Involvement of process parameters and various modes of application of TiO2 nanoparticles in heterogeneous photocatalysis of pharmaceutical wastes – a short review
Santanu Sarkara,
Ranjana Dasa,
Heechul Choib and
Chiranjib Bhattacharjee*a
aDepartment of Chemical Engineering, Jadavpur University, Kolkata, India. E-mail: c.bhatta@gmail.com; cbhattacharyya@chemical.jdvu.ac.in; Fax: +91 33 2414 6203; Tel: +91 98364 02118
bSchool of Environmental Science and Engineering, Gwangju Institute of Science and Technology, Gwangju, Korea
First published on 17th October 2014
Abstract
In recent years, the occurrence of persistent organic compounds in industrial as well as municipal effluents is becoming a serious threat to the environment. The pharmaceutical compounds present along with those organics have a detrimental effect on our environment. Among various well established wastewater treatment technologies, advanced oxidation processes (AOPs) using TiO2 nanoparticles have shown promising results against various organic wastewater pollutants. This study represents an in-depth review of applications of TiO2 in the treatment of various pharmaceutical wastes and the effects of associated process controlling parameters. It also highlights aspects of different application techniques of TiO2 nanoparticles and the involvement of reaction kinetics in photocatalytic degradation.
1. Introduction
Increasing human and livestock populations, along with industrial developments, have raised concerns for environmental protection, waste management and pollution control along with the supply of safe drinking water. It has been estimated that approximately 4 billion people all over the world are facing limited access to a clean and sanitized water supply, and millions are dying from waterborne diseases annually.1 These statistical figures are steadily growing, due to an increase in water pollution from micro pollutants and contaminants discharged to the natural water cycle.2,3 Studies have shown that pharmaceutical wastes are directly entering the sewage treatment process4–6 and adversely affecting aquatic life.7–9 In the present context of a safe water supply, the necessity for the development of advanced, low-cost and high efficiency wastewater treatment technologies is becoming obvious. Techniques related to the removal of suspended solids, health-threatening coliforms and soluble refractory organic compounds from wastewater are very tedious and expensive.10 In the current scenario, processes such as adsorption, coagulation and concentration of the pollutants using phase change methodology do not completely eliminate or destroy the pollutants.11 Due to their high operating cost and generation of secondary pollutants, processes of sedimentation, filtration and chemical wastewater treatments are also not well accepted.12–15 To overcome the limitations of conventional wastewater treatments and effective utilization of available economic resources, various advanced treatment technologies have been adopted, optimized, and applied.12–14 Among well adopted technologies, membrane filtration, advanced oxidation processes (AOPs), and UV irradiation have proven beneficial in the removal of a wide range of challenging contaminants.Advanced oxidation processes (AOPs) are the modern trend of pharmaceutical and municipal waste water treatment. The AOPs can be categorized as: photolysis, ozonization, Fenton's oxidation, heterogeneous photocatalysis, electrochemical oxidation, ultrasound irradiation and wet air oxidation.16 According to Klavarioti et al.,16 ozonization and heterogeneous photocatalysis are the most accepted water and wastewater treatment methodologies. Several researchers have observed that ozonization is more suitable for the purification of drinking water17,18 than pharmaceutical waste water treatment because the latter requires several downstream processing steps. Hence, heterogeneous photocatalysis using nanoparticles has become more effective in the treatment of pharmaceutical wastewater. Nanoparticles offer high surface area, which presents a higher surface reaction rate, and they are also commercially available and photochemically stable. Due to their light absorption properties, charge transport characteristics, excited-state lifetimes and favourable combination of electronic structure,19 semiconductors, namely TiO2 and MnO2, are mostly used as photocatalysts in AOPs.
This review aims to study the application of TiO2 nanoparticles in the field of heterogeneous photocatalysis for treatment of pharmaceutical wastes as well as several other related parameters that control the process efficiency. Moreover, this review also covers several methods utilizing TiO2 nanoparticles where the processes are more acceptable as well as more economic. The involvement of different reaction kinetics related to photocatalytic degradation of pharmaceutical wastes using TiO2 has also been studied here.
2. Heterogeneous photocatalysis-a new trend of advanced oxidation process
Developments toward heterogeneous photocatalysis (HPC) in the presence of TiO2 are gradually rising and represent the highest percentage among all AOPs.16,20,85 The most important literature studies available on the photocatalytic degradation of pharmaceutical components, which have demonstrated the effectiveness of photocatalysis as a promising process for pharmaceutical wastewater treatment, have been summarized in Table 1.| Pharmaceutical wastes | Solvent matrix | AOP parameters | Reaction kinetics | Major observations | ||
|---|---|---|---|---|---|---|
| Nanoparticle | Sources | pH | ||||
| a NM – not mentioned. | ||||||
| 17β-Estradiol 0.05–3 μM | Acetonitrile/water | Immobilized TiO2 | UV light 300–400 nm | 1–12 | Langmuir–Hinshelwood (L–H) kinetics | Up to 98% degradation was possible after 3.5 hours of irradiation time. The variation of the reaction rate with pH was established, and it reached the maximum at pH 12. Thus, photocatalysis was more efficient than photolysis.21 |
| 17β-Estradiol, estrone, ethinylestradiol 0.1 mg L−1 | Deionized water | Immobilized nanostructured TiO2 | 20 W Backlight, 359 nm | NMa | First-order kinetics | Without catalyst, 90% removal was possible in 2 hours of irradiation but the same degradation was observed in the presence of TiO2 within 30 minutes.22 |
| 17β-Estradiol 1 μM | Deionized water | Suspended Degussa TiO2 (P25) | UV-365 nm | NM | First-order kinetics | The total removal of 17β-estradiol was possible, and it lost its activity. After 3 hours, the mineralization process completed and all byproducts were identified.23 |
| 17β-Estradiol, estrone 250 μg L−1 | Deionized water and industrial effluent | Immobilized TiO2 | UV-black fluorescent lamp | NM | First-order kinetics | The rate of reaction depended on the diffusion constant. As the rate of diffusion of substrate molecules increased with the surface area of catalyst and the temperature, the rate also increased with increase of both parameters.24 |
| Buspirone 15 mg L−1 | Distilled water | Suspended P25 | Artificial sunlight | NM | First-order kinetics | Several structures of byproducts were identified with different mechanisms. Maximum drug removal was possible during photocatalysis.27 |
| Unconjugated and conjugated estrone and estradiol 1 mM | Distilled water | Immobilized TiO2 on glass beads | UV lamp | NM | First-order kinetics | 17-glucuronide and estrone were completely depredated within 4 h of UV irradiation. Conjugates remained unaltered after 6.5 hours of the oxidation process.28 |
| Clofibric acid, carbamazepine, iomeprol and iopromide | Distilled water | Suspended P25 and Hombikat UV100 | 1000W Xe short-arc lamp <400 nm | 6.5 | L–H kinetic model | P25 showed better degradation of clofibric acid and carbamazepine than Hombikat UV100 but in the case of iomeprol, this was reversed; higher adsorption of iomeprol by Hombikat UV100 was observed. Possible degradation pathways were established.29 |
| 17β-Estradiol, estrone, 17α-ethylestradiol, 10 μg L−1 | Distilled water | Immobilized TiO2 | High-pressure mercury UVA lamp | NM | First-order kinetics | The photocatalysis process was much faster than direct photolysis. Immobilized TiO2 inside the photoreactor enabled the reuse of the catalyst more than one time. Thus, the efficiency of the process was improved.30 |
| 17α-Ethinylestradiol, 17β-estradiol, estriol 0.1–3 μM | Acetonitrile/water | Immobilized P25 | UVA & UVB | 3–4 | First-order degradation kinetics | Photocatalysis was more effective than photolysis. The rate of reaction increased with light intensity and initial substrate concentration. Pt and Ag were added in small amounts to the system to boost the reaction; only Pt enhanced the performance of the reaction.31 |
| Iomeprol, clofibric acid, carbamazepine ∼2 mg L−1 | Deionized water | Suspended P25 or Hombikat TiO2 | 254 nm | 6.8 | Not mentioned | Microfiltration with backwashing enhanced the reusability of TiO2 and the membrane. Hombikat was more active in the pilot plant than other types of TiO2 but batch studies showed reverse results.32 |
| Carbamazepine, clofibric acid, iomeprol 0.5–5.2 mg L−1 | Spiked lake water | Suspended P25 or Hombikat TiO2 | Artificial sunlight | 6.5 | Pseudo-first order kinetics | P25 had more activity than other nanoparticles. The rate of degradation decreased and increased with increasing substrate concentration and TiO2 loading, respectively. The total mineralization was performed by photocatalysis.33,34 |
| Lincomycin 10–75 μM | Distilled water | P25 coupled with nanofiltration | Sunlight | 6.3 | First-order kinetics | A membrane photoreactor enabled the separation of nanoparticles and byproducts during degradation of the drug. Thus, TiO2 could be used more than one time.35 |
| Sulfamethazine 10–70 mg L−1 | Distilled water | Suspended P25 or ZnO | 350–400 nm | 4.8 | First-order kinetics | The result showed that ZnO was more effective than TiO2. In the presence of H2O2 the degradation rate increased; the rate also increased with catalyst loading.36 |
| B-Estradiol 0.5 mg L−1 | Distilled water | Suspended TiO2 | 366 nm | 3–11 | NM | Both the adsorption and degradation increased considerably with increasing pH as the OH radical content of the reaction mixture increased simultaneously.37 |
| Diclofenac, 0.76–15 mg L−1 | Distilled water | Suspended P25 | Artificial sunlight | Ambient pH | L–H kinetic model | Response surface methodology (RSM) was employed to optimize catalyst loading and drug concentration. Finally, the toxicity was removed.38 |
| Triclosan 15–37 μM | Distilled water and surface river water | Suspended P25 or anatase TiO2 | 300–450 nm | 5 | First-order kinetics | P25 was more active than other types of TiO2. Up to a certain range, the degradation rate increased with catalyst loading. Detection of intermediates, byproducts and pathways was achieved for complete removal of the drug after 60 minutes of reaction. Degradation was slower in the case of river water than in distilled water.39 |
| Triclosan 9 mg L−1 | Distilled water | Suspended P25 | UV-365 nm | NM | NM | Up to 95% degradation of triclosan was possible. The mineralization efficiency increased with the addition of H2O2. Byproducts and degradation pathways were identified.40 |
| Furosemide, ranitidine, ofloxacin, phenazone, etc. 5–10 mg L−1 | Distilled water and surface river water | Suspended P25/MP UV coupled with nanofiltration | 125 W medium pressure Hg lamp | 2–12 | First-order kinetics | The degradation rate increased with pH. Filtration separated catalyst particles for reuse. Characteristics of different types of membranes were studied in both alkali and acid media.41 |
| α-Methyl-phenylglycine, 500 mg L−1 | Distilled water | Suspended P25 | Sunlight | 2.7–2.9 | NM | Complete drug degradation was possible, and the chemical oxygen demand (COD) value reduced to 504 mg L−1 after 1500 minutes of irradiation time. LCA (Life cycle assessment) measured the environmental impact of degraded by-products.43 |
| Tetracycline 40 mg L−1 | Deionized water | Suspended P25 | HPLN (>254 nm), solarium (300–400 nm), black light (365 nm) | NM | NM | Photocatalysis was more effective than photolysis. The rate of oxidation was higher under UV and solarium radiation. Partial mineralization was possible. However, the antibacterial activity of the byproducts was reduced completely after 1 h of irradiation.44 |
| Sulfamethoxazole 25–200 mg L−1 | Distilled water | Suspended P25 | Artificial sunlight | 2–11 | First-order kinetics | The effects of catalyst loading and pH were studied. After 6 hours of irradiation, aromatic compounds were found due to the presence of sulphur and nitrogen-containing aromatics.45 |
| Sulfamethoxazole 5–500 μM | Deionized water with NOM and bicarbonates | Suspended P25 or anatase or rutile TiO2 | UV-(324–400 nm) | 3–11 | Pseudo-first order reaction followed by L–H kinetics | The rate of the reaction depended on the concentration of drug, the concentration of TiO2, and the pH of the medium. P25 showed more activity compared to other nanoparticles. Intermediates and by-products were identified and mechanism pathways were established.46 |
| Salbutamol 15 mg L−1 | Distilled water | Suspended P25 | Artificial sunlight | 2.5–9.5 | L–H kinetics | RSM was implemented to optimize catalyst loading and pH. The by-products and the pathway of the reaction were identified. The toxicity level gradually decreased.47 |
| 17α-Ethinylestradiol, 17β-estradiol, estriol 0.8 mg L−1 | Distilled water | Immobilized P25 | Artificial sunlight or UV-350 nm | NM | First-order kinetics | Among all heterogeneous photocatalysis, UV-irradiated photocatalysis was the most effective. A TiO2 immobilization technique was employed to improve performance by recycling the photocatalyst.48 |
| Gemfibrozil, tamoxifen 2.5–50 mg L−1 | Deionized water | Suspended P25 or anatase TiO2 | UV-360 nm | 10 | Pseudo-first order kinetics followed by L–H model | The impact of photocatalysis on tamoxifen was negligible as it was degraded using photolysis, but the opposite was observed for gemfibrozil. P25 was more effective than other types of TiO2. The pathway of reaction was understood and verified.49 |
| Estrone, 17β-estradiol 0.1–1 μg L−1 | Deionized water | Suspended P25 | UV-253 nm or UV-238–579 nm | 2–10 | First-order kinetics | The reaction at 253 nm was three times faster than at 238–579 nm. Degradation increased with catalyst loading and H2O2 addition and also depended on pH. Humic substances facilitated degradation due to photosensitization.50 |
| Imipramine, 15 mg L−1 | Deionized water | Suspended P25 combined with Fenton | Xenon arc lamp, 290 nm | NM | L–H kinetics model | The combined effects of H2O2 and Fe2+ on photocatalytic degradation were analyzed. The application of an artificial neural network (ANN) was introduced for optimization. The by-products were identified; however, they were found to be as toxic as imipramine and resistant to photocatalysis.51 |
| Chloramphenicol, 10–80 mg L−1 | Deionized water | Suspended P25 or anatase TiO2 or ZnO | 320–400 nm | 5 | L–H kinetics model | The rate of degradation increased with substrate and catalyst concentration as well as the addition of H2O2. P25 was more effective than ZnO. The complete pathway of the reaction was identified. Within 90 minutes, the target drug was completely removed.52 |
| Diclofenac, naproxen, ibuprofen, 25–200 mg L−1 | Deionized water | Suspended P25 | Artificial sunlight | NM | First-order kinetics | The total organic carbon (TOC) decreased with TiO2 and O2 concentration. Temperature affected the degradation of naproxen. Byproducts were identified; however, the post biological treatment could be performed for byproducts of ibuprofen.53 |
| Paracetamol 2–10 mM | Deionized water | Suspended P25 | UV-254 nm or UV-365 | 3.5–11 | Pseudo-first order kinetics | The rate of degradation was much higher under UVC irradiation than UVA. Up to certain concentrations of TiO2, the value of the reaction rate constant increased, and it also depended on the initial substrate, O2 concentration and pH of the reaction medium. The byproducts were identified.54 |
| Triclosan | Deionized water | Suspended P25 in photo reactor | UVA lamp | 6–8 | Pseudo-first order followed by L–H kinetic model | The intermediates during photocatalysis were identified. Fewer toxic elements are formed using TiO2 photocatalysis compared to other methods. This method could reduce the formation of dioxin with effective mineralization of triclosan.56 |
| Mixture of amoxicillin 10 mg L−1, carbamazepine 5 mg L−1, diclofenac 2.5 mg L−1 | Deionized water | Suspended P25 | Artificial UV 300–420 nm | 4–5.5 | Pseudo-first order followed by L–H kinetic model | Effective mineralization, detoxification and degradation could be possible using heterogeneous photocatalysis. The intricate relationships between process parameters were identified.57,59 |
| Ofloxacin and the β-blocker atenolol, initial concentration 5–20 mg L−1 | Deionized water | P25 suspension in the range of 50–1500 mg L−1 | UVA lamp 350–400 nm | 3–10 | L–H kinetic model | The effects of different process parameters were studied, and addition of H2O2 enhanced the effect of photocatalytic reaction. Its intermediates were more stable and less toxic compared to parent molecule.61 |
| Diclofenac, initial concentration 5–20 mg L−1 | Deionized water | Six types of TiO2 suspension | UVA lamp 350–400 nm | 6 | NM | The conversion rate for different types of TiO2 was analyzed, and an effective type was identified. H2O2 enhanced the photocatalytic reaction.62 |
| Sulfachlorpyridazine, sulfapyridine and sulfisoxazole | Deionized water | TiO2 suspension | High-pressure mercury lamp, 365 nm | 3–11 | Pseudo-first order followed by L–H kinetic model | Up to 90% removal of sulfa drug was possible after less than 60 minutes of illumination time. The pH of the reaction matrix played a significant role in photocatalytic degradation. However, the rate of degradation increased with catalyst loading.63 |
| Oxolinic acid 20 mg L−1 | Deionized water | P25 suspension | Cylindrical black light lamp as UV source | 7.5–11 | Pseudo-first order kinetics | Formation of intermediates was confirmed. Optimization showed that 1.0 g L−1 TiO2 concentration at pH 7.5 was the most favourable condition for photocatalysis.64 |
| Amoxicillin, ampicillin and cloxacillin with concentrations of 104, 105 and 103 mg L−1, respectively | Distilled water | TiO2 suspension | UVA lamp 365 nm | 5–11 | Pseudo-first order kinetics | The degradation of drugs was very low at 300 nm irradiation and maximum degradation was achieved at pH 11. Complete degradation was possible at pH 5 in the presence of H2O2 with TiO2. Rate constants were calculated for the degradation of different drugs.65 |
| Carbamazepine, clofibric acid and iomeprol | Deionized water | TiO2 suspension with activated carbon in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
UV Hg lamp, below 300 nm | 7.5 | Pseudo-first order kinetics | High removal efficiency was observed using TiO2 suspension. The addition of activated carbon reduced the intermediates, although it enhanced turbidity of the current system. The lower affinity of clofibric acid towards activated carbon provided a higher surface area, which led to faster degradation rates.66 |
| Sulfonamides (sulfathiazole, sulfamethoxazole and sulfadiazine) | Deionized water | P25, FeCl3 | UV lamp, maximum irradiation 366 nm | 3–8 | First and second order kinetics | Removal of sulphonamides was 15 times higher in the presence of FeCl3 and HCl along with TiO2 than TiO2 alone.67 |
| Chloramphenicol, initial concentration 6.6–23.4 mg L−1 | Deionized water | P25 suspension | Mercury lamp, 365 nm | 4–9 | NM | The parameters were optimized using RSM and optimized parameters were pH 6.4, TiO2 concentration 0.94 g L−1 and initial substrate concentration 19.97 mg L−1.68 |
| Indomethacin, concentration 0.1 to 1.5 mmol L−1 | Deionized water | P25 with activated carbon | 125 W medium pressure mercury lamp | NM | Pseudo-first order kinetics followed by L–H kinetic model | Langmuir, Freundlich, and Sips isotherms were used to describe adsorption. With the increase of TiO2 concentration, the rate of adsorption and rate of reaction increased, and those reached the maximum value with 10% of TiO2 concentration.69 |
| Caffeine, diclofenac, glimepiride and ibu-profen with initial concentration of 100 μg L−1 and 25 μg L−1 for methotrexate | Deionized water | TiO2 modified with SiO2 | Solar bath | NM | Zero order or pseudo-first order kinetics | TiO2 was successfully modified with SiO2. Both wastewater and simulated solution were used for the experimental study. Lower degradation was observed in the case of real wastewater. Removal percentage of those drugs was 79–96%.70 |
| Atenolol, metoprolol and propranolol with initial concentration of 50–200 μM | Milli-Q water | P25 | High-pressure mercury UV lamp, 365 nm | 3–11 | Pseudo-first order kinetics followed by L–H kinetic model | Complete mineralization of substrates and intermediates was possible by photocatalysis. Adsorption played the major role in photocatalytic degradation. The pathway of the reaction and intermediates were identified.71 |
| Lamivudine, concentration 100 μM | Distilled water | P25 | Mercury lamp, 365 nm | 3–11 | First-order kinetics followed by L–H kinetic model | Maximum degradation was possible at pH 9, TiO2 1 g L−1 and with initial substrate concentration of 100 μM. The process was optimized using RSM. A tentative reaction mechanism was established.72 |
| Trimethoprim, initial concentration 2–50 mg L−1 | Distilled water | P25 | Artificial UV lamp, 352 nm | 3–8 | First-order kinetics | Rate of degradation decreased with increasing drug concentration, but the rate remained constant above UV intensity 47 mW cm−2 and TiO2 concentration of 0.5g L−1. A continuous mode of degradation was successfully attempted.73 |
| Sulfamethoxazole, initial concentration 2.5–30 mg L−1 | Ultrapure water | P25, Hombikat, Millennium PC-50/100/105/500 | UVA lamp, 350–400 nm. | 5–6.7 | L–H kinetic model | Effectiveness of distinct type of TiO2 catalyst was analyzed and P25 was the most effective catalyst. At the same type, the influences of different process parameters were investigated.74 |
| Amoxicillin and cloxacillin | Wastewater | TiO2 suspension | Artificial UV lamp, 365 nm | 5 | NM | The most desirable conditions were AOP followed by SBR; the process showed 57% removal efficiency, which was the limitation of the whole process.75 |
| Carbamazepine, concentration 10 mg L−1 | Distilled water | TiO2 slurry form used in MBR | UVA lamp, 360 nm | NM | Pseudo-first order followed by L–H kinetics | Up to 95% carbamazepine removal was possible with 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 recycle ratio and below the concentration of 10 mg L−1 it was not biologically degradable.76 1 recycle ratio and below the concentration of 10 mg L−1 it was not biologically degradable.76 |
| Norfluoxetine, lincomycin, etc. Initial concentration 0.1 g L−1 | Ultra pure water | TiO2 nanowire membrane | 100 W artificial UV irradiation | 6.7 | Pseudo-first order kinetics | A successful attempt was made to synthesize a TiO2 nanowire membrane. In the presence of UV light this was more effective than normal TiO2 to degrade pharmaceutical materials.77 |
| Norfloxacin | Deionized water | C–TiO2 suspension, 0–2.0 g L−1 | Mercury lamp, 420 nm | 2.5–11.8 | L–H kinetics | The most important observation from the recycling study was that degradation efficiency was nearly the same for fresh and used TiO2. The addition of OH− enhanced the performance of the system.78 |
| Amoxicillin trihydrate | Deionized water | TiO2 and Sn/TiO2 nano particle suspension | 15W (UVC) mercury lamp, 254 nm | 7 | Pseudo-first order reaction followed by L–H kinetic model | Sn doping enhanced the adsorption efficiency due to enhancement in the generation of hydroxyl radicals, band gap energy, specific surface area, decrement in crystal size, etc. Thus, the degradation efficiency of photocatalysis increased.79 |
| Levofloxacin, initial concentration 20 mg L−1 | Deionized water | P25, concentration of 0.05–0.5 g L−1 suspension | UVC lamp, 254 nm | 6.5 | NM | A comparative study was made between ozonization and heterogeneous photocatalysis. The intermediates were more favorable in the oxidation process, and it showed higher mineralization efficiency compared to heterogeneous photocatalysis. The intermediates had no antibacterial properties.80 |
| Venlafaxin, atorvastatin, ibuprofen, naproxen, gemfibrozil, lincomycin, norfluoxetine, etc. | Deionized water | TiO2 nanowires suspension | Low pressure mercury lamp, 264 and 365 nm | 7.4 | Pseudo-first order reaction kinetics | Study indicated that the degradation mechanism of waste was a simultaneous process of surface adsorption and photocatalytic degradation. TiO2 nanowires showed more effective mineralization compared to normal TiO2 nanoparticles.81 |
| Famotidine, tamsulosin and solifenacin | Distilled water | Tetra(4-carboxyphenyl) porphyrin (TCPP)-TiO2 composite | 500 W halogen lamp and sunlight | NM | NM | Study showed that a higher degradation rate of famotidine by the nanocomposite was observed compared to unmodified P25. Recycling and reuse of the photocatalyst was possible in the case of the composite materials. Solar photolysis showed promising results.82 |
| Carbamazepine | Deionized water | TiO2 suspension | UVC | 4–11 | L–H kinetic model | Complete degradation of carbamazepine was observed via photocatalysis in 30 min. The addition of O2 improved the activity of the catalyst.83 |
| Carbamazepine, initial concentration 1 mg L−1 | Deionized water | N-doped TiO2 suspension | Hg vapor lamp | 2–8 | NM | The surface coating of modified N-doped TiO2 ensured multiple uses of the same catalyst. The modified catalyst surface did not absorb the substrate. Less removal of carbamazepine was observed in the presence of other organic matter. The performance of AOP was reduced with increasing alkalinity.84 |
| Ibuprofen, initial concentration 5–60 mg L−1 | Ultrapure water | P25 suspension | UV–Vis solarium lamp | 3–9 | NM | Study showed that pH ∼7 is favourable for the photocatalytic reaction. The catalyst activity was enhanced using an optimal catalyst to substrate ratio. Intermediates were identified, and these had a great impact on the reaction.86 |
| Sulfamethoxazole, diclofenac sodium, hydrochlorothiazide, 4-acetamidoantipyrine, nicotine and ranitinide hydrochloride with initial concentration of 10 mg L−1 | Milli-Q water | P25 suspension | 150 W medium pressure mercury UV lamp, 320 nm | NM | First-order kinetics; for 4-acetamido-antipyrine, zero order | After 6 h irradiation time, over 90% removal was possible. 20% of total organic carbon was removed; however, intermediates were not indicated here.87 |
| Naproxen and carbamazepine with initial concentrations of 60.1 and 125 mg L−1 respectively | Deionized water | P25 nanobelt suspension | 100 W middle pressure mercury UV lamp, 365 nm | 4–10 | Pseudo-first order kinetics | TiO2 nanoparticles were modified to synthesize a nanobelt photocatalyst. The effect of adsorption on photocatalysis was also studied. Moreover, experiments with the addition of foreign substances to enhance the activity of nanoparticles were also attempted. Intermediates were not identified.88 |
| Diclofenac | Deionized water | C- and C, N-codoped TiO2 suspension | Artificial UV | Pseudo-first order kinetics | Up to 60% COD removed using modified nanoparticles. The anastase had better activity than the rutile phase. Complete mineralization was not possible using modified TiO2.89 | |
| Carbamazepine and carbamazepine epoxide, acridine, and acridone with initial concentration of 10 μg mL−1 | MilliQ water | TiO2 and ZnO nanoparticle suspension | Artificial solar illumination, Xenon lamp (1500 W lamp, 300–800 nm) | 3–11 | Pseudo-first order kinetics | In the presence of ZnO nanoparticles, the activity of TiO2 nanoparticles was reduced. Lower pH was favourable for degradation when only TiO2 was introduced but in the case of ZnO, lower and higher pH levels (3 and 11) were favourable conditions. Higher ionic strength of the reaction mixture increased the reaction rate in the presence of TiO2 only. Intermediates were detected.90 |
| 5-Fluorouracil, 200 μg/L and cyclophosphamide (27.6 mg L−1) | Milli-Q water | Aldrich-TiO2, P25, and ZnO suspension | UV lamp, 8 W/254 nm | 3–10 | NM | In this comparative study, it was observed that P25 was the best photocatalyst and complete removal was possible within 2 h for 5-fluorouracil and within 4 h for cyclophosphamide. Catalyst loading was optimised at 20 mg L−1. However, the effect of initial substrate concentration was not described. Byproducts were identified.91 |
| Chlorhexidine digluconate, initial concentration of 500–1500 mg L−1 | Deionized water | Aldrich-TiO2, P25 suspension | UVA lamp, 10 W, 365 nm | 4–11 | NM | Approximately 70% chlorhexidine removal was possible. The effect of all parameters was studied and optimization was performed using both ANFIS and RSM. Toxicological tests were performed to confirm that the byproducts had no detrimental effects on the environment.92–94 |
HPC by definition is the oxidation of target molecules with the help of active radicals using nanoparticles as a catalyst under UV light illumination. As soon as the UV radiation energy becomes equal to or larger than the band gap energy, the minimum energy required to move one electron from the valance band to the conduction band, electrons (e−) move from the valance band to conduction at the photocatalytic surface which results in the generation of holes (h+). The photo holes convert the water (H2O) molecules to hydroxyl radicals (OH˙) and hydrogen cations (H+). The formed OH˙ works as a strong oxidizing agent and it transforms organic substances, mainly present in pharmaceutical wastewater, into carbon dioxide, water and other often less hazardous materials than the original substance.
The overall mechanism for the process can be represented using eqn (1)–(4).
| Photocatalytic nanoparticle + hγ → e− + h+ | (1) |
| e− + O2 → O−2 | (2) |
| h+ + H2O → OH˙ + H+ | (3) |
| OH˙ + Pharmaceutical waste → CO2 + H2O + Others | (4) |
In heterogeneous photocatalysis two reactions occur consecutively; the first reaction is the reduction of oxygen and the second is the oxidation of pharmaceutical wastes by the hydroxyl radical. During the excitation of TiO2 nanoparticles, electrons are accumulated in the conduction band, which causes the generation of a hole. Therefore, it is necessary to consume electrons to increase the efficiency of HCP in the presence of a nano photocatalyst; here, the electrons are consumed by the dissolved oxygen. The overall reaction has been schematically represented in Fig. 1. Some important parameters which influence photocatalytic oxidation are light intensity, catalyst concentration, substrate concentration, temperature and pH of the medium.21–93
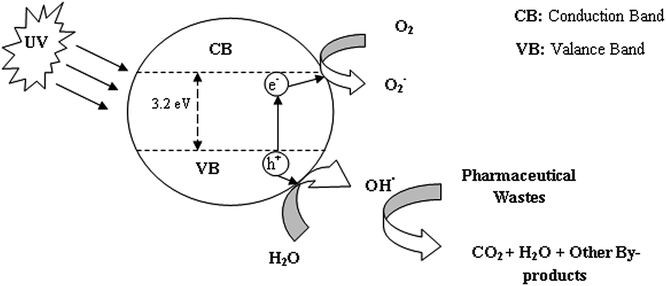 | ||
| Fig. 1 Schematic representation of photocatalysis of pharmaceutical wastes on the surface of TiO2 nanoparticles. | ||
3. Process parameters involved in the photocatalytic degradation process
HPC is a surface phenomenon where reaction occurs inside the active site of the catalyst in the presence of UV irradiation. As with every chemical reaction, several parameters, such as catalyst loading, substrate concentration, pH of the medium, presence of other materials, etc. affect the reaction rate during the photocatalytic degradation. Several research groups have highlighted those parameters and their influences on photocatalysis. In the subsequent sections, the adverse effects of process parameters on photocatalytic degradation will be discussed.3.1. Effect of initial substrate concentration
The initial concentration of the target molecule is the primary constraint for the photocatalytic reaction, and the catalyst loading is dependent on this parameter. In the photocatalytic reaction, substrate molecules are adsorbed on the active surface of the catalyst; therefore, the degradation rate significantly depends on the population of target molecules on the active surface. If the substrate concentration is higher, competition between target molecules for attachment to the catalyst surface becomes more pronounced and thus the rate of degradation decreases with an increase in the initial substrate concentration. Many researchers have studied the variation of rate with initial concentration of pharmaceutical waste. Among them, Coleman et al.30 reported that the degradation rate of 17β-oestradiol was linearly dependent on the initial substrate concentration. A slightly different observation, noted by Hu et al.,46 is that initially, the rate of the reaction increased with increasing aqueous concentration of sulfonamides up to a certain value. According to them, the initial increment was expected because the substrate adsorption rate at the catalyst surface was higher at the initial stage and after that, the reaction rate was independent of initial substrate loading. After the initial stage of reaction, no active sites on the TiO2 surface are available for further adsorption until completion of the earlier reaction. At that time, an increment in substrate loading does not affect the reaction rate. However, Yang et al.54 determined that the degradation rate of paracetamol decreased with an increase in its initial concentration. They explained that at higher concentrations, a large number of substrate molecules occupied the maximum number of active sites of the catalyst and thus caused a decrement in available protons for the oxidation process. After that, Mendez-Arriaga et al.,53 Rizzo et al.,57 Hapeshi et al.,61 Achilleos et al.,62 Yanga et al.,63 Yang et al.,71 An et al.,72 Ho et al.,73 Chen and Chu,78 Mohammadi et al.,79 Das et al.92 and Sarkar et al.93 reported similar results to Yang et al.54 with their various target materials. Xekoukoulotakis et al.74 observed that the rate of degradation initially increased with an increase in the initial substrate concentration up to a certain level, but after that it decreased with increasing initial concentration. They gave a similar explanation to that had been already stated by Hu et al.46 Therefore, initial substrate concentration plays a crucial role in photocatalytic degradation and as it increases, the rate of degradation along with efficiency of the photocatalysis decreases.3.2. Effect of catalyst loading
The rate of degradation of pharmaceutical wastes directly depends upon the catalyst loading, as the available active surface area of the catalyst increases with its concentration and thus the surface reaction is also enhanced. In 2006, Calza et al.38 reported that the efficiency of photocatalytic degradation increased with TiO2 concentration up to a certain level and after that, the situation was reversed due to scattering of the incident UV-irradiation occurring on the TiO2 surface. Thereafter, Hu et al.46 determined that the degradation rate is not always directly proportional to the catalyst loading, and they explained that this was due to a decrease in the number of photons or increase in UV scattering from the catalyst surface with an increase in TiO2 loading. Sakkas et al.47 made the same observation up to 600 mg L−1 TiO2 concentration, but after that particular value, the rate of reaction decreased due to shrinkage of the active volume of the active sites. In the same year, Abellan et al.45 varied TiO2 concentration in a range of 0.0 to 2.0 g L−1 to degrade sulfamethoxazole, and their observation was as same as Hu et al.;46 however, they explained the behaviour as a consequence of uneven competition between nanoparticles for light absorption at higher concentrations. Moreover, an increment of the rate of degradation of paracetamol with increasing TiO2 loading was described by Yang et al.54 up to 0.8 g L−1; the rate decreased beyond 5 g L−1 TiO2 concentration due to a decrease in light penetration through the active sites. Boroski et al.58 made a similar statement from their experimental observations.58 The observations of Abellan et al.45 and Yang et al.54 on reaction rate behaviour and catalyst loading were alike for different target molecules, as previously mentioned. However, Yang et al.54 did not describe any decrement in reaction rates at higher concentrations of TiO2, but stated that the rate remained constant. Other groups of researchers, Mendez-Arriaga et al.53 and Giraldo et al.,64 supported the statement made by Hu et al.46 The screening effect of UV irradiation was sometimes found to be responsible for a decrement in the rate of photocatalysis for catalyst loading higher than the threshold value.61,65 Though catalyst loading is expected to be directly proportional to substrate concentration, an excess of catalyst provides an obstacle for light penetration on the active surface. Thus, a proper ratio of initial substrate and catalyst concentration should be maintained.62,72 However, Yanga et al.63 and Ho et al.73 did not consider the effect of excess catalyst loading as they observed that rate of reaction increased with increasing TiO2 loading. Accepting the findings of Hu et al.,46 Elmolla and Chaudhuri65 explained that the decrement of the degradation rate at a higher concentration of TiO2 was due to agglomeration and sedimentation of TiO2. Though the rate increases with catalyst loading, Boroski et al.,58 Elmolla and Chaudhuri,65 Xekoukoulotakis et al.,74 Chen and Chu,78 Mohammadi et al.79 and Nasuhoglu et al.80 reported that lower UV penetration through available active sites at higher concentrations of TiO2 was the vital cause for the decrement in reaction rates, which was formerly mentioned by Yang et al.54 It appears that with an increase of TiO2, the opacity of the reaction solution increases, and as a result UV irradiation is obstructed by the working solution; certainly, some portion of UV irradiation should be absorbed by the working solution. Very recently Lin and Lin91 reported that higher concentrations of TiO2 (>20 mg L−1) enhanced light scattering, which reduced light penetration through the active surface of the catalyst. Therefore, catalyst loading should be optimized for photocatalytic degradation of pharmaceutical waste so that UV light can penetrate the TiO2 suspension and provide active sites with the necessary energy for activation.3.3. Influences of various phases of TiO2 nanoparticles
Current ly, various phases of TiO2 nanoparticles are easily available such as Degussa (P25), Hombikat, anatase, rutile, etc. Many researchers have made comparative studies to identify the efficiency of various phases of TiO2 for pharmaceutical wastewater treatment. A large number of studies46,33 showed that P25 was more effective than Hombikat TiO2 as the latter contains only anatase phase and had higher adsorption capacity whereas P25 was a mixture of anatase and rutile phases and had a higher surface area. In 2006, Rafqah et al.39 made a comparative study of the degradation of triclosan to establish the higher effectiveness of P25 compared to PC50 and PC500. According to their report, P25 was a mixture of anatase and rutile where as others contained pure anatase phase. Achilleos et al.62 added new information about P25 and Hombicat UV100; according to their study, P25 showed higher degradation rates compared to Hombicat and they explained that P25 had a slower electron-hole recombination rate, and Hombicat had a faster interfacial electron transfer rate. Therefore, the availability of electrons and holes on P25 helped to increase the degradation rate. Xekoukoulotakis et al.74 compared the activity of six different types of TiO2; P25 showed the best result, as the rates of recombination of holes and electrons were slower than those of the other types; an explanation had already been provided by Achilleos et al.62 Therefore, literature reports indicate that P25 is the best choice of catalyst for the photocatalytic degradation of pharmaceutical wastes and that type of TiO2 has already been used by a large number of researchers.3.4. Effect of pH of the solution matrix
Variation of pH means alteration in the concentration of H+ and OH− ions in a reaction mixture. The concentration of hydroxyl radical plays a vital role in photocatalytic degradation; this can be explained using eqn (4), as OH˙ is a key species for oxidation. Thus, the increase of pH means an increase of OH− and its transformation to OH˙ radicals as well. Therefore, the rate of oxidation improves further in the presence of OH˙ radicals. That principle is applicable, to some extent, but actual surface chemistry is more relevant to explain the effect of pH. As the photocatalytic reaction is a surface phenomenon, the rate of degradation of any compound depends on the attachment rate of the substrate on the active surface of the catalyst. That attachment rate totally depends on the pH of the medium and can be explained with the help of point of zero charges (pzc) of TiO2 at pH 6.25. Thus, the TiO2 surface is positively charged in acidic media; on the other hand, it is negatively charged in alkaline media. If the substrate is negatively charged, an acidic matrix is favourable for absorption, whereas a positively charged substrate shows better absorption in alkaline medium. The above explanation can also be described with the help of the logarithmic acid dissociation constant (pKa) of the substrate. Acidic medium is favourable for lower pKa values, and the opposite is true for substrates with higher pKa values. Therefore, the pH of the reaction medium greatly influences the efficiency of photocatalysis. The chemical nature of the substrate also affects the reaction behaviour. Coleman et al.21 observed the variation in reaction rates within a pH range from 1 to 12. They explained that when the pH increased, the formation of hydroxyl radicals increased, and thus the oxidation process was also enhanced up to pH 7, which was quite obvious. After that, the rate decreased up to pH 10 due to the formation of large amounts of phenoxide ion from oestradiol, which competed with hydroxyl ion for absorption. However, subsequently in the range of pH 10 to pH 12 the phenoxide ions were neutralized by unprotonated hydroxyl radicals and formed neutral phenoxy radicals which could react at the TiO2 surface. Due to neutralization of the phenoxide ions, the reaction rate increased after pH 10. Moreover, Hu et al.,46 Sakkas et al.,47 Yang et al.,54 Chen and Chu78 and Avisar et al.84 cited that the rate of photocatalytic reaction increased in the alkaline range of pH as at higher pH (>7) the formation of OH− was also enhanced, which caused the reduction of photoholes. Sakkas et al.47 reported that at lower pH, the active surface area of the photocatalyst was reduced due to agglomeration of TiO2. However, Hu et al.46 observed that the degradation of sulfamethoxazole was not affected with variation of the pH of the medium though removal of total organic content (TOC) was enhanced with increasing pH. Contrastingly, Rizzo et al.57 did not observe any improvement in the degradation kinetics of triclosan with an increase in pH since the pKa value of the substrate varied from 7.9 to 8.1, but the rate decreased in acidic medium due to inhibition of the hydroxyl radical. At the same time, a different observation was made by Giraldo et al.,64 where they stated that degradation of oxolinic acid was favored at pH > 7.5 but less than 11 as at pH > 11 the repulsion between oxolinic acid and the catalyst surface increased. Moreover, Hapeshi et al.61 stated that pH of the solution lower than the pKa value of the substrates was favourable but that the medium should be alkaline in nature. A pH value higher than the pKa value lessened the attraction between the substrate and the catalyst surface and as a result, the rate of reaction decreased. Recently, Yanga et al.,63 An et al.,72 Xekoukoulotakis et al.74 and Chen and Chu78 generalized the influence of pH on reaction rate that the pH of the medium depended on the pKa value of the substrate. According to their suggestion, acidic medium was favourable for substrates with lower pKa values and alkaline medium was favourable for reverse cases. During the degradation of amoxicillin, Elmolla and Chaudhuri65 mentioned that at acidic pH both the substrate and catalyst surfaces were positively charged, and hence surface repulsion was enhanced; on the other hand, in alkaline media both surfaces carried negative charges and thus repulsive force was generated. Therefore, they reported that neutral pH was more favourable than acidic pH but alkaline pH was acceptable due to the greater availability of OH radicals, which favoured the reaction, as well as the instability of the target material at higher pH. Moreover, Boroski et al.58 found improved degradation of pharmaceutical waste in acidic media as negatively charged pollutants showed better absorption on the TiO2 surface in that particular condition. Therefore, acidic media favoured the above reaction. In general, the degradation depends on the pKa value of the reactants or the surface charge of the pollutants and the pH of the media should be adjusted accordingly.3.5. Effect of energy source
Like all other parameters, light intensity plays an important role in photocatalytic reactions. The band gap energy of 3.2 eV for TiO2 nanoparticles is supplied from the light energy of UV-irradiation or sunlight. In some instances, visible light fails to provide this amount of energy for photocatalysis. At an earlier stage in the field of photocatalytic degradation of pharmaceutical waste, Augugliaro et al.35 achieved the degradation of lincomycin in a solar photoreactor in the presence of TiO2 nanoparticles. The following year, Munõz et al.43 tried the same type of experiment and achieved successful removal of pharmaceutical wastes. Thereafter, as per the available literature, only one other group of researchers70 attempted to carry out the photocatalysis experiment under sunlight. Although earlier researchers35,43,70 reached their targets successfully using solar illumination, most researchers opted to use UV irradiation due to the uncertainty of solar radiation. In 2005, Coleman et al.31 studied the dependency of the reaction rate on light intensity. They found that the rate of degradation of 17β-oestradiol was proportional to the square root of UV intensity. Moreover, Abellan et al.45 carried out the experiment under a UV wavelength range of 240–310 nm to differentiate the effect of photocatalysis and photolysis. They found that photolysis had an impact upon the photocatalysis process. In the next year Yang et al.54 performed the photolysis of paracetamol in the presence of UVC (254 nm) irradiation but not UVA (365 nm), as paracetamol showed negligible absorbance above the wavelength of 350 nm and for the same reason; the rate of photocatalytic degradation was higher under a UVC source. They also proposed that rate was approximately proportional to the square of light intensity. In 2010 Elmolla and Chaudhuri65 made a general comment that if the substrate absorbed a certain wavelength of UV irradiation, it could not be degraded at that particular wavelength, but it would be degraded at a higher range of UV irradiation. The effect of light intensity on the degradation process was studied by Ho et al.,73 and they mentioned that the rate increased with light intensity up to a certain range. Consequently, light intensity and wavelength can be varied for different substrates, and these should be chosen properly to achieve effective degradation. Very recently, the current research group92 has also analysed the dependency of photocatalytic degradation of chlorhexidine on UV intensity, and it was observed that with increasing intensity the rate also increased up to 80 μW cm−2, but that the rate decreased after that intensity. They92 reported that this was because the hole-electron recombination rate also increased at a higher intensity; therefore, the hole-electron availability for photooxidation was reduced after a certain range of UV intensity. Thus, the wavelength and intensity of irradiation are target specific, and these should be adjusted very rationally.3.6. Effect of temperature variation
Heterogeneous photocatalysis in the presence of TiO2 nanoparticles is a combination of diffusion and reaction phenomena. First, the substrate molecules are adsorbed by means of surface diffusion on the catalyst surface, then they are degraded inside the active pores of TiO2. The change of temperature does not affect the reaction rate, it affects the rate of diffusion of substrate molecule on the catalyst surface. Very few literature reports are available where the dependency of photocatalytic reaction rate on temperature has been studied. This is because heterogeneous photocatalysis is less sensitive to temperature throughout the system. Among these, Nakashima et al.,24 considering that the rate of the reaction was dependent on the substrate diffusion constant on the TiO2 surface, showed that the reaction rate constant increased with temperature as the substrate diffusion constant increased with temperature. Chatzitakis et al.52 studied the temperature effect on photocatalytic degradation of chloramphenicol in the temperature range of 3 °C to 57 °C. They observed that the degradation rate increased up to 45 °C, and after that it decreased. They attributed these findings to the fact that after 45 °C, dissolved O2, which is one of the major parameters, disappeared from the system. However, temperature is regarded as a minor parameter for the photocatalytic reaction. Liang et al.88 found that in the range of 4–60 °C, the rate of reaction increased with increase of temperature; they attempted to explain this with the help of activation energy, although they found that in that particular range of temperature the activation energy was constant. Therefore, they88 stated that with increase of temperature the rate of diffusion of substrate molecules on the catalyst surface increased and hence the rate of photocatalysis increased. From the above discussion, it may be concluded that ambient temperature is a favourable condition for photocatalysis.3.7. Factors influencing the performance of nanoparticles
The presence of foreign substances may enhance the activity of nanoparticles. Some research has already been published where different foreign materials were added to the process to enhance the performance of heterogeneous photocatalysis. In 2005, Coleman et al.31 attempted to enhance the performance of TiO2 in the presence of Pt and Ag. They reported that no such effect was observed during the degradation of low concentrations of organic pollutants in the presence of Pt or Ag, whereas in the presence of Pt, the degradation rate of high concentrations of bisphenol A and resorcinol was increased, and thus, they concluded that incorporation of metal ion was substrate specific. The effect of peroxide on the activity of nanoparticles was investigated by Yu et al.40 According to their report, the addition of a very small amount of H2O2 enhanced the rate of degradation of triclosan, and as a result less time was required for its removal. They ascribed this result to the fact that H2O2 was a better electron acceptor than O2 in the studied system. Meanwhile, Calza et al.51 explained the effect of H2O2 as due to the enhanced rate of formation of hydroxyl ion, thereby leading to the improvement of the reaction rate, using the following equations:| H2O2 + hγ → 2OH˙ | (5) |
| H2O2 + O2− → OH˙ + OH− + O2 | (6) |
They also studied the additional effect of a small amount of ferrous (Fe2+) ion along with a lower amount of H2O2. As per their suggestion, this helped to produce hydroxyl radicals, although the excess amount of Fe2+ reduced the performance of the reaction due to the formation of ferric (Fe3+) ions. Chatzitakis et al.52 described the influence of H2O2 as being quite similar to the reason described by Calza et al.51 They also illustrated that hydroxyl radicals enhanced the formation of peroxy compounds with TiO2, which enhanced the reaction performance, although the increase in the reaction rate was limited up to certain concentrations of H2O2. Studies by Hapeshi et al.,61 Achilleos et al.,62 Elmolla and Chaudhuri65,75 also supported the observation by Calza et al.;51 however, higher concentrations of peroxide reduced mineralization rates, as it absorbed oxidizing agent and acted as an electron and radical scavenger. KIO3 has been shown to enhance the performance of photocatalysis,78 as it functioned as a recombinant between hole and electron, which resulted in a higher rate of OH˙ generation. Very recently, Mohammadi et al.79 tested the doping effect of Sn in the TiO2 matrix. They observed modifications in the surface area, band gap energy and crystal structure. Higher surface area, uniform particle size distribution and lower band gap energy ensured higher rates of waste degradation. 2-propanol and KI may also function as OH˙ radical scavengers. The OH˙ radical functions as the main oxidising agent; thus, with the introduction of 2-propanol in the photocatalytic system, the rate of reaction increases.71,88,91 Therefore, it may be concluded that the introduction of a material, which enhances OH˙ radical concentration and Sn doping improves the performance of photocatalysis.
3.8. Influence of dissolved oxygen
Oxygen works as an oxidizing agent; therefore, it plays a significant role in AOP. In 2007, Abellan et al.45 made a successful attempt to identify the necessity of dissolved oxygen in the photocatalytic reaction. They stated that a sufficient amount of oxygen was required for the oxidation process to produce hydroxyl radicals. In the following year, Yang et al.54 tested the effect of O2 concentration, and their finding was quite similar to that of Abellan et al.45 They explained that H2O2 was generated during the reaction with the help of O2. H2O2 played a major role in the formation of hydroxyl radicals and subsequently reduced the effective wavelength for photocatalysis. To ensure the effectiveness of dissolved O2, Mendez-Arriaga et al.53 observed the degradation behaviour of ibuprofen in an excess supply of O2 and ascertained a noticeably high degradation rate with reduced irradiation time. According to Xekoukoulotakis et al.,74 Nasuhoglu et al.80 and Im et al.,83 the presence of O2 could reduce the possibility of recombination between generated electrons and holes, and the excess holes could solely degrade waste molecules; this could lead to the formation of hydroxyl radicals, which enhance the rate of the degradation process. Thus, it is necessary to maintain the desired O2 concentration level in the photocatalytic system.3.9. Influence of flow rate over a continuous system
Numerous researchers have studied the degradation rate of pharmaceutical wastes in batch mode. However, batch study has several disadvantages, i.e. a constant amount of liquid can be handled at a time, the process is time consuming, process parameters cannot be altered during the reaction, and in some cases it is not commercially viable. To alleviate these issues, continuous mode can be a better alternative for downstream treatment. The flowing stream can be easily handled by a continuous reacting system. The most important parameter of such a system is retention time; this must be optimized depending on the desired conversion. The retention time is a measure of how long a reactant particle is retained inside a reactor. It is directly dependent upon the flow rate through the system. Lower flow rate means higher retention time and higher rate of mineralization of pharmaceutical waste. Ho et al.73 first implemented such a mode of operation for the degradation of trimethoprim, an antibiotic, and adjusted the flow rate of the stream to achieve the maximum degradation of the target drug for the continuous system.4. Mode of application of nanoparticles
During photocatalytic degradation, different modes of applications were observed by several groups of researchers, although it was observed that in most cases nanoparticle suspension was used during photocatalysis for pharmaceutical waste degradation.22,27,32–47,49–57,59–76,78–81,83,84,86–94 In suspension mode both nanoparticles and the pharmaceutical compound are placed together into the reactor and stirring is provided to keep the suspension stable under UV irradiation. It is very difficult to recover nanoparticles from the suspension; therefore, TiO2 is lost after the reaction, which is the major limitation of that technique. Thus, immobilization techniques have been adopted by several groups of researchers to enhance the reusability of nanoparticles as well as to make the process more economic.21,22,24,26,28,30,31,48,77,82 Two main types of immobilization have been observed so far; the first is the coating of nanoparticles on a substrate material, and the second is the entrapment of nanoparticles inside a porous structure. All modes of applications of nanoparticles are shown in Fig. 2. To overcome the limitations of suspension mode, Coleman et al.21 implemented the immobilization of TiO2 on Ti-6Al-4V alloy and achieved 98% removal of 17-β-oestradiol within 3.5 h. In 2002, Tanizaki et al.22 implemented the concept of a photobioreactor of high sensitivity where TiO2 nanoparticles were immobilized in a porous quartz structure to increase the available surface area. TiO2 has also been used in immobilized conditions inside a polytetrafluoroethylene mesh sheet.24 Coleman et al.53 used another type of photocatalytic reactor in batch mode where a TiO2 coated reactor surface was used. Doll and Frimmel32 and Ziegmann and Frimmel66 designed a pilot-scale setup of a microfiltration unit to separate TiO2 from the treated solution to enhance the reusability of the nanoparticles. To fulfil the same purpose, Augugliaro et al.35 used a solar membrane photoreactor. In 2006, Molinari et al.41 induced membrane technology to separate byproducts and TiO2 after the photocatalytic reaction in a batch system to make the process cost effective. In 2007, Abellan et al.45 applied a tubular quartz reactor to carry out the photocatalytic reaction and two parabolic mirrors were used to concentrate UV irradiation towards the target solution; however, TiO2 suspension was used during the degradation reaction. For the same purpose, Coleman et al.48 developed a ‘sol–gel spiral reactor’ under both solar and UV irradiation where immobilized and suspended TiO2 were used; the UV radiated system showed the best performance. To enhance the performance of the system, Mendez-Arriaga et al.53 incorporated a stirred tank reactor with integrated O2 supply, temperature and pH indicator, thermostatic control bath, etc. All areas exposed to the catalyst were covered with Teflon or made of glass to enhance the effectiveness of the TiO2. To treat wastewater from the pharmaceutical industry directly, Ziemiańska et al.67 followed conditioning, microfiltration and coagulation methods before photocatalytic degradation to reduce the waste load from the solution matrix. Ziemiańska et al.67 and Laera et al.76 introduced a membrane bioreactor (MBR) along with photocatalysis for wastewater treatment where they maintained a certain recycle ratio to achieve a sufficient amount of drug removal.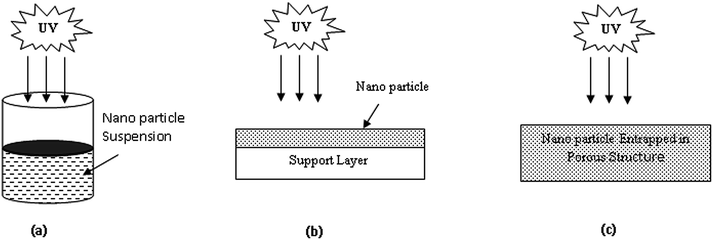 | ||
| Fig. 2 Different modes of application of TiO2 nanoparticles: (a) suspension mode; (b) surface immobilization; (c) entrapped in porous structure. | ||
In 2011, a new technology was adopted by Hu et al.77 to use nanoparticles for pharmaceutical wastewater treatment. They synthesized a TiO2 nanowire membrane on titanium foil. This type of membrane showed more effective degradation than normal P25 and enhanced the reusability of TiO2. A possible explanation was provided by Hu et al.77,81 that the energy distribution over the nanowire membrane surface was better than that of other types of TiO2; thus, it presented an effective photocatalytic effect. Murphy et al.82 compared the effectiveness of TiO2 when it was employed as a suspension as well as in composite form, and they found higher degradation rates for the latter case due to large surface area availability. Moreover, the composite could be reused as its activity was considerably available. Very recently, Avisar et al.84 used “sol–gel N-doped TiO2” which was deposited over microscope glass slides. They found this was more advantageous than powdered TiO2 considering the homogeneous mixing and scattering of light during the reaction; their technique enhanced the reusability of nanoparticles and also made the process more economic. Therefore, reuse and enhancement of the active surface area of TiO2 are important areas of research and development. Only then can photocatalysis be implemented in wastewater treatment for commercial purposes.
5. Kinetics involved in the photocatalytic reaction
The photocatalytic degradation process is basically classified as a heterogeneous catalytic reaction. It has already been discussed in earlier sections that the reaction rate varies significantly with different process parameters such as initial substrate concentration, catalyst loading, pH, dissolved oxygen, etc. The effects of those parameters on photocatalysis can be well described in terms of the reaction rate constant. Therefore, it is very important to understand the reaction kinetics of photocatalytic degradation.Some groups of researchers22–24,27,28,30,31,36,39,41,45,48,50,53,67,73,87 described the degradation kinetics as first order, which was presented as:
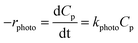 | (7) |
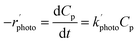 | (8) |
| ln(C0p/Cp) = k′photot | (9) |
The initial concentration of pharmaceutical waste is represented as C0p. The rate constant values were calculated from the slope of the plot of −ln(C0p/Cp) vs. time. However, in recent years, heterogeneous catalytic reaction kinetics have been interpreted using Langmuir–Hinshelwood (L–H) kinetics,55 and since photocatalysis belongs to a similar reaction category, in many cases, the photocatalytic degradations were described by the L–H kinetics model.29,38,46,49,51,52,56,59,63,69,71,72,74,78,79,83 According to the L–H model, the reaction takes place inside the catalyst pore surface where the substrate material is adsorbed through the catalyst pores. The rate is represented as
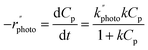 | (10) |
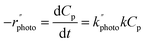 | (11) |
Therefore, three different types of kinetic models can be adopted during the photocatalytic degradation of pharmaceutical wastewater under UV irradiation depending on the experimental observation.
6. Conclusions
Considering the impact of pharmaceutical wastes on the aquatic environment, it is desirable to take necessary action against the alarming threat posed by discharged pharmaceutical waste. The present study has attempted to describe the importance of the photocatalytic degradation process and consequences of the process variables. Some of the major observations have been described here. Among all other treatment methodologies, heterogeneous photocatalysis has already been established as a future trend for the treatment of pharmaceutical waste, and by this process, maximum removal of drug components can be achieved with the production of no secondary pollutants. Several process parameters are involved in AOP using nanoparticles, and these need to be optimized. To make the process more economic as well as more acceptable to researchers and industries, the recycling and reuse of nanoparticles should be investigated. Moreover, optimization of the process parameters and kinetic studies are required for better understanding of the degradation process. In most cases, researchers have used batch processes; very few literature reports are available regarding the continuous oxidation process, though the latter can enhance the performance of the system, making the continuous process more acceptable for large scale wastewater treatment.In most of the reviewed literature, the immense potential of the photocatalytic degradation approach is well accepted but further improvements are still needed for better exploitation by pharmaceutical industries. Above all, the recycling and reuse of the photocatalyst in a continuous wastewater treatment process is the main area which must be developed in future for large scale implementation of heterogeneous photocatalysis. Moreover, the impact of the photocatalyst on the environment has not yet been studied according to the available literature, and that will be another area of exploration. The present article has highlighted advantages as well as limitations of heterogeneous photocatalysis of pharmaceutical wastes and thus, this study may be a valuable addition in further developments in photocatalysis. As a consequence, heterogeneous photocatalysis, with some necessary modification, can be used as a potential alternative to the traditional tedious processes for municipal as well as industrial pharmaceutical wastewater treatment.
Notes and references
- S. Malato, P. Fernandez-Ibanez, M. I. Maldonado, J. Blanco and W. Gernjak, Catal. Today, 2009, 147, 1–59 CrossRef CAS PubMed.
- T. Wintgens, F. Salehi, R. Hochstrat and T. Melin, Water Sci. Technol., 2008, 57, 99–107 CrossRef CAS PubMed.
- S. Suarez, M. Carballa, F. Omil and J. M. Lema, Rev. Environ. Sci. Biotechnol., 2008, 7, 125–138 CrossRef CAS.
- M. Carballa, F. Omil, J. M. Lema, M. Llompart, C. Garcia-Jares, I. Rodriguez, M. Gomez and T. Ternes, Water Res., 2004, 38, 2918–2926 CrossRef CAS PubMed.
- R. Hirsch, T. A. Ternes, K. Haberer, A. Mehlich, F. Ballwanz and K. L. Kratz, J. Chromatogr. A, 1998, 815, 213–223 CrossRef CAS.
- T. A. Ternes, Water Res., 1998, 32, 3245–3260 CrossRef CAS.
- K. A. Kidd, P. J. Blanchfield, K. H. Mills, V. P. Palace, R. E. Evans, J. M. Lazorchak and R. W. Flick, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 8897–8901 CrossRef CAS PubMed.
- R. Lange, T. H. Hutchinson, C. P. Croudace, F. Siegmund, H. Schweinfurth, P. Hampe, G. H. Panter and J. P. Sumpter, Environ. Toxicol. Chem., 2001, 20, 1216–1227 CrossRef CAS.
- J. L. Oaks, M. Gilbert, M. Z. Virani, R. T. Watson, C. U. Meteyer, B. A. Rideout, H. L. Shivaprasad, S. Ahmed, M. J. Chaudhry, M. Arshad, S. Mahmood, A. Ali and A. A. Khan, Nature, 2004, 427, 630–633 CrossRef CAS PubMed.
- W. Viessman Jr and M. J. Hammer, Water Supply and Pollution Control, Addison Wesley Longman Inc, California, USA, 6th edn, 1998 Search PubMed.
- P. V. A. Padmanabhan, K. P. Sreekumar, T. K. Thiyagarajan, R. U. Satpute, K. Bhanumurthy, P. Sengupta, G. K. Dey and K. G. K. Warrier, Vacuum, 2006, 80, 11–12 CrossRef PubMed.
- U. I. Gaya and A. H. Abdullah, J. Photochem. Photobiol., C, 2008, 9, 1–12 CrossRef CAS PubMed.
- H. Yang and H. Cheng, Sep. Purif. Technol., 2007, 56, 392–396 CrossRef CAS PubMed.
- J. Lu, T. Zhang, J. Ma and Z. Chen, J. Hazard. Mater., 2009, 162, 140–145 CrossRef CAS PubMed.
- H. M. Coleman, C. P. Marquis, J. A. Scott, S. S. Chin and R. Amal, Chem. Eng. J., 2005, 113, 55–63 CrossRef CAS PubMed.
- M. Klavarioti, D. Mantzavinos and D. Kassinos, Environ. Int., 2009, 35, 402–417 CrossRef CAS PubMed.
- T. A. Ternes, M. Meisenheimer, D. McDowell, F. Sacher, H. J. Brauch, B. Haist-Gulde, G. Preuss, U. Wilme and N. Zulei-seibert, Environ. Sci. Technol., 2002, 36, 3855–3863 CrossRef CAS.
- W. Hua, E. R. Bennett and J. R. Letcher, Water Res., 2006, 40, 2259–2266 CrossRef CAS PubMed.
- T. Thiruvenkatachari, S. Vigneswaran and S. Moon, Korean J. Chem. Eng., 2008, 25, 64–72 CrossRef PubMed.
- N. Serpone, and E. Pelizzetti, Photocatalysis: Fundamentals and applications, John Wiley & Sons, New York, 1989 Search PubMed.
- H. M. Coleman, B. R. Eggins, J. A. Byrne, F. L. Palmer and E. King, Appl. Catal., B, 2000, 24, L1–L5 CrossRef CAS.
- T. Tanizaki, K. Kadokami and R. Shinohara, Bull. Environ. Contam. Toxicol., 2002, 68, 732–739 CrossRef CAS.
- Y. Ohko, K. I. Iuchi, C. Niwa, T. Tatsuma, T. Nakashima and T. Iguchi, et al., Environ. Sci. Technol., 2002, 36, 4175–4181 CrossRef CAS.
- T. Nakashima, Y. Ohko, Y. Kubota and A. Fujishima, J. Photochem. Photobiol., A, 2003, 160, 115–120 CrossRef CAS.
- T. E. Doll and F. H. Frimmel, Chemosphere, 2003, 52, 1757–1769 CrossRef CAS.
- H. M. Coleman, E. J. Routledge, J. P. Sumpter, B. R. Eggins and J. A. Byrne, Water Res., 2004, 38, 3233–3240 CrossRef CAS PubMed.
- P. Calza, M. Pazzi, C. Medana, C. Baiocchi and E. Pelizzetti, J. Pharm. Biomed. Anal., 2004, 3, 9–19 CrossRef PubMed.
- K. Mitamura, H. Narukawa, T. Mizuguchi and K. Shimada, Anal. Sci., 2004, 20, 3–4 CrossRef CAS.
- T. E. Doll and F. H. Frimmel, Water Res., 2004, 38, 955–964 CrossRef CAS PubMed.
- H. M. Coleman, M. I. Abdullah, B. R. Eggins and F. L. Palmer, Appl. Catal., B, 2005a, 55, 23–30 Search PubMed.
- H. M. Coleman, K. Chiang and R. Amal, Chem. Eng. J., 2005b, 113, 65–72 Search PubMed.
- T. E. Doll and F. H. Frimmel, Water Res., 2005a, 39, 847–854 Search PubMed.
- T. E. Doll and F. H. Frimmel, Water Res., 2005b, 39, 403–411 Search PubMed.
- T. E. Doll and F. H. Frimmel, Catal. Today, 2005c, 101, 195–202 Search PubMed.
- V. Augugliaro, E. Garcĩa-Lŏpez, V. Loddo, S. Malato-Rodrĩguez, I. Maldonado, G. MarcĩRaffaele Molinari and L. Palmisano, Sol. Energy, 2005, 79, 402–408 CrossRef CAS PubMed.
- S. Kaniou, K. Pitarakis, I. Barlagianni and I. Poulios, Chemosphere, 2005, 60, 372–380 CrossRef CAS PubMed.
- T. Malygina, S. Preis and J. Kallas, Int. J. Photoenergy, 2005, 7, 187–191 CrossRef CAS.
- P. Calza, V. A. Sakkas, C. Medana, C. Baiocchi, A. Dimou and E. Pelizzetti, et al., Appl. Catal., B, 2006, 67, 197–205 CrossRef CAS PubMed.
- S. Rafqah, P. Wong-Wah-Chung, S. Nelieu, J. Einhorn and M. Sarakha, Appl. Catal., B, 2006, 66, 119–125 CrossRef CAS PubMed.
- J. C. Yu, T. Y. Kwong, Q. Luo and Z. Cai, Chemosphere, 2006, 65, 390–399 CrossRef CAS PubMed.
- R. Molinari, F. Pirillo, V. Loddo and L. Palmisano, Catal. Today, 2006, 118, 205–213 CrossRef CAS PubMed.
- W. Baran, J. Sochacka and W. Wardas, Chemosphere, 2006, 65, 1295–1299 CrossRef CAS PubMed.
- I. Munõz, J. Peral, J. A. Ayllon, S. Malato, P. Passarinho and X. Domenech, Water Res., 2006, 40, 3533–3540 CrossRef PubMed.
- C. Reyes, J. Fernandez, J. Freer, M. A. Mondaca, C. Zaror and S. Malato, et al., J. Photochem. Photobiol., A, 2006, 184, 141–146 CrossRef CAS PubMed.
- M. N. Abellan, B. Bayarri, J. Gimenez and J. Costa, Appl. Catal., B, 2007, 74, 233–241 CrossRef CAS PubMed.
- L. Hu, P. M. Flanders, P. L. Miller and T. J. Strathmann, Water Res., 2007, 41, 2612–2626 CrossRef CAS PubMed.
- V. A. Sakkas, P. Calza, C. Medana, A. E. Villioti, C. Baiocchi and E. Pelizzetti, et al., Appl. Catal., B, 2007, 77, 135–144 CrossRef CAS PubMed.
- H. M. Coleman, V. Vimonses, G. Leslie and R. Amal, Water Sci. Technol., 2007, 55, 301–306 CrossRef CAS.
- S. Yurdakal, V. Loddo, V. Augugliaro, H. Berber, G. Palmisano and L. Palmisano, Catal. Today, 2007, 129, 9–15 CrossRef CAS PubMed.
- Y. Zhang, J. L. Zhou and B. Ning, Water Res., 2007, 41, 19–26 CrossRef CAS PubMed.
- P. Calza, V. A. Sakkas, A. Villioti, C. Massolino, V. Boti and E. Pelizzetti, et al., Appl. Catal., B, 2008, 84, 379–388 CrossRef CAS PubMed.
- A. Chatzitakis, C. Berberidou, I. Paspaltsis, G. Kyriakou, T. Sklaviadis and I. Poulios, Water Res., 2008, 42, 386–394 CrossRef CAS PubMed.
- F. Mendez-Arriaga, S. Esplugas and J. Gimenez, Water Res., 2008, 42, 585–594 CrossRef CAS PubMed.
- L. Yang, L. E. Yu and M. B. Ray, Water Res., 2008, 42, 3480–3488 CrossRef CAS PubMed.
- K. K. Vasanth, K. Porkodi and F. Rocha, Catal. Commun., 2008, 9, 82–84 CrossRef PubMed.
- H. Son, G. Ko and K. D. Zoh, J. Hazard. Mater., 2009, 166, 954–960 CrossRef CAS PubMed.
- L. Rizzo, S. Mericb, D. Kassinosc, M. Guidab, F. Russob and V. Belgiornoa, Water Res., 2009, 43, 979–988 CrossRef CAS PubMed.
- M. Boroski, A. C. Rodrigues, J. C. Garcia, L. C. Sampaio, J. Nozaki and N. Hioka, J. Hazard. Mater., 2009, 162, 448–454 CrossRef CAS PubMed.
- L. Rizzo, S. Meric, M. Guida, D. Kassinos and V. Belgiorno, Water Res., 2009, 43, 4070–4078 CrossRef CAS PubMed.
- Y. Liu, X. Gan, B. Zhou, B. Xiong, J. Li, C. Dong, J. Bai and W. Cai, J. Hazard. Mater., 2009, 171, 678–683 CrossRef CAS PubMed.
- E. Hapeshi, A. Achilleos, M. I. Vasquez, C. Michael, N. P. Xekoukoulotakis, D. Mantzavinos and D. Kassinos, Water Res., 2010, 44, 1737–1746 CrossRef CAS PubMed.
- A. Achilleos, E. Hapeshi, N. P. Xekoukoulotakis, D. Mantzavinos and D. Fatta-Kassinos, Chem. Eng. J., 2010, 161, 53–59 CrossRef CAS PubMed.
- H. Yanga, G. Li, T. An, Y. Gao and J. Fu, Catal. Today, 2010, 153, 200–207 CrossRef PubMed.
- A. L. Giraldo, G. A. Peňuela, R. A. Torres-Palma, N. J. Pino, R. A. Palominos and H. D. Mansilla, Water Res., 2010, 44, 5158–5167 CrossRef CAS PubMed.
- E. S. Elmolla and M. Chaudhuri, Desalination, 2010, 252, 46–52 CrossRef CAS PubMed.
- M. Ziegmann and F. H. Frimmel, Water Sci. Technol., 2010, 61.1, 273–281 CrossRef PubMed.
- J. Ziemiańska, E. Adamek, A. Sobczak, I. Lipska, A. Makowski and W. Baran, Physicochem. Probl. Miner. Process., 2010, 45, 127–140 Search PubMed.
- J. Zhang, D. Fu, Y. Xu and C. Liu, J. Environ. Sci., 2010, 22(8), 1281–1289 CrossRef CAS.
- S. Basha, D. Keane, A. Morrissey, K. Nolan, M. Oelgemöller and J. Tobin, Ind. Eng. Chem. Res., 2010, 49, 11302–11309 CrossRef CAS.
- W. Alahmad and M. A. Alawi, Jordan J. Pharm. Sci., 2010, 3(2), 126–135 CAS.
- H. Yang, T. An, G. Li, W. Song, W. J. Cooper, H. Luo and X. Guo, J. Hazard. Mater., 2010, 179, 834–839 CrossRef CAS PubMed.
- T. An, J. An, H. Yang, G. Li, H. Feng and X. Nie, J. Hazard. Mater., 2011, 197, 229–236 CrossRef CAS PubMed.
- D. Ho, S. Vigneswaran, H. H. Ngo, H.-K. Shon, J. Kandasamy, C.-Y. Chang and J.-S. Chang, Sustainable Environ. Res., 2011, 21(3), 149–154 CAS.
- N. P. Xekoukoulotakis, C. Drosou, C. Brebou, E. Chatzisymeon, E. Hapeshi, D. Fatta-Kassinos and D. Mantzavinos, Catal. Today, 2011, 161, 163–168 CrossRef CAS PubMed.
- E. S. Elmolla and M. Chaudhuri, Desalination, 2011, 272, 218–224 CrossRef CAS PubMed.
- G. Laera, M. N. Chong, B. Jin and A. Lopez, Bioresour. Technol., 2011, 102, 7012–7015 CrossRef CAS PubMed.
- A. Hu, X. Zhang, K. D. O. Peng Peng, Y. N. Zhou and M. R. Servos, J. Hazard. Mater., 2011, 189, 278–285 CrossRef CAS PubMed.
- M. Chen and W. Chu, J. Hazard. Mater., 2012, 219–220, 183–189 CrossRef CAS PubMed.
- R. Mohammadi, B. Massoumi and M. Rabani, Int. J. Photoenergy, 2012 DOI:10.1155/2012/514856.
- D. Nasuhoglu, A. Rodayan, D. Berk and V. Yargeau, Chem. Eng. J., 2012, 189–190, 41–48 CrossRef CAS PubMed.
- A. Hu, X. Zhang, D. Luong, K. D. Oakes, M. R. Servos, R. Liang, S. Kurdi, P. Peng and Y. Zhou, Waste Biomass Valorization, 2012, 3, 443–449 CrossRef CAS.
- S. Murphy, C. Saurel, A. Morrissey, J. Tobin, M. Oelgemöller and K. Nolana, Appl. Catal., B, 2012, 119–120, 156–165 CrossRef CAS PubMed.
- J.-K. Im, H.-S. Son, Y.-M. Kang and K.-D. Zoh, Water Environ. Res., 2012, 84(7), 554–561 CrossRef CAS.
- D. Avisar, I. Horovitz, L. Lozzi, F. Ruggieri, M. Baker, M.-L. Abel and H. Mamane, J. Hazard. Mater., 2013, 244–245, 463–471 CrossRef CAS PubMed.
- L. G. Bousiakou, K. Mohsin, P. Lianos, A. J. Fatani, E. Kalkani and G. A. Karikas, Pharmakeftiki, 2013, 25(2), 37–48 CAS.
- J. Choina, H. Kosslicka, Ch. Fischer, G.-U. Flechsig, L. Frunza and A. Schulz, Appl. Catal., B, 2013, 129, 589–598 CrossRef CAS PubMed.
- F. Martínez, M. J. Lõpez-Munõz, J. Aguado, J. A. Melero, J. Arsuaga, A. Sotto, R. Molina, Y. Segura, M. I. Pariente, A. Revilla, L. Cerro and G. Carenas, Water Res., 2013, 47, 5647–5658 CrossRef PubMed.
- R. Liang, A. Hu, W. Li and Y. N. Zhou, J. Nanopart. Res., 2013 DOI:10.1007/s11051-013-1990-x.
- W. Buda and B. Czech, Water Sci. Technol., 2013, 68(6), 1322–1328 CrossRef CAS PubMed.
- L. Haroune, M. Salaun, A. Ménard, C. Y. Legault and J.-P. Bellenger, Sci. Total Environ., 2014, 475, 16–22 CrossRef CAS PubMed.
- H. H.-H. Lin and A. Y.-C. Lin, Water Res., 2014, 48, 559–568 CrossRef CAS PubMed.
- R. Das, S. Sarkar, S. Chakraborty, H. Choi and C. Bhattacharjee, Ind. Eng. Chem. Res., 2014, 53(8), 3012–3020 CrossRef CAS.
- S. Sarkar, R. Chowdhury, R. Das, S. Chakraborty, H. Choi and C. Bhattacharjee, RSC Adv., 2014, 4, 21141–21150 RSC.
- R. Das, S. Sarkar and C. Bhattacharjee, Journal of Water Process Engineering, 2014, 2, 79–86 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2014 |
