3)3 suspension prepared had a pH of about 1.5, and on adding NH4HCO3 solution, the pH approached 3.5. The morphology of the mid-product particles obtained (pH 3.5) was almost identical to that of pure Al2O3 particles (Fig. 6(a)). However, on exploration by electron beam, a large quantity of tiny particles were seen to be located on the particle surface (Fig. 6(b)). This indicated that a thin layer of a second phase had formed homogeneously on the surface of the Al2O3 nanoparticles, similar to that obtained with urea as precipitant. After further addition of NH4HCO3 solution (pH 6.8) the thickness of the second phase layer on the surface of Al2O3 nanoparticle increased (Fig. 6(c)). At the same time, some independent nanoparticles about 10–50 nm in diameter were noticed in the precursor. Based on the earlier discussion, these nanoparticles were identified as self-assembled Y-compound.
Compared with the Al2O3–Y(NO3)3–urea system, the only difference in the reaction process of the Al2O3–Y(NO3)3–NH4HCO3 system was its slow reaction rate. As is well known, the supersaturation, S, plays a key role in chemical precipitation, and is in general given by:
| S = aAaB/Ksp | (1) |
The low solubility product constants of Y2(CO3)3 (1.03 × 10−31) and Y(OH)3 (1.00 × 10−22) made it easy to reach supersaturation. When urea was used as the precipitant (Fig. 3), once the reaction temperature reached 83 °C, a large proportion of the urea decomposed, supplying precipitating ligands and reaching high supersaturation. A considerable quantity of very small Y-compound particles were then formed, in a process which is described as a burst nucleation. In competition with this nucleation process the nuclei were unable to grow, but assembled to lower the total surface energy. On the other hand, using NH4HCO3 as precipitant (Fig. 7), the slow reaction process gave a relatively low degree of supersaturation, leading to a small number of Y-compound nuclei in the reaction system. As the reaction proceeded these nuclei were able to grow, and the Y-compound nanoparticles provided cores for further assembly of Y-compound nanoparticles, comparable to the assembly on the precoated Al2O3 nanoparticles. In addition to the increase in thickness of the Y-compound coating layer, individual Y-compound nanoparticles were formed in the system. Thus, in the Al2O3–Y(NO3)3–NH4HCO3 system, it could be inferred that the absence of the burst nucleation process corresponded to the increased formation of individual Y-compound nanoparticles.
In general, the results described previously revealed that the formation of the core–shell structure by a partial wet process consisted of two stages. Firstly, the slow formation of the Y-compound took place on the surface of the Al2O3 particles, initiated by the adsorption of Y ions and weak hydrolysis of the urea. Secondly, a large number of very small primary precipitated particles were formed in the suspension as a result of the burst nucleation process. Driven by the high surface energy of the nanoparticles, the small particles assembled and formed a shell structure on the precoated Al2O3 nanoparticles, lowering the total surface energy. Compared to the more familiar mechanism of core–shell structure formation,35–37 the driving force in the burst nucleation process was not only the traditional electrostatic attraction between the template–core and the shell materials, but also the result of the high surface energy of the nanoparticles.
Conclusions
The mechanism of the core–shell structured YAG precursor formation was intensively studied. A two-stage mechanism is proposed to describe the formation process. Initially, the surface of Al2O3 particles adsorbs carbonate and hydroxyl groups and forms very small Y-compound nuclei on the surface of the Al2O3 nanoparticles. The burst nucleation process then occurs at the urea decomposition temperature, and large numbers of small Y-compound nuclei are formed simultaneously throughout the suspension. By self-assembly of the Y-compound nanoparticles on the surface of the precoated Al2O3 nanoparticles, Al2O3/Y-compound core–shell nanostructures are formed. During most of the process, the driving force is the electrostatic attraction between the opposite surface charges of Al2O3 and Y-compound nanoparticles. However, the high surface energy of the Y-compound nanoparticles originating from the burst nucleation process plays a key role in completing the core–shell structure formation. Surface modification with negatively charged PAA− blocks both the precoating process and the assemblage processes, leading to failure of the core–shell structure formation. Following clarification of the mechanism of core–shell structure formation, the partial wet chemical route involving a burst nucleation process may be applied to the morphology-controllable synthesis of other binary oxides.Acknowledgements
This research was supported by an NSFC grant (NSFDYS: 50990303, 51372142, 51102040, 51402172), the Innovation Research Group (IRG: 51321091), and the China Postdoctoral Science Foundation (2013M541909).References
- E. A. Ahmad, G. Mallia, D. Kramer, A. R. Kucernak and N. M. Harrison, J. Mater. Chem. A, 2013, 1, 11152 CAS.
- J. Griesbauer, T. Korner, T. Wehlus, A. Heinrich, B. Stritzker, J. Simon and W. Mader, CrystEngComm, 2011, 13, 77 RSC.
- J. G. Li, T. Ikegami, J. H. Lee, T. Mori and Y. Yajima, J. Eur. Ceram. Soc., 2001, 21, 139 CrossRef CAS.
- J. M. Patete, X. H. Peng, C. Koenigsmann, Y. Xu, B. Karn and S. S. Wong, Green Chem., 2011, 13, 482 RSC.
- Y. D. Liu, J. Goebl and Y. D. Yin, Chem. Soc. Rev., 2013, 42, 2610 RSC.
- M. Veith, S. Mathur, A. Kareiva, M. Jilavi, M. Zimmer and V. Huch, J. Mater. Chem., 1999, 9, 3069 RSC.
- S. A. H. Tabrizi, E. T. Nassaj and H. Sarpoolaky, J. Alloys Compd., 2008, 456, 282 CrossRef PubMed.
- X. J. Xu, X. D. Sun, H. Liu, J. G. Li, X. D. Li, D. Huo and S. H. Liu, J. Am. Ceram. Soc., 2012, 95, 3821 CrossRef CAS PubMed.
- D. J. Sordelet, M. Akinc, M. L. Panchula, Y. Han and M. H. Han, J. Eur. Ceram. Soc., 1994, 14, 123 CrossRef CAS.
- Y. J. Li, Y. C. Zhang and J. X. Leng, Key Eng. Mater., 2014, 602–603, 110 CrossRef CAS.
- H. Z. Wang, L. Gao and K. Niihara, Mater. Sci. Eng., A, 2000, 288, 1 CrossRef.
- M. L. Saladino, G. Nasillo, D. C. Martino and E. Caponetti, J. Alloys Compd., 2010, 491, 737 CrossRef CAS PubMed.
- X. L. Zhang, D. Liu, Y. H. Sang, H. Liu and J. Y. Wang, J. Alloys Compd., 2010, 502, 206 CrossRef CAS PubMed.
- A. Aboulaich, J. Deschamps, R. Deloncle, A. Potdevin, B. Devouard, G. Chadeyron and R. Mahiou, New J. Chem., 2012, 36, 2493 RSC.
- X. Li, H. Liu, J. Y. Wang, F. Han and R. I. Boughton, J. Am. Ceram. Soc., 2004, 87, 2288 CrossRef CAS PubMed.
- S. Bogatko and P. Geerlings, Phys. Chem. Chem. Phys., 2012, 14, 8058 RSC.
- Y. H. Sang, H. Liu, X. D. Sun, X. L. Zhang, H. M. Qin, Y. H. Lv, D. Huo, D. Liu, J. Y. Wang and R. I. Boughton, J. Alloys Compd., 2011, 509, 2407 CrossRef CAS PubMed.
- A. Sahraneshin, S. Takami, D. Hojo, T. Arita, K. Minami and T. Adschiri, CrystEngComm, 2012, 14, 6085 RSC.
- Y. H. Sang, Y. H. Lv, H. M. Qin, X. L. Zhang, H. Liu, J. Y. Wang, X. D. Sun and R. I. Boughton, Ceram. Int., 2012, 38, 1635 CrossRef CAS PubMed.
- P. Palmero, C. Esnouf, L. Montanaro and G. Fantozzi, J. Eur. Ceram. Soc., 2005, 25, 1565 CrossRef CAS PubMed.
- H. M. Qin, H. Liu, Y. H. Sang, Y. H. Lv, X. L. Zhang, Y. Y. Zhang, T. Ohachi and J. Y. Wang, CrystEngComm, 2012, 14, 1783 RSC.
- H. M. Qin, X. H. Zhang, H. Liu, Y. H. Sang and J. Y. Wang, CrystEngComm, 2013, 15, 5076 RSC.
- M. Yao, W. M. Chen, X. Fan, C. M. Liu, X. M. Meng, L. Guo and C. P. Chen, CrystEngComm, 2011, 13, 2593 RSC.
- J. X. Yu, N. Du, H. Zhang and D. R. Yang, RSC Adv., 2013, 3, 7713 RSC.
- G. Sahu, K. Wang, S. W. Gordon, W. L. Zhou and M. A. Tarr, RSC Adv., 2012, 2, 3791 RSC.
- M. K. Han, X. W. Yin, L. Kong, M. Li, W. Y. Duan, L. T. Zhang and L. F. Cheng, J. Mater. Chem. A, 2014, 2, 16403 CAS.
- Y. H. Sang, H. M. Qin, H. Liu, L. L. Zhao, Y. N. Wang, H. D. Jiang and J. Y. Wang, J. Eur. Ceram. Soc., 2013, 33, 2617 CrossRef CAS PubMed.
- J. G. Li, X. D. Li, X. D. Sun and I. Takamasa, J. Phys. Chem. C, 2008, 112, 11707 CAS.
- X. S. Li, V. K. Michaelis, T. C. Ong, S. J. Smith, I. Mckay, P. Müller, R. G. Griffin and E. N. Wang, CrystEngComm, 2014, 16, 2950 RSC.
- J. Q. Jiao, X. Liu, W. Gao, C. W. Wang, H. J. Feng, X. L. Zhao and L. P. Chen, CrystEngComm, 2009, 11, 1886 RSC.
- N. Venkatachalam, Y. Saito and K. Soga, J. Am. Ceram. Soc., 2009, 92, 1006 CrossRef CAS PubMed.
- S. Som and S. K. Sharma, J. Phys. D: Appl. Phys., 2012, 45, 415102 CrossRef.
- V. K. Ivanov, A. E. Baranchikov, A. S. Vanetsev, A. S. Shaporev, O. S. Polezhaeva, Y. D. Tretyakov, P. P. Fedorov and V. V. Osiko, Russ. J. Inorg. Chem., 2007, 52, 1321 CrossRef.
- J. G. Li, X. Li, X. D. Sun, T. Ikegami and T. Ishigaki, Chem. Mater., 2008, 20, 2274–2281 CrossRef CAS.
- J. Geng, X. D. Jia and J. J. Zhu, CrystEngComm, 2011, 13, 193 RSC.
- D. A. Walker, B. Kowalczyk, M. O. de la Cruz and B. A. Grzybowski, Nanoscale, 2011, 3, 1316 RSC.
- J. F. L. Duval, J. Merlin and P. A. L. Narayana, Phys. Chem. Chem. Phys., 2011, 13, 1037 RSC.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra09856k |
| ‡ These authors contributed equally to the study. |
| This journal is © The Royal Society of Chemistry 2014 |