Capillarity-driven (self-powered) one-dimensional photonic crystals for refractometry and (bio)sensing applications†
Abstract
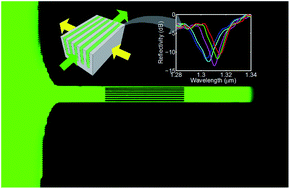
In this work, we advance the state-of-the-art knowledge on photonic crystals by demonstrating the effective and reliable operation of vertical one-dimensional photonic crystals by capillarity, i.e. without the use of external pumps, for self-powered refractometry and label-free (bio)sensing applications. As a proof-of-concept, an all-silicon self-powered drop-and-measure platform exploiting a vertical one-dimensional photonic crystal as a sensing element is fabricated and tested by the capillary infiltration of both ethanol–water mixtures (used as the benchmark for refractometry) and Bovine Serum Albumin (BSA) aqueous solutions at different BSA concentrations (used as the benchmark for biosensing). Excellent analytical performance is achieved for both refractometry and biosensing, in terms of reproducibility and linearity, as well as sensitivity and limit of detection, thus paving the way towards a novel class of self-powered drop-and-measure platforms for chemical/biochemical point-of-care analysis by exploiting the photonic crystals operating under capillary action as label-free transducers.

 Please wait while we load your content...
Please wait while we load your content...