High performance solid polymer electrolyte with graphene oxide nanosheets†
Abstract
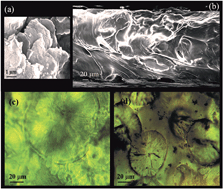
Two dimensional graphene oxide (GO) sheets with high surface area and excellent mechanical properties are introduced into a solid polyethylene oxide/lithium salt electrolyte. Nearly two orders of magnitude improvement in ion conductivity and a 260% increase in the tensile strength of the polymer electrolyte are achieved with only 1 wt% GO content. GO fillers show improved thermo-mechanical stability for the polymer electrolyte and they appear to significantly enhance the performance of the Li ion battery.
- This article is part of the themed collection: Polymers for Electrochemical Energy Storage

 Please wait while we load your content...
Please wait while we load your content...