Ultrasensitive determination of bisphenol A in water by inhibition of copper nanoclusters-enhanced chemiluminescence from the luminol–KMnO4 system
Shuangjiao Xua,
Mao Denga,
Yanyan Suia,
Yingxue Zhangb and
Funan Chen*a
aThe Key Laboratory of Luminescence and Real-time Analytical Chemistry, Ministry of Education, School of Chemistry and Chemical Engineering, Southwest University, Chongqing, 400715, China. E-mail: chenfn@swu.edu.cn; Fax: +86-23-68258363; Tel: +86-18883255598
bCollege of Chemistry, Chongqing Normal University, Chongqing, China
First published on 11th September 2014
Abstract
In the present work, a highly sensitive chemiluminescence (CL) system for determination of trace bisphenol A (BPA) was established. The method was based on the inhibitory effect of luminol CL by BPA on the Cu NCs-enhanced luminol–KMnO4 CL system for the first time. Water-soluble Cu NCs capped with BSA were synthesized. Under the optimized conditions, the inhibited CL intensity was proportional to the concentration of BPA ranging from 1.0 × 10−9 to 1.0 × 10−5 mol L−1, with a detection limit (3σ) of 1.2 × 10−10 mol L−1. It indicated that the concentration level is lower by almost 1–3 orders of magnitude compared with alternatives. The relative standard deviation (RSD) of the method was 1.7% with 9 repeated measurements of 1.0 × 10−6 mol L−1 BPA. The proposed method has also been applied to determine BPA in water samples with good accuracy and precision. The CL spectra, UV-visible spectroscopy and radical scavengers were conducted to explore the possible mechanism.
Introduction
Bisphenol A (2,2-bis(4-hydroxyphenyl)propane, BPA), as shown in Fig. 1A, has been widely used as an intermediate in the manufacture of polycarbonate (PC), epoxy resins and other plastics.1,2 Since the first discovery of the release of BPA from polycarbonate bottles at high temperature and pressures, more attention has been paid to research on BPA toxicity.3,4 It was found that BPA mainly involved its potential human health risk at low doses due to its estrogenic activities. Moreover, BPA is possibly associated with cancer, pleiotropic actions in the brain and cardiovascular system.5–7 Therefore, it is of great importance to develop a simple, ultrasensitive, selective method for determination of trace amounts of BPA. In particular, for drinking water, BPA released from the plastic packages or cups should be accurately monitored on-site for food safety. The analytical methods have been widely available for detection of BPA, including fluorescence spectrometry,8–11 liquid chromatography (LC),12,13 gas chromatography coupled with mass spectrometry (GC-MS),14–16 immunochemical assay,17–19 and electrochemical sensor.20–22 However, these techniques for determination of BPA suffer from low sensitivity, narrow linear range along with the use of toxic organic solvents and expensive instruments.In recent years, CL analysis method has been applied in different fields,23–29 because of its high sensitivity, wide linear range, simple instrumentation, and no background scattering light interference. Wang et al.30 had demonstrated the feasibility of CL detection for BPA, based on the inhibitory effect of BPA on the CL reaction between luminol and potassium hexacyanoferrate. Lin et al.31 had investigated that BPA could inhibit the CL from gold(III) and peroxymonocarbonate and then determined it in water. Nevertheless, the detection limit and range wide obtained with these methods are not enough for the trace analysis of BPA in real samples. Therefore, it is a valuable analytical task to develop an alternative CL method for determination BPA.
Lately, chemiluminescence study has been extended to nanomaterials system, providing amplified CL emission. It opens up a new way to enhance the inherent sensitivity and expand novel applications in detection. As an example, noble nanomaterials of gold, silver, platinum nanoparticles have always been used as CL amplifiers to enhance many traditional CL systems,32–34 but their applications are restricted owing to the high cost. Recently, our group has found that copper nanoclusters (NCs) can also greatly enhance the CL of luminol–H2O2 system.35 Compared with the noble metals Au and Ag, the metal Cu is relatively abundant, inexpensive, and readily available from commercial sources.
Metal nanoclusters consisting of several to tens of atoms have newly stood out.36–39 Their unique physical, electrical, and optical properties have made metal NCs as promising candidates in the fields of catalysis, chemical sensors, electronics devices, and biological imaging.39,40 Until now, the applications of metal NCs in analytical fields are mainly concentrated on their fluorescence properties and very little on their catalytic properties for biological or chemical sensing application.35,41,42 Therefore, it is significant to explore new sensing platforms based on their catalytic activities of metal NCs. As far as we know, there are not any reports about Cu NCs-enhanced CL assay for BPA.
In this paper, we developed a highly sensitive method for detection of BPA based on the inhibitory effect of luminol–KMnO4–Cu NCs by BPA. The incorporation of Cu NCs into the luminol–KMnO4 system induces catalytic effect on the CL assay, thus allowing for quantification of BPA with a linear range of 1.0 × 10−9 to 1.0 × 10−5 mol L−1 and a detection limit of 1.2 × 10−10 mol L−1 (3σ). The proposed method has been used to detect BPA in the water samples with satisfactory results. Moreover, the possible mechanism was carefully studied in term of UV-visible, CL spectra and radical scavengers in this work.
Experimental
Reagents and materials
All chemicals and reagents were of analytical grade and used as received without further purification, and ultrapure water was used throughout. BPA was purchased from Tokyo chemical industry Co., Ltd. (Tokyo, Japan). The stock solution of BPA (1.0 × 10−2 mol L−1) was prepared with ethanol and then diluted to the desired concentrations with water. A 1.0 × 10−2 mol L−1 stock solution of luminol (3-aminophthalhydrazide) was prepared by dissolving luminol (Sigma) in 0.1 mol L−1 sodium hydroxide solution. Working solutions of luminol were prepared by diluting the stock solution. Bovine serum albumin (BSA) was purchased from Sangon Biotech Co., Ltd. (Shanghai, China). CuSO4·5H2O was purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Potassium permanganate and ascorbic acid (AA) were commercially purchased from Chongqing Chemical Regent Company (Chongqing, China).Synthesis of BSA–Cu nanoclusters
BSA modified Cu NCs were synthesized in aqueous solution following the previously reported method.43 In a typical experiment, 1 mL aqueous CuSO4·5H2O solution (20 mM) was added to BSA solution (5 mL, 15 mg mL−1) under vigorous stirring for 5 min at room temperature. Then, the solution PH was adjusted to 12 by adding NaOH solution and the mixture was allowed to proceed under vigorous stirring at 55 °C for 8 h. The solution was then dialyzed in ultra-pure water for 48 h to remove unreacted Cu2+. The final solution was stored at 4 °C in refrigerator when not in use.General procedure for CL analysis
The chemiluminescence detection was carried out on a laboratory-built flow injection CL system (Xi'an Remax Company, Xi'an, China), consisting of two peristaltic pumps to deliver the reactants to the flow cell (Fig. 1B). One delivered KMnO4 and Cu NCs (or with samples) with two channels at a flow rate (per tube) of 1.9 mL min−1. The other pump was used to carry luminol solution at the same flow rate. The PTFE tubing (0.8 mm i.d.) was used to connect all components in the flow system. A six-way injection valve equipped with an 8 cm long sampling loop was used to inject. The CL signal produced was detected by a photomultiplier tube (operated at −500 kV), and was then recorded by a computer equipped with a data acquisition interface. Data acquisition and treatment were performed with BPCL software running under Windows XP. Determination of BPA was performed based on the net CL intensity of ΔI = I0 − Is, where I0 and Is denote the CL intensity in the absence and presence of BPA, respectively.Sample preparation
Tap water and bottled drinking water was analyzed without any pretreatment. River water samples were filtered with a 0.45 μm filter to remove particulate matter. To a 50 mL flask, 5 mL water samples and 0.75 mL of 1.0 × 10−4 mol L−1 EDTA were added and diluted to the desired concentration with ultrapure water. The above solutions were then used for BPA detection.Results and discussion
Inhibition of BPA on the Cu NCs-enhanced CL assay
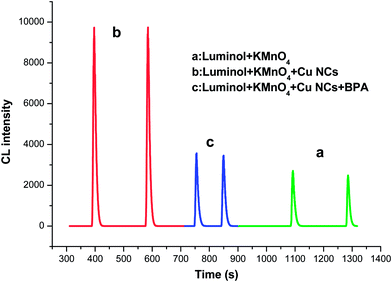
Luminol–KMnO4 CL reaction, a popular CL reaction, has been extensively applied for the detection of various substances. As show in Fig. 2, the oxidation of luminol by KMnO4 generates weak CL in alkaline media. However, the CL signal intensity could be enhanced by about 3 times as soon as adding the Cu nanocluster. Then, the CL intensity will decrease remarkably after incorporation of BPA into the Cu NCs-enhanced CL system, thus permitting to sensitively detect trace BPA.Optimization of the reaction conditions
The reaction conditions were optimized for the CL system shown in Fig. 3. It is in the basic solution that luminol reacts with KMnO4 to produce light emission. Therefore, the concentration of NaOH in luminol solution was investigated in the range of 0.01–0.5 mol L−1 (Fig. 3A). With the increase of concentration from 0.01 to 0.3 mol L−1, the CL intensity increased sharply and then changes slowly beyond 0.3 mol L−1. Therefore, 0.3 mol L−1 was selected as optimum in the present system.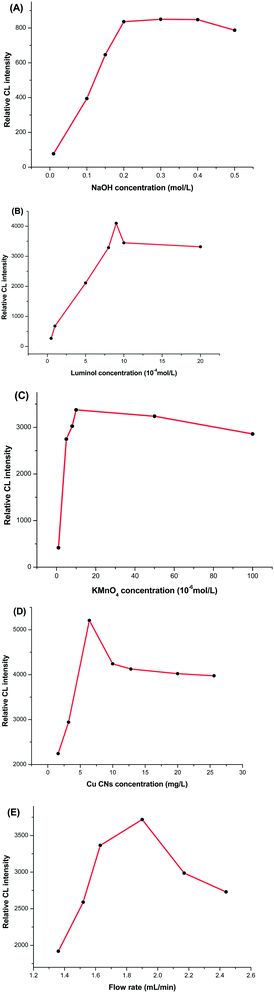 | ||
| Fig. 3 Effects of the reaction conditions on the luminol–KMnO4–Cu NCs–BPA CL system. (A) NaOH (B) luminol (C) KMnO4 (D) Cu NCs (E) flow rate. | ||
Luminol is a key factor on the signals as a common CL reaction reagent. The effect of luminol concentration on the CL was studied in the range from 5.0 × 10−5 to 2.0 × 10−3 mol L−1 (Fig. 3B), the CL intensity increased with increasing luminol concentration in the range of 5.0 × 10−5 to 9.0 × 10−4 mol L−1. However, when the concentration of luminol was above 9.0 × 10−4 mol L−1, only slight changes in the CL intensity were observed. Therefore, 9.0 × 10−4 mol L−1 was chosen as the optimal luminol concentration in the present study.
As an oxidant, KMnO4 concentration influences the CL intensity. The effect of KMnO4 concentration on the CL was tested in the range of 1.0 × 10−6 to 1.0 × 10−4 mol L−1. As can be seen from Fig. 3C, the optimal KMnO4 concentration was set at 1.0 × 10−5 mol L−1 due to maximum ΔI. The effects of the concentration of Cu NCs and the flow rate were also discussed (Fig. 3D and E). In view of the CL intensity and the consumption of the regents, the optimized conditions for CL system were as follows: 9.0 × 10−4 mol L−1 luminol in NaOH solution (0.3 mol L−1), 1.0 × 10−5 mol L−1 KMnO4, 12.8 mg L−1 Cu NCs and 1.90 mL min−1 flow rate.
Mechanism discussion
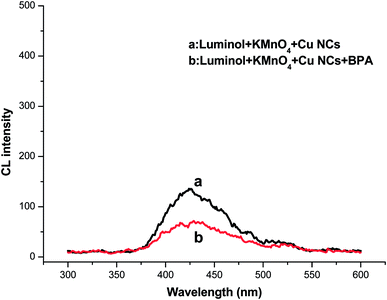
The CL-generation mechanism for luminol oxidation in aqueous solution had summarized by Merényi et al.44 to occur in three basic steps: (1) oxidation of luminol to the luminol radical; (2) oxidation of the luminol radical to hydroxy hydroperoxide, the key intermediate; (3) decomposition of hydroxyl hydroperoxide with or without the emission of CL, among which step 1 was supposed to be the rate-determining step of luminol CL.45 The presence of oxygen-related radicals (for example, OH˙, O2˙−, and other radical derivatives) as oxidants was expected to occur, during the luminol oxidation processes. As for luminol–KMnO4 system, the CL reaction of superoxide radical with luminol in alkaline solution was catalyzed in presence of KMnO4.A F-7500 mode fluorescence spectrophotometer has been used to discuss the CL mechanism of luminol–KMnO4–Cu NCs in the presence or absence of BPA. The CL spectra were obtained after turning off light entrance slot. As shown in Fig. 4, the maximal emission peak located at 425 nm clearly in both cases, indicating that the luminophor was still the excited-sate 3-aminophthalate anions, the oxidation product of luminol. Therefore, the adding of Cu NCs did not generate a new luminophor for this CL system. The enhanced CL signals were thus attributed to the possible catalysis from Cu NCs.
In order to further confirm the possible mechanism, the UV-visible absorption spectra were recorded. As shown in Fig. 5, the maximum absorption peaks of Cu NCs is observed at around 325 nm and luminol–KMnO4 system has three absorption peaks at around 300 nm, 315 nm and 350 nm, respectively. Therefore, the light absorption of the mixed system was approximately equivalent to the sum of the light absorption of the two individuals, which suggests that no interaction exists between the species after the reaction. As a result, the enhancement of CL signals had derived from the catalytic effects of Cu NCs. Similar results were also accomplished for luminol–K3Fe(CN)6–ZnSe QDs system.46
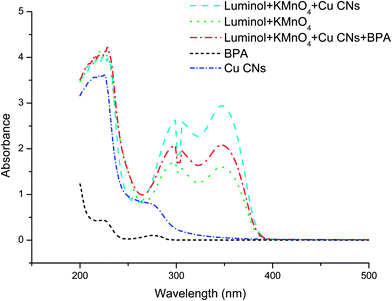 | ||
| Fig. 5 UV-vis absorption spectra. Luminol: 9.0 × 10−4 mol L−1 in NaOH solution (0.3 mol L−1), KMnO4: 1.0 × 10−5 mol L−1, Cu NCs: 12.8 mg L−1, BPA: 1.0 × 10−5 mol L−1. | ||
The reaction of luminol with KMnO4 in alkaline solution without a catalyst underwent weak CL. According to some literatures,32,35,46 we assumed that the catalyst Cu NCs may interact with the reactants or the intermediates of the reaction of luminol with KMnO4. When Cu NCs were used as the catalyst, the formation of active oxygen-containing reactant intermediates such as O2˙− and OH˙ were reported.35 It was identified by the experiments showing that the CL could be quenched even at a relatively low concentration by ascorbic acid, which is an efficient active oxygen scavenger. The generated OH˙ was stabilized on the surface of Cu NCs via partial electron exchange interactions. OH˙ radicals reacted with luminol anion generating luminol radicals. It was followed by the reaction between O2˙− and luminol radicals, and then generated luminol intermediate, which could enhance the CL of luminol.
Upon in corporation of BPA into the luminol–KMnO4–Cu NCs system, the absorption peak of BPA at 275 nm disappeared, which could be caused by oxidation of hydroxyl groups in BPA by KMnO4. The consumption of KMnO4 gives rise to decrease in the amount of 3-aminophthalate ions and then CL emission.32
Analytical performance
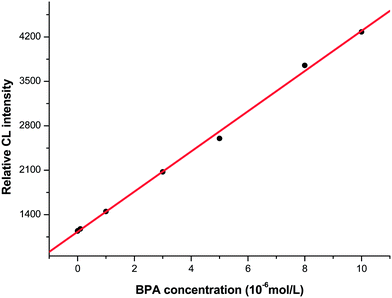
Under the optimum conditions described above, the linear calibration range prolonged over 4 orders of magnitude from 1.0 × 10−9 to 1.0 × 10−5 mol L−1 for BPA (Fig. 6). The regression equation is ΔI = 1130.3 + 316.4 [BPA] (mol L−1), r = 0.9990 (n = 8). The limit of detection (LOD, 3σ) for BPA was 0.12 nM. The relative standard deviation (RSD) was 1.7% for 10−6 mol L−1 BPA (n = 9). Moreover, as compared with the analytical methods previously reported in literatures in terms of LOD and linear range (Table 1), the proposed method shows high sensitivity for the determination of BPA.| Method | Sample | Linear range (μM) | Detection limit (μM) | Reference |
|---|---|---|---|---|
| CL1 | Plastic | 0.8–12 | 0.3 | 30 |
| CL2 | Polycarbonate | 0.05–50 | 0.01 | 26 |
| CL3 | Plastic | 0.3–80 | 0.08 | 31 |
| LC-MS | Plastic | 0.09–44 | 0.06 | 13 |
| LC-FL | Water | 0.04–4.4 | 0.06 | 10 |
| GC-MS | Water | 0.04–219 | 0.004 | 14 |
| ECL | Water | 0.1–11 | 0.083 | 20 |
| FL | Water | 0.1–1.8 | 0.009 | 8 |
| CL4 | Water | 0.001–10 | 0.00012 | This method |
Interference study
The selectivity of the proposed method was evaluated by analyzing a standard solution of 10−6 mol L−1 BPA, to which varying amounts of possible interference were added. With respect to 10−6 mol L−1 BPA, the tolerable limit of each exotic species was considered as a relative error less than the 5% level. The results were summarized in Table 2. Most of the ions and organics had no essential effect on the detection of 10−6 mol L−1 BPA. River water samples usually contain a mass of metal ions that may interfere with BPA determination. However, the interferences from these metal ions could be eliminated with the addition of EDTA. Therefore, the results indicated that the proposed CL system is highly selective for BPA.| Species | Tolerance ratioa | Species | Tolerance ratioa |
|---|---|---|---|
| a The ratio of concentration between the interfering substance and BPA, i.e. [ion]/[BPA]. | |||
| Na+ | 1000 | CO32− | 400 |
| K+ | 1000 | HPO42− | 300 |
| Ca2+ | 500 | Ethanol | 300 |
| Mg2+ | 500 | Glucose | 600 |
| Pb2+ | 300 | Citric acid | 100 |
| Zn2+ | 300 | Tartaric acid | 100 |
| Fe3+ | 100 | Malic acid | 100 |
| Cl− | 1000 | TBHQ | 50 |
| NO3− | 1000 | BHT | 50 |
| C2O42− | 500 | BHA | 50 |
| SO42− | 500 | m-Nitrophenol | 10 |
| HCO3− | 400 | Salicylic acid | 50 |
Analytical applications
In order to evaluate the applicability and reliability of the proposed method, it was applied to the determination of BPA in real samples, such as tap water, bottled drinking water, and river water. From Table 3, bottled drinking water exhibits higher BPA concentration level than tap water. Meanwhile, the longer time barreled drinking water aged, the higher BPA content was detected in water sample. For comparison, two samples of bottled drinking water were also determined by traditional CL method.30 The data show that the two methods are in excellent agreement. The recovery of BPA in water samples ranged from 97.4 to 102.5% through standard addition experiments, which demonstrated the proposed CL system was satisfactory for BPA analysis.| Samples | Added (μM) | Found (μM) | Recovery (%) |
|---|---|---|---|
| a 6 months.b 12 months.c Traditional CL method.30d Not detected. | |||
| Tap water | 0 | NDd | |
| 0.50 | 0.491 | 98.2 | |
| 1.00 | 0.990 | 99.0 | |
| Bottled drinking water 1a | 0 | 0.12 | |
| 2.00 | 2.10 | 99.1 | |
| 3.00 | 3.08 | 98.7 | |
| Bottled drinking water 2b | 0 | 0.28 | |
| 4.00 | 4.18 | 97.6 | |
| 4.15c | |||
| 5.00 | 5.30 | 100.4 | |
| 5.24c | |||
| River water | 0 | 5.15 | |
| 0.10 | 5.19 | 98.9 | |
| 1.50 | 6.86 | 102.5 | |
Conclusion
In summary, Cu NCs were found to enhance the CL signals between luminol and KMnO4 greatly, and then BPA displays a significant inhibition on the luminol–KMnO4–Cu NCs CL system probably due to the oxidation of hydroxyl group in BPA by KMnO4. The proposed method was highly sensitive for trace BPA with a linear range of 1.0 × 10−9 to 1.0 × 10−5 mol L−1.The detection limit of 1.2 × 10−10 mol L−1 is lower than other methods by almost 1–3 orders of magnitude. Moreover, the proposed method was successfully applied for detection BPA in water samples. In addition, based on the CL spectra, UV-visible spectroscopy and radical scavengers, a possible CL reaction mechanism was put forward. This work was of great significance for investigation of new and efficient catalysts for CL systems.Acknowledgements
This work was supported by Science and Technology Research Project of Chongqing Educational Committee (CSTC, 2010BB8328). We thank Prof. H. Z. Zheng and Prof. Y. M. Huang for measurements.References
- A. Ballesteros-Gomez, S. Rubio and D. Perez-Bendito, J. Chromatogr. A, 2009, 1216, 449–469 CrossRef CAS PubMed.
- M. Rezaee, Y. Yamini, S. Shariati, A. Esrafili and M. Shamsipur, J. Chromatogr. A, 2009, 1216, 1511–1514 CrossRef CAS PubMed.
- T. K. Jensen, J. Toppari and N. Keiding, Clin. Chem., 1995, 41, 1896–1901 CAS.
- L. Sharp, R. J. Black and E. F. Harkness, Scottish Cancer Intelligence Unit, 1993 Search PubMed.
- K. D. Lin, W. P. Liu and J. Gan, Environ. Sci. Technol., 2009, 43, 3860–3864 CrossRef CAS.
- H. S. Yin, L. Cui, S. Y. Ai, H. Fan and L. S. Zhu, Electrochim. Acta, 2010, 55, 603–610 CrossRef CAS PubMed.
- M. Portaccio, D. Di Tuoro, F. Arduini, M. Lepore, D. G. Mita, N. Diano, L. Mita and D. Moscone, Biosens. Bioelectron., 2010, 25, 2003–2008 CrossRef CAS PubMed.
- J. Fan, H. Q. Guo, G. G. Liu and P. G. Peng, Anal. Chim. Acta, 2007, 585, 134–138 CrossRef CAS PubMed.
- X. Wang, H. L. Zeng, Y. L. Wei and J. M. Lin, Sens. Actuators, B, 2006, 114, 565–572 CrossRef CAS PubMed.
- J. H. Kang, F. Kondo and Y. Katayama, Anal. Chim. Acta, 2006, 555, 114–117 CrossRef CAS PubMed.
- X. Y. Cao, F. Shen, M. W. Zhang, J. X. Bie, X. Liu, Y. l. Luo, J. J. Guo and C. Y. Sun, RSC Adv., 2014, 4, 16597–16606 RSC.
- E. Maiolini, E. Ferri, A. L. Pitasi, A. Montoy, M. Di Giovanni, E. Errani and S. Girotti, Analyst, 2014, 139, 318–324 RSC.
- S. N. Pedersen and C. Lindholst, J. Chromatogr. A, 1999, 864, 17–24 CrossRef CAS.
- P. Varelis and D. Balafas, J. Chromatogr. A, 2000, 883, 163–170 CrossRef CAS.
- K. K. Selvaraja, G. Shanmugama, S. Sampatha, D. G. Joakim Larssonb and B. R. Ramaswamy, Ecotoxicol. Environ. Saf., 2014, 99, 13–20 CrossRef PubMed.
- M. L. Ocaa, M. C. Ortiza, A. Herreroa and L. A. Sarabiab, Talanta, 2014, 106, 266–280 CrossRef PubMed.
- M. Aminur Rahman, M. J. A. Shiddiky, J. S. Park and Y. B. Shim, Biosens. Bioelectron., 2007, 207, 2464–2470 CrossRef PubMed.
- G. R. Marchesini, E. Meulenberg, W. Haasnoot and H. Irth, Anal. Chim. Acta, 2005, 528, 37–45 CrossRef CAS PubMed.
- Z. L. Mei, H. Q. Chu, W. Chen, F. Xue, J. Liu, H. N. Xu, R. Zhang and L. Zheng, Biosens. Bioelectron., 2013, 39, 26–30 CrossRef CAS PubMed.
- J. A. Rather and K. D. Wael, Sens. Actuators, B, 2013, 176, 110–117 CrossRef CAS PubMed.
- Y. Zhang, Y. Liu, X. B. Ji, C. E. Banks and W. Zhang, J. Mater. Chem., 2011, 21, 14428–14431 RSC.
- Z. X. Zheng, Y. L. Du, Z. H. Wang, Q. L. Feng and C. M. Wang, Analyst, 2013, 138, 693–701 RSC.
- C. Dodeigne, L. Thunus and R. Lejeune, Talanta, 2000, 76, 6871–6876 Search PubMed.
- P. M. Easton, A. C. Simmonds, A. Rakishev, A. M. Egorov and L. P. Candeias, J. Am. Chem. Soc., 1996, 118, 6619–6624 CrossRef CAS.
- S. Ahmed, N. Kishikawa, K. Ohyama, T. Maki, H. Kurosaki, K. Nakashima and N. Kuroda, J. Chromatogr. A, 2009, 1216, 3977–3984 CrossRef CAS PubMed.
- F. Pan, L. Liu, S. Dong and C. Lu, Spectrochim. Acta, Part A, 2014, 128, 393–397 CrossRef CAS PubMed.
- L. R. Luo, Z. J. Zhang, L. J. Chen and L. F. Ma, Food Chem., 2006, 97, 355–360 CrossRef CAS PubMed.
- M. Yamasuji, T. Shibata, T. Kabashima and M. Kai, Anal. Biochem., 2011, 413, 50–54 CrossRef CAS PubMed.
- F. Chen, S. Mao, H. Zeng, S. Xue, J. Yang, H. Nakajima, J. M. Lin and K. Uchiyama, Anal. Chem., 2013, 85, 7413–7418 CrossRef CAS PubMed.
- S. H. Wang, X. T. Wei, L. Y. Du and H. S. Zhuang, Luminescence, 2005, 20, 46–50 CrossRef CAS PubMed.
- C. Lu, J. G. Li, Y. Yang and J. M. Lin, Talanta, 2010, 22, 1576–1580 CrossRef PubMed.
- Z. Zhang, H. Cui, C. Lai and L. Liu, Anal. Chem., 2005, 77, 3324–3329 CrossRef CAS PubMed.
- J. Z. Guo, H. Cui, W. Zhou and W. Wang, J. Photochem. Photobiol., C, 2008, 93, 89–96 CrossRef PubMed.
- S. L. Xu and H. Cui, Luminescence, 2007, 22, 77–87 CrossRef CAS PubMed.
- S. J. Xu, F. N. Chen, M. Deng and Y. Y. Sui, RSC Adv., 2014, 4, 15664–15670 RSC.
- L. Shang, S. J. Dong and G. U. Nienhaus, Nano Today, 2011, 6, 401–418 CrossRef CAS PubMed.
- J. Zheng, P. R. Nicovich and R. M. Dickson, Annu. Rev. Phys. Chem., 2007, 58, 409–431 CrossRef CAS PubMed.
- I. Díez and R. H. A. Ras, Nanoscale, 2011, 3, 1963–1970 RSC.
- Y. Z. Lu and W. Chen, Chem. Soc. Rev., 2012, 41, 3594–3601 RSC.
- H. Qian, M. Zhu, Z. Wu and R. Jin, Acc. Chem. Res., 2012, 45, 1470–1478 CrossRef CAS PubMed.
- L. Hu, Y. Yuan, L. Zhang, J. Zhao and S. Majeed, Anal. Chim. Acta, 2013, 762, 83–86 CrossRef CAS PubMed.
- M. Deng, S. J. Xu and F. N. Chen, Anal. Methods, 2014, 6, 3117–3123 RSC.
- N. Goswami, A. Giri, M. S. Bootharaju, P. L. Xavier, T. Pradeep and S. K. Pal, Anal. Chem., 2011, 83, 9676–9680 CrossRef CAS PubMed.
- G. Merényi, J. Lind and T. E. Eriksen, J. Biolumin. Chemilumin., 1990, 5, 53 CrossRef PubMed.
- T. G. Burdo and W. R. Seitz, Anal. Chem., 1975, 47, 1639 CrossRef CAS.
- C. Guo, H. J. Zeng, X. J. Ding, D. He, J. J. Li, R. Yang and L. B. Qu, J. Lumin., 2013, 134, 888–892 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |