Hollow tungsten carbide/carbon sphere promoted Pt electrocatalyst for efficient methanol oxidation†
Zaoxue Yan*,
Fan Li,
Jimin Xie* and
Xuli Miu
School of Chemistry and Chemical Engineering, Jiangsu University, Zhenjiang, 212013, PR China. E-mail: yanzaoxue@163.com; xiejm391@sohu.com; Fax: +86 11 88791800; Tel: +86 11 88791708
First published on 8th December 2014
Abstract
The surface carbon thickness and particle size of tungsten carbide (WC) are critical to its synergistic effect on noble metal based electrocatalysts. We report the synthesis of hollow tungsten carbide/carbon sphere composites (HWCSs) with a thinner surface carbon layer and smaller particle size. The polystyrene sphere (PS) as a template and P123 as a surfactant favor the dispersion of carbon and WC precursors, leading to HWCSs with a smaller-sized WC and thinner surface carbon. The above factors in turn favor a stable loading of smaller Pt particles on HWCSs, resulting in excellent electrocatalytic activity (due to the synergistic effect) and stability (due to stronger interaction force between WC and Pt) of Pt/HWCS in electrocatalyzing methanol oxidation in acidic media. The present method is imagined to easily prepare other small-sized carbide particles with a thinner surface carbon layer.
1. Introduction
The commercialization of low temperature fuel cells depends on a significant decrease in cost.1–5 Reducing the amount of the noble metal-based electrocatalyst6,7 is an important aim. It is reported that tungsten carbide (WC) has a synergistic effect on noble metal based electrocatalysts both in the anodic alcohol oxidation and cathodic oxygen reduction reaction.8–15 The use level of a noble metal can be significantly reduced to achieve the same or higher activity when WC is added. The literature indicates that the synergistic effect between carbides and noble metals increases with decreasing carbide particle size16,17 and surface carbon thickness.18 On the other hand, the WC particles reported before were large or coated with a thick surface carbon layer,19–22 resulting in a low specific surface area and inferior contact with the loaded noble metals.Considerable efforts have been made to obtain small-sized WC and other early transition metal carbide particles.23–30 Cui et al. prepared WC nanoparticles with a diameter of 15 nm by in situ carbonization with mesoporous silica as a template, in which the template was used to impede the conglomeration of WC.31 Lu et al. prepared WC particles, also with the diameter of about 15 nm, using a microwave heating method, which avoided the growth of WC particles.32 Liang et al. reduced the WC size to 2–5 nm in diameter on the carbon nanotubes with tungsten hexacarbonyl as the tungsten precursor.33 Recently, Fu's and Shen's groups reported the novel synthesis of carbides with a diameter as small as 2–10 nm using an ion-exchange method.34–37 A new formation mechanism of pure-phase WC that was directly transformed from FeWO4 at temperatures below 600 °C was also put forward.38 Although small-sized WC particles have been obtained using various methods, most of the WC were wrapped with large blocks of carbon or graphite substrates,39–42 reducing the contact with the loaded metals.
This paper reports hollow tungsten carbide/carbon sphere composites (HWCSs) with a hollow structure, small size of WC and thin surface carbon, which lead to closer contact and a stronger interaction force between the WC and the loaded Pt particles. Excellent catalytic activity (synergistic effect) and excellent stability (stronger interaction force) were obtained towards methanol electro-oxidation (MOR).
2. Experimental
2.1 Preparation of the HWCS-1
Typically, 1.0 g polystyrene spheres (denoted as PSs), 2.0 g glucose, 1.8 g ammonium metatungstate (AMT, (NH4)6W7O24·6H2O, A.R.) and some poly(ethylene glycol)-block-poly(propylene glycol)-block-poly(ethylene glycol) (denoted as P123) were impregnated in 40 ml of deionized water. The mixture was heated in a 50 ml autoclave at 180 °C for 12 h. The precipitate was washed with an ethanol solution and dried at 90 °C. Subsequently, the dried product was heated in a microwave oven (1000 W) for 160 s to remove the PSs. The product was then heated to 1000 °C for 7 h in a nitrogen atmosphere and treated with a 1.0 mol L−1 HCl solution to remove a small amount of W2C, and the final HWCS-1 composite was obtained. The “-1” in “HWCS-1” means 1 g PSs was used in the experimental process. P123, as a long-chain dispersant, is often used to synthesize porous substances,43,44 and it is used to disperse the WC precursors. The P123 with masses of 0.00, 0.01, 0.03, 0.06, and 0.22 g in HWCS-1 composites correspond to a P123 content of 0.0 wt%, 0.5 wt%, 1.5 wt%, 3.0 wt% and 10.0 wt%, respectively, based on all carbon sources.2.2 Preparation of the HWCS-2
The preparation of HWCS-2 is similar to the preparation of HWCS-1. The difference is that 2 g of PSs and 1.5 wt% of P123 were added. The “-2” in “HWCS-2” means 2 g PSs were used in the preparation process.2.3 Preparation of HCS-1 (hollow carbon spheres)
The preparation of HCS-1 is similar to the preparation of HWCS-1. The difference is that 1 g of PSs, 1.5 wt% of P123 and no AMT were added. The “-1” in “HCS-1” also means that 1 g PSs was used in the preparation process.2.4 Preparation of Pt electrocatalysts
Pt supported on the HWCS-1 (denoted as Pt/HWCS-1), HWCS-2 (denoted as Pt/HWCS-2) or HCS-1 (denoted as Pt/HCS-1) was prepared. Typically, HWCS-1, HWCS-2 or HCS-1 (50 mg) was added to a mixture of H2PtCl6 (containing 50 mg Pt) and 20 ml glycol, and dispersed to a uniform ink in an ultrasonic bath for 30 min. The pH of the mixture was adjusted to 10 with a 2 mol L−1 NaOH/glycol solution and the resulting solution was then placed into a microwave oven (900 W) for heating at 12 s on and 12 s off procedure for 7 times. Then, the mixture was washed with deionized water and dried in vacuum at 60 °C for 4 h. The Pt content in the resulting product was targeted at 50 wt%.2.5 Preparation of electrodes
4 mg of Pt/HWCS-1, Pt/HWCS-2, Pt/HCS-1 or commercial Pt/C (TKK, Japan) was dispersed in 1.95 ml ethanol and 0.05 ml 5 wt% Nafion suspension (DuPont, USA) under ultrasonic agitation to form the electrocatalyst ink. The electrocatalyst ink (0.005 ml) was deposited on a glassy carbon rod (0.25 cm2) and dried at room temperature. The total Pt loadings were controlled at 0.02 mg cm−2.2.6 Electrochemical characterization
The electrochemical measurements were performed in a three-electrode cell on a potentiostat at 30 °C with a mixed solution of 0.5 mol L−1 H2SO4 and 1.0 mol L−1 methanol solution as electrolyte. Platinum foil (1.0 cm2) and a saturated calomel electrode (SCE) were used as the counter and reference electrodes, respectively. Prior to the electrochemical measurements, the electrolytic solution was purged with high-purity N2 for 30 min to remove the dissolved oxygen.2.7 Physical characterization
The morphology of the synthesized materials was characterized by transmission electron microscopy (TEM, JOEP JEM-2010, JEOL Ltd.) operating at 200 kV, and thermal field emission environmental SEM-EDS-EBSD (Quanta 400F, FEI/OXFORD/HKL, Czech/France). The surface and pore structure were determined on a Physical Adsorption Instrument (ASAP 2400, Micrometeritics Co., USA). The crystal structures were determined on an X-ray diffractometer (XRD, D/Max-IIIA, Rigaku Co., Japan, CuK1, λ = 1.54056 Å radiation).3. Results and discussion
Fig. 1a shows the XRD patterns of HWCS-1 (before the HCl treatment) with a P123 content of 0.0 wt%, 0.5 wt%, 1.5 wt%, 3.0 wt%, and 10.0 wt%. The diffraction peaks at 2θ of 31.5°, 35.6° and 48.3° were assigned to the (001), (100) and (101) planes of the WC crystal by comparing the JCPDS cards. The diffraction peaks at 2θ of 34.5°, 38.0°, 39.6°, and 52.3° were assigned to the (100), (002), (101), and (102) planes of the W2C crystal. The WC (101) peak can be used to calculate the crystal size according to the Scherrer eqn (1)
D = Kλ/(β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ) θ)
| (1) |
 | ||
| Fig. 1 (a) XRD patterns of HWCS-1 with different P123 mass content, and (b) the effect of P123 mass content on crystal size of WC. | ||
Fig. 2a shows a SEM image of HWCS-1 before treatment with HCl, which shows white dots dispersed uniformly on black spheres. Fig. 2b presents a TEM image, which shows uniform black dots. These white and black dots were determined by XRD to be almost pure WC (Fig. 1). From the WC particle size distribution (Fig. 2e) measured randomly from 100 particles (Fig. 2b), the average diameter of WC in HWCS-1 was calculated to be 14.3 nm, which is consistent with the XRD result (14.6 nm). For comparison, the morphology of HWCS without P123 before the HCl treatment was also characterized, and its SEM and TEM images are shown in Fig. 2c and its inset, respectively. From the images, serious conglomeration of white or black dots were observed, and the average diameter of the dots was calculated as 24.0 nm (see Fig. 2f). On the other hand, the XRD result (Fig. 1) shows dominant W2C dots. The above results confirm that the larger precursor particles are difficult to transform to WC.
The effect of the amount of PS on the size of WC and surface carbon thickness was also studied. Typically, the amount of PS in the preparation process was doubled and HWCS-2 was obtained, whose TEM image is shown in Fig. 2d. Obviously, the carbon thickness46 in HWCS-2 was less than that in HWCS-1, in other words, the WC in HWCS-2 has more exposure. The WC particle size distribution in HWCS-2 is shown in Fig. 2g, and the corresponding WC average particle size was calculated to be 13.4 nm, which is slightly smaller than that in HWCS-1. The WC particle size is restricted by the carbon layer thickness.
The HWCS-1 and HWCS-2 were further characterized by BET analysis. Fig. S1† shows the N2 adsorption–desorption isotherms and pore size distributions. Table 1 summarizes the surface areas, pore volumes and average pore diameters. HWCS-1 has a total surface area of 433 m2 g−1, total pore volume of 0.25 cm3 g−1 and an average pore diameter of 0.46 nm, and HWCS-2 has a total surface area of 738 m2 g−1, total pore volume of 0.46 cm3 g−1 and an average pore diameter of 0.49 nm. Obviously, HWCS-2 has an improved surface structure than HWCS-1.
| Sample | Total surface area/m2 g−1 | Micropore area/m2 g−1 | Total pore volume/cm3 g−1 | Micropore volume/cm3 g−1 | Average pore diameter/nm |
|---|---|---|---|---|---|
| HWCS-1 | 433 | 361 | 0.25 | 0.17 | 0.46 |
| HWCS-2 | 738 | 598 | 0.46 | 0.30 | 0.49 |
The higher surface area, more exposure (or thinner surface carbon) and smaller-sized WC of HWCSs are expected to give Pt/HWCSs better performance. Pt particles were loaded on the HWCS-1, HWCS-2 and HCS-1 to form electrocatalysts. Fig. 3 shows the XRD patterns of the three electrocatalysts. The peaks at 39.8°, 46.2° and 67.5° 2θ were assigned to the (111), (200) and (220) facets of a Pt crystal by comparing the JCPDS cards. According to Scherrer eqn (1), the Pt particle size in Pt/HCS-1 was calculated to be 3.6 nm. The Pt particle size in Pt/HWCS-1 and Pt/HWCS-2 cannot be accurately calculated because the Pt peaks overlapped with the WC peaks. On the other hand, the FWHMs of the Pt peaks on Pt/HWCS-1 and Pt/HWCS-2 were obviously wider than that on Pt/HCS-1, indicating smaller Pt sizes in Pt/HWCS-1 and Pt/HWCS-2. The reports suggest that stronger interaction force exist between the carbides and noble metals,47,48 which should account for the loading of smaller Pt particles on HWCSs.
Fig. 4a presents a TEM image of Pt/HWCS-1; no conglomeration can be observed. Fig. 4b and c show the Pt and WC crystal lattices in Pt/HWCS-1. Fig. 4d shows sparser and smaller WC and Pt particles in Pt/HWCS-2. Fig. 4e shows conglomerated Pt particles in Pt/HCS-1. All the WC and Pt particle sizes shown in Fig. 4 are consistent with the TEM and XRD results (Fig. 2 and 3). In addition, the EDS patterns (inset in Fig. 4a and d) for Pt/HWCS-1 and Pt/HWCS-2 confirm the coexistence of Pt, C and W (the peaks for Cu and Cr come from the sample bracket).
The improvement in MOR is significant for improving the performance of proton exchange membrane fuel cells. Fig. 5a shows the cyclic voltammograms of MOR on the Pt/HWCS-1, Pt/HWCS-2, Pt/HCS-1 and Pt/C (TKK) electrodes. The peak current density and onset potential for each electrocatalyst are listed in Table 2. In the following sections, we will first compare the performance of Pt/HWCS-1, Pt/HCS-1 and Pt/C, and then compare that of Pt/HWCS-1 and Pt/HWCS-2.
| Electrocatalyst | Pt mass contenta | Peak current density (mA mgPt−1) | Onset potential (V) | EASA (m2 g−1) |
|---|---|---|---|---|
| a The data were determined by inductively coupled plasma-atomic emission spectrometry (ICP). | ||||
| Pt/HWCS-1 | 47.9% | 1517 | 0.25 | 71.9 |
| Pt/HWCS-2 | 48.3% | 1860 | 0.24 | 82.9 |
| Pt/HCS-1 | 47.1% | 926 | 0.39 | 50.1 |
| Pt/C (TKK) | 47.6% | 773 | 0.40 | 44.9 |
First, the Pt/HCS-1 has slightly higher performance than the Pt/C (TKK), which should result from the high specific surface area of HCS-1. In addition, the Pt/HWCS-1 has considerably higher performance than the Pt/HCS-1, which should result from the addition of WC. The higher performance of Pt/HWCS-1 than that of Pt/C (TKK) was attributed to both the high specific surface area of HWCS-1 and the existence of WC in Pt/HWCS-1. The peak current density on Pt/HWCS-1 (1517 mA mgPt−1) is 1.96 times higher than that on Pt/C (TKK) (773 mA mgPt−1), and the onset potential for MOR on Pt/HWCS-1 is negatively shifted by about 150 mV. This is a significant improvement because a direct methanol fuel cell gives an output voltage of less than 0.5 V at a reasonable current density, leading to an expected 30% improvement in electrical efficiency.
The electrochemical active surface areas (EASAs) can be used to analyze the performance of the synthesized materials. Fig. 5b shows the cyclic voltammograms of the four electrodes in a 0.5 mol L−1 H2SO4 solution. Their EASAs can be calculated using the following eqn (2):48
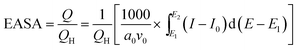 | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, which are obviously smaller than the corresponding ratios of peak current densities (1.96
1, which are obviously smaller than the corresponding ratios of peak current densities (1.96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Therefore, the EASA is not the sole factor determining the performance of the electrocatalysts. There should be a synergistic effect, as mentioned above, to further promote the catalytic activity of Pt/HWCS-1.
1). Therefore, the EASA is not the sole factor determining the performance of the electrocatalysts. There should be a synergistic effect, as mentioned above, to further promote the catalytic activity of Pt/HWCS-1.
Second, we compared the Pt/HWCS-1 and P/HWCS-2 electrocatalysts (Fig. 5). The Pt/HWCS-2 has a peak current density of 1860 mA mgPt−1, which is 1.23 times higher that of Pt/HWCS-1. The EASA of Pt/HWCS-2 (82.9 m2 g−1) is 1.15 times that of Pt/HWCS-1. Moreover, the Pt/HWCS-2 has a more negative onset potential than Pt/HWCS-1. The results indicate that the HWCS-2 with more exposure of WC has a greater promotion effect on Pt than HWCS-1.
The stabilities of the Pt/HWCS-1, Pt/HWCS-2 and Pt/C (TKK) electrodes for MOR are shown in Fig. 5c and d. The shadows are the cycling difference between the 1st cycle and the 5000th cycle. The activity of the commercial Pt/C (TKK) decreased by 21.7% from 767.8 mA mgPt−1 to 601.1 mA mgPt−1 by comparing the peak current density. The activity of the Pt/HWCS-1 decreased by 6.7% from 1517 mA mgPt−1 to 1416 mA mgPt−1, whereas the activity of the Pt/HWCS-2 decreased by 5.1% from 1860 mA mgPt−1 to 1766 mA mgPt−1. The results indicate that WC supported electrocatalysts are more stable than the sole carbon supported electrocatalyst, and the WC with more exposure leads to higher stability of the Pt loaded electrocatalyst.
The excellent activity and stability of Pt/HWCSs are explained by the synergistic effect and strong interaction force between the WC support and the active Pt metal. The literature showed that a synergistic effect exists between carbides and noble metals due to electron transfer between them.48–50 The surface electronic structure of Pt/WC was very different from that of Pt/C, and showed an obvious anti-CO-poisoning effect in the electrooxidation of methanol. This may be the origin of the excellent activity of the Pt/HWCSs. On the other hand, electron transfer between WC and Pt indicates a high linkage (strong interaction force) between them, which accounts for the excellent electrocatalytic stability of Pt/HWCS. Moreover, the greater exposure of WC will lead to closer contact with the loaded Pt particles, and then higher activity and stability can be obtained.
4. Conclusions
HWCSs with more exposure (or thinner surface carbon) and smaller WC particle size were synthesized. The thickness of surface carbon around WC can be reduced by improving the PS to glucose ratio. The WC particle size can be reduced by both increasing the amount of P123 as a dispersant and improving the PS to glucose ratio. The smaller-sized WC particles with more exposure lead to a stable loading of smaller-sized Pt particles and closer contact between them, which not only show excellent activity for catalyzing MOR due to the synergistic effect, but also show greater stability due to the stronger interaction force. The present work can be used to prepare other small-sized particles with a thin surface carbon layer.Acknowledgements
This work was financially supported by China Postdoctoral Science Foundation (2014T70481), National Natural Science Foundation of China (21306067), Natural Science Foundation of Jiangsu (BK20130490), College Natural Science Research Program of Jiangsu Province (12KJB150007) and China Postdoctoral Science Foundation (2013M541609). Jimin Xie thanks the Industry High Technology Foundation of Jiangsu (BE2013090).References
- Y. Dong, H. Pang, H. Yang, J. Jiang, Y. Chi and T. Yu, RSC Adv., 2014, 4, 32791–32795 RSC.
- C. W. Liu, C. M. Lai, J. N. Lin, L. D. Tsai and K. W. Wang, RSC Adv., 2014, 4, 15820–15824 RSC.
- Y. Zhou, X. Hu, Y. Xiao and Q. Shu, Electrochim. Acta, 2013, 111, 588–592 CrossRef CAS PubMed.
- Y. Bing, H. Liu, L. Zhang, D. Ghosh and J. Zhang, Chem. Soc. Rev., 2010, 39, 2184–2202 RSC.
- Q. Wang, X. Wang, Z. Chai and W. Hu, Chem. Soc. Rev., 2013, 42, 8821–8834 RSC.
- M. K. Jeona, K. R. Leea, W. S. Leea, H. Daimonb, A. Nakaharab and S. I. Woo, J. Power Sources, 2008, 185, 927–931 CrossRef PubMed.
- M. Liu, R. Zhang and W. Chen, Chem. Rev., 2014, 114, 5117–5160 CrossRef CAS PubMed.
- Y. W. Lee, A. R. Ko, D. Y. Kim, S. B. Han and K. W. Park, RSC Adv., 2012, 2, 1119–1125 RSC.
- M. D. Obradović, B. M. Babić, V. R. Radmilović, N. V. Krstajić and S. L. Gojković, Int. J. Hydrogen Energy, 2012, 37, 10671–10679 CrossRef PubMed.
- K. Wang, Y. Wang, Z. Liang, Y. Liang, D. Wu, S. Song and P. Tsiakaras, Appl. Catal., B, 2014, 147, 518–525 CrossRef CAS PubMed.
- M. Rahsepar, M. Pakshir, P. Nikolaev, A. Safavi, K. Palanisamy and H. Kim, Appl. Catal., B, 2012, 127, 265–272 CrossRef CAS PubMed.
- N. R. Elezović, B. M. Babić, L. Gajić-Krstajić, P. Ercius, V. R. Radmilović, N. V. Krstajić and L. M. Vračar, Electrochim. Acta, 2012, 69, 239–246 CrossRef PubMed.
- Z. Y. Chen, C. A. Ma, Y. Q. Chu, J. M. Jin, X. Lin, C. Hardacre and W. F. Lin, Chem. Commun., 2013, 49, 11677–11679 RSC.
- C. Ma, L. Kang, M. Shi, X. Lang and Y. Jiang, J. Alloys Compd., 2014, 588, 481–487 CrossRef CAS PubMed.
- T. G. Kelly and J. G. Chen, Chem. Soc. Rev., 2012, 41, 8021–8034 RSC.
- Z. Yan, H. Meng, P. K. Shen, R. Wang, L. Wang, K. Shi and H. Fu, J. Mater. Chem., 2012, 22, 5072–5079 RSC.
- Z. Yan, H. Wang, M. Zhang, Z. Jiang, T. Jiang and J. Xie, Electrochim. Acta, 2013, 95, 218–224 CrossRef CAS PubMed.
- D. V. Esposito, S. T. Hunt, Y. C. Kimmel and J. G. Chen, J. Am. Chem. Soc., 2012, 134, 3025–3033 CrossRef CAS PubMed.
- Y. Wang, S. Song, V. Maragou, P. K. Shen and P. Tsiakaras, Appl. Catal., B, 2009, 89, 223–228 CrossRef CAS PubMed.
- C. Ma, L. Kang, M. Shi, X. Lang and Y. Jiang, J. Alloys Compd., 2014, 588, 481–487 CrossRef CAS PubMed.
- H. Singh and O. P. Pandeyn, Ceram. Int., 2013, 39, 6703–6706 CrossRef CAS PubMed.
- P. K. Shen, S. Yin, Z. Li and C. Chen, Electrochim. Acta, 2010, 55, 7969–7974 CrossRef CAS PubMed.
- S. Yin, M. Cai, C. Wang and P. K. Shen, Energy Environ. Sci., 2011, 4, 558–563 CAS.
- Y. Liu and W. E. Mustain, ACS Catal., 2011, 1, 212–220 CrossRef CAS.
- Y. Yan, B. Xia, X. Qi, H. Wang, R. Xu, J. Y. Wang, H. Zhang and X. Wang, Chem. Commun., 2013, 49, 4884–4886 RSC.
- X. Tao, Y. Li, J. Du, Y. Xia, Y. Yang, H. Huang, Y. Gan, W. Zhang and X. Li, J. Mater. Chem., 2011, 21, 9095–9102 RSC.
- Q. Zhu, S. Zhou, X. Wang and S. Dai, J. Power Sources, 2009, 193, 495–500 CrossRef CAS PubMed.
- A. T. Garcia-Esparza, D. Cha, Y. Ou, J. Kubota, K. Domen and K. Takanabe, ChemSusChem, 2013, 6, 168–181 CrossRef CAS PubMed.
- F. Teng, J. Wang, X. An, B. Lu, Y. Su, C. Gong, P. Zhang, Z. Zhang and E. Xie, RSC Adv., 2012, 2, 7403–7405 RSC.
- K. G. Nishanth, P. Sridhar, S. Pitchumani and A. K. Shukla, Fuel Cells, 2012, 12, 146–152 CrossRef CAS.
- X. Cui, X. Zhou, H. Chen, Z. Hua, H. Wu, Q. He, L. Zhang and J. Shi, Int. J. Hydrogen Energy, 2011, 36, 10513–10521 CrossRef CAS PubMed.
- J. L. Lu, Z. H. Li, S. P. Jiang, P. K. Shen and L. Li, J. Power Sources, 2012, 202, 56–62 CrossRef CAS PubMed.
- C. Liang, L. Ding, A. Wang, Z. Ma, J. Qiu and T. Zhang, Ind. Eng. Chem. Res., 2009, 48, 3244–3248 CrossRef CAS.
- R. Wang, C. Tian, L. Wang, B. Wang, H. Zhang and H. Fu, Chem. Commun., 2009, 3104–3106 RSC.
- G. He, Z. Yan, M. Cai, P. K. Shen, M. R. Gao, H. B. Yao and S. H. Yu, Chem.–Eur. J., 2012, 18, 8490–8497 CrossRef CAS PubMed.
- Z. Hu, Z. Yan, P. K. Shen and C. J. Zhong, Nanotechnology, 2012, 33, 485404 CrossRef PubMed.
- Z. Yan, M. Zhang, J. Xie and P. K. Shen, J. Power Sources, 2013, 243, 336–342 CrossRef CAS PubMed.
- Z. Yan, M. Cai and P. K. Shen, Sci. Rep., 2013, 3, 1646 Search PubMed.
- X. Yang, Y. C. Kimmel, J. Fu, B. E. Koel and J. G. Chen, ACS Catal., 2012, 2, 765–769 CrossRef CAS.
- Y. C. Kimmel, D. V. Esposito, R. W. Birkmire and J. G. Chen, Int. J. Hydrogen Energy, 2012, 37, 3019–3024 CrossRef CAS PubMed.
- T. Ryu, H. Y. Sohn, K. S. Hwang and Z. Z. Fang, J. Am. Ceram. Soc., 2009, 92, 655–660 CrossRef CAS PubMed.
- S. Sharma and B. G. Pollet, J. Power Sources, 2012, 208, 96–119 CrossRef CAS PubMed.
- Z. Fu, Q. M. Huang, X. D. Xiang, Y. L. Lin, W. Wu, S. J. Hu and W. S. Li, Int. J. Hydrogen Energy, 2012, 37, 4704–4709 CrossRef CAS PubMed.
- M. Lei, T. Z. Yang, W. J. Wang, K. Huang, R. Zhang, X. L. Fu, H. J. Yang, Y. G. Wang and W. H. Tang, Int. J. Hydrogen Energy, 2013, 38, 205–211 CrossRef CAS PubMed.
- Y. Wang, S. Song, P. K. Shen, C. X. Guo and C. M. Li, J. Mater. Chem., 2009, 19, 6149–6153 RSC.
- Z. Yan, Z. Hu, C. Chen, H. Meng, P. K. Shen, H. Ji and Y. Meng, J. Power Sources, 2010, 195, 7146–7151 CrossRef CAS PubMed.
- Z. Yan, J. Xie, P. K. Shen, M. Zhang, Y. Zhang and M. Chen, Electrochim. Acta, 2013, 108, 644–650 CrossRef CAS PubMed.
- Z. Yan, G. He, P. K. Shen, Z. Luo, J. Xie and M. Chen, J. Mater. Chem. A, 2014, 2, 4014–4022 CAS.
- R. Wang, Y. Xie, K. Shi, J. Wang, C. Tian, P. Shen and H. Fu, Chem.–Eur. J., 2012, 18, 7443–7451 CrossRef CAS PubMed.
- G. Cui, P. K. Shen, H. Meng, J. Zhao and G. Wu, J. Power Sources, 2011, 196, 6125–6130 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra08184f |
| This journal is © The Royal Society of Chemistry 2015 |




