Droplet detection: simplification and optimization of detecting conditions towards high sensitivity quantitative determination of melamine in milk without any pretreatment†
Xiang Lin,
Wu-Li-Ji Hasi*,
Xiu-Tao Lou,
Shuang Lin,
Fang Yang,
Bao-Shen Jia,
Dian-Yang Lin and
Zhi-Wei Lu*
National Key Laboratory of Science and Technology on Tunable Laser, Harbin Institute of Technology Harbin 150001, China. E-mail: hasiwuliji@126.com; Zhiwei_Lu@sohu.com
First published on 17th September 2014
Abstract
A new droplet configuration was developed to simplify and optimize the SERS detecting condition using silver colloid as SERS substrate. Samples were measured in solution to improve the evenness of SERS response. The influences of the reflectivity of substrates and height of sample solution on Raman intensity have been studied systematically. Aluminum tape was employed as the supporting platform for SERS detection. Based on the high reflectivity and hydrophobicity of aluminum tape, this configuration is both highly efficient and convenient. The detection limit of melamine is 10−2 ppb in water and the distribution of SERS intensity is regular over the droplet profile with good reproducibility. Under this configuration, melamine in milk can be quantitatively detected without any sample pretreatment in a special order of agent addition. A good linear relationship was obtained at concentrations ranging from 0.05 to 10 ppm (R2 = 0.9963). Our scheme has advantages such as simplicity, improved reproducibility, less time and good accuracy over other methods, which offers tremendous potential for the on-site examination of trace melamine in three types of milk (pasteurized milk, skimmed milk, sweet milk).
Introduction
Melamine is a nitrogen-rich (66% by mass) chemical, which is commonly used to produce melamine resin. Unfortunately, since 2007, melamine has been added to wheat gluten, pet food, and various milk products as an adulterant to fake protein in order to reduce production costs by unreliable manufacturers.1 However, over ingestion of melamine can cause renal failure and even death in humans and pets.2 Therefore, the identification and detection of melamine is of vital importance for human health. In order to detect melamine in food matrixes, such as wheat gluten and milk, various methods have been reported, including gas chromatography (GC), liquid chromatography/mass spectrometry (LC/MS), high performance liquid chromatography coupled to ultraviolet detection (HPLC-UV), enzyme linked immunosorbent assays (ELISA),3 matrix-assisted laser desorption ionization (MALDI)4 and colorimetric assays using gold (Au) and silver (Ag) nanoparticles (NPs).5,6 However, some of these methods require complex instruments and long measurement time; some methods require extensive sample preparation and data analysis; and some methods suffer from interferences caused by other analytes. Thus, a rapid method with high sensitivity and specificity for real-time detection of melamine in milk is urgently needed.As an emerging detection technology, SERS technique has been employed to detect melamine in milk using various SERS substrates such as gold/silver colloid,7,8 Ag-nanoparticle-modified single Ag nanowire,9 gold nanofinger10 and cyclodextrin-decorated silver nanoparticles.11 Among them, metal colloid requires an easy preparation method and simple sample preparation (needing only mixing of the sample with colloid). Recently, metal colloids are widely used in trace detection of target analytes,12–14 since using aggregated silver nanoparticles as SERS substrate has allowed trace detection down to the single molecule level.15 However, because metal colloid is a kind of liquid substrate, it is easily affected by environmental conditions such as pH,16 size,17 shape,18 aggregating agents species19–21 and surface charge of nanoparticles and analytes.22 Conversely, these drawbacks provide potential for improving SERS enhancement when dealing with a special condition. The routine approach for SERS detection using a metal colloid is dropping the mixture of sample and colloid on silicon or quartz wafers and waiting for the droplet to dry, which is time-consuming and has poor uniformity. Recently, M. Culha et al. proposed a suspended configuration from a hydrophobic surface to improve testing uniformity and sensitivity;23,24 however, this method also requires the sample to be dried.
Herein, we proposed a simple SERS measurement method by directly detecting the sample droplet in solution, which overcomes both disadvantages of the poor uniformity and time-consuming sample pretreatment. After further optimizing the detection conditions, a 10 ppt (part per trillion) level of detection of melamine in water was achieved under this configuration. Using a particular order of reagent addition, combined with an internal standard method, our setup is capable to quantitatively detecting trace melamine (0.05–10 ppm) in milk without any sample pretreatment.
Experimental section
2.1 Reagents
Silver nitrate (AgNO3) and sodium citrate were obtained from Sinopharm Chemical Reagent Co., Ltd. trichloroacetic acid (TCA) was obtained from Tianjin Kemiou Chemical Reagent Co., Ltd., sodium chloride (NaCl) was obtained from Xilong Chemical Co., Ltd., deionized water was used for all procedures, and milk was purchased from a local supermarket.2.2 Sample preparation
Firstly, melamine standard stock solution (1 × 103 ppm) was prepared. A series of standard melamine solutions of different concentrations were prepared by diluting the stock solution with water. The solutions of milk spiked with 10, 5, 2.5, 1, 0.5, 0.25, 0.1 and 0.05 ppm melamine were prepared by adding 1 mL of each standard melamine solution to 9 mL of milk. 1 mL of deionized water was added to 9 mL of milk as a blank control.2.3 SERS measurement
Raman spectra were collected with a portable compact laser Raman Spectrometer BWS415-785H (B&W Tek, Inc.). The excitation wavelength of the laser is 785 nm. The spectrometer provides a Raman spectrum over the range of 68 to 3200 cm−1 with a spectral resolution better than 3 cm−1. The SERS detections were conducted using 5 s exposure time and a laser power of 150 mW. The typical concentration of melamine for SERS measurements was 100 ppb unless otherwise stated. Boxcar averaging was used to smooth the raw spectrum, and a baseline correction routine was performed to obtain the final spectrum with the background subtracted.2.4 Preparation and characterization of silver colloid
Silver colloid was prepared according to the method of Lee and Meisel.25 Briefly, 45 mg of silver nitrate was added to 250 mL of deionized water, which was then brought to a boil in a flask under vigorous stirring. Sodium citrate (5 mL, 1%) was added, and the solution was kept boiling for 1 hour. Finally, the solution turned greenish brown, which indicated the formation of silver colloid, at which time it was cooled passively. The morphology of silver colloid was observed by a Hitachi SU8010 field emission scanning electron microscope (SEM) at an accelerating voltage of 15 kV (Fig. S1†).Results and discussion
When metal colloid is used, most of time, a droplet of the mixture of colloid and sample is spotted onto a surface followed by drying of the droplet, which is time-consuming. Moreover, a “coffee-ring” phenomenon generated during the drying process would cause an uneven SERS response within the dried droplet area.24,26 In this study, detection of sample in solution was used to save detection time and improve the reproducibility of Raman signal. The influence of different detection configurations on SERS detection sensitivity was studied first. To date, various materials, such as colorimetric cells,11 glass slides27 and silicon wafers,28 have been used to load sample when using metal colloid as SERS substrate. Here, Raman responses of samples loaded on colorimetric cells, quartz wafers, aluminum tape and silicon wafers were compared. Melamine was used as Raman probe because of its high Raman activity and significance in food safety. The strongest Raman peak at 701 cm−1, which is assigned to the in-plane deformation vibration mode,29 is used for melamine identification. When using a colorimetric cell as sample container, the measurement was performed under configuration 1 shown in Fig. 1(a), which requires 2 mL of sample, whereas for the other three substrates, the measurement was performed under configuration 2, in which only 10 μL of sample is needed to drop on substrates without further drying. | ||
| Fig. 1 (a) Two configurations to perform SERS measurement; (b) SERS spectra of sample droplets on different substrates. | ||
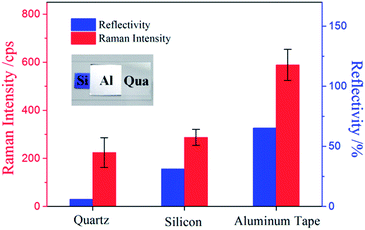
SERS spectra of sample droplets using the above configurations were shown in Fig. 1(b), in which aluminum tape exhibits the strongest SERS intensity while quartz wafer demonstrates the lowest. Silicon wafer has a strong Raman peak at 523 cm−1 and a strong fluorescence background in the range from 1500 cm−1 to 3000 cm−1, which would cause distortion and interference to SERS detection. The colorimetric cell, quartz wafer and silicon wafer all exhibit poorer SERS performance than aluminum tape. The different Raman responses using different supporting platforms can be attributed to two parameters: reflectivity30 of the surface and extinction on propagation through the sample.31
To investigate the effect of reflectivity of surface on SERS response, sample droplets were spotted on three different surfaces with the same droplet height. As shown in Fig. 2, the Raman intensity is positively associated with the reflectivity of surface.
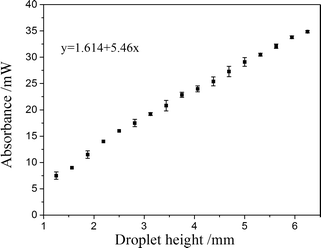
Then, the influence of droplet height on laser extinction was demonstrated in Fig. 3. With increase in sample height, the extinction of Raman signal also increases monotonously and linearly. Therefore, we anticipated that the collected Raman scattering decreases as the droplet height increases.
In view of this, droplet height may directly impact the SERS response. We gradually added 100 ppb melamine solution in a home-made vertical cylindrical vessels using aluminium tape as the bottom. The height was changing from 1.2 mm to 4.7 mm with a step size of about 0.3 mm. As shown in Fig. 4, when the sample height increases, the Raman intensity increases first, and then decreases after reaching the maximum value. This changing trend can be attributed to the change of the excitation and extinction of Raman scattering light through the sample droplet. When the sample height is low, few of the melamine molecules could be irradiated and excited by the incident laser. In this case, even though there is almost no extinction of Raman scattering light caused by the melamine solution, the Raman signal is weak as the sample amount cannot make full use of the incident laser. As sample height increases, the amount of melamine molecules excited by the incident laser also increases. In this case, the excitation of Raman signals grows faster than the extinction; therefore, the detected Raman intensity increases. However, with continued increase of sample height, the excited Raman signals stop increasing as the amount of sample molecules is enough to take full use of the incident laser. On the other hand, as shown in Fig. 3, the loss caused by the solution absorption still increases linearly. Therefore, the Raman intensity gradually decreases after reaching the maximum value. In addition, we have to change the melamine concentration (within the range of 10 ppb to 1 ppm) to verify if the optimal sample height is dependent on the concentration of melamine solution (Fig. S2†). The result shows that the changing trend of Raman intensity with sample height does not rely on the concentration of melamine solution, which is conducive to the quantitative detection of melamine in a wide concentration range. Therefore, the hydrophobicity of supporting platform, which determines the height of a droplet with a certain volume, is the key property of the supporting platform when using the configuration that a sample droplet is free dropped on a surface. As shown in Fig. 4(b), aluminum tape has the maximum contact angle, which is suitable for the SERS detection of a droplet sample; hence, we used aluminium tape as a supporting platform to load the sample.
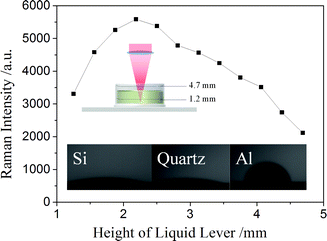 | ||
| Fig. 4 Raman intensity of a melamine solution droplet at different droplet heights. Inset: Hydrophobicity of silicon wafer, quartz wafer and aluminum tape. | ||
To evaluate the uniformity of the Raman intensity acquired from a free droplet sample, the laser probe was scanned through the droplet profile. As shown in Fig. 5(a), the distribution of Raman intensity is central symmetric and is more regular than that of a dried droplet. The effect of focus position on SERS intensity was also demonstrated in Fig. 5(b). The position when the focus of laser beam at the surface of aluminum tape was set as coordinate origin. The result shows that the highest SERS intensity is achieved when the incident laser is exactly focussed on the surface of the aluminum tape.
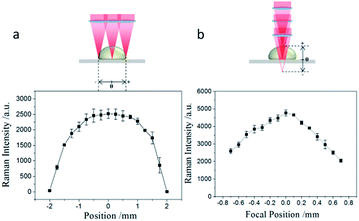 | ||
| Fig. 5 (a) Raman intensity collected along sample droplet at different positions. (b) The effect of focus position on the intensity of Raman scattering light. | ||
It is well known that the SERS performance of metal colloid strongly depends on the volume ratio of sample solution to Ag colloid, the concentration of Cl− and the pH of the detection solution.32–34 After optimizing the detection conditions, see ESI,† 40 μL of melamine solution, 160 μL of silver colloidal solution, 50 μL of 4 mol L−1 NaCl solution, 50 μL of 4 mol L−1 NaOH solution, and 10 μL of mixing solution was used to detect melamine in water. As shown in Fig. 6, the detection limit of melamine in water could reach 0.01 ppb. This provides a feasible way for SERS detection of trace analytes.
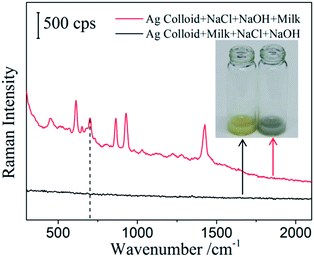
Detection of melamine in milk using SERS technology always requires protein precipitation and centrifugation of the milk sample, which involves the use of toxic organic reagents and equipment. Furthermore, the need of pretreatment makes fast in situ SERS detection of melamine in real samples a mirage. Here, we demonstrate a simple method to detect melamine in milk without any pretreatment using the configuration proposed above. Firstly, we investigate the effect of order of addition of sample on the SERS performance and the result was shown in Fig. 7. One protocol (red line) is mixing silver colloid and aggregating agents first, followed by adding milk. The other protocol (black line) is mixing silver colloid and milk sample first, followed by adding the aggregating agents. If milk is added first, the mixture appears yellow and almost no Raman peaks appear. If aggregating agents are added first, the mixture appears emerald green and many Raman peaks of milk arise. Milk contains large molecular weight species such as various proteins and fat, and many other low molecular weight species such as vitamins and other minerals. Ag NPs would adsorb on these ingredients and thus hinder the aggregation of Ag NPs, which is the precondition of SERS hotspot generation. In addition, some species of milk could compete with the melamine molecules for adsorption in hot spots between Ag NPs. Furthermore, melamine may interact with proteins in milk to form complexes which also hinder melamine molecules trapped by SERS hotspots. Adding the milk sample first will prevent the formation of SERS hot spots which is essential to the Raman enhancement.10,27 On the contrary, adding aggregating agents first will effectively form silver nanoparticle aggregates that mainly consist of SERS hot spots, which in turn amplifies the Raman signals of milk.
From Fig. 7, it is worth noting that there is also a Raman peak at 701 cm−1 in the spectrum of blank milk sample, in addition to several other Raman peaks at 614 cm−1, 652 cm−1, 865 cm−1, 929 cm−1, 1424 cm−1 and 1459 cm−1. These Raman fingerprint peaks can be assigned to other ingredients in milk and have been observed in a previous study.7 These characteristic peaks can be used as internal standards for quantitative determination of melamine in milk; thus, the variation caused by environments can be ignored. To select the most reliable characteristic peak as internal standard, 15 Raman spectra of a blank milk sample were collected and the ratio of Raman intensity of four apparent Raman peaks (614 cm−1, 865 cm−1, 929 cm−1 and 1424 cm−1) to that of 701 cm−1 were calculated. The Raman spectra and calculated results were shown in Fig. 8(a) and (b), respectively. The results indicate that the variation of the ratio of Raman intensity at 929 cm−1 to that of 701 cm−1 is minimal with the RSD of 4.75%. Therefore, the characteristic peak at 925 cm−1 was selected as the internal standard to quantitatively detect melamine in milk. Experimental errors caused by measurement conditions and other factors can be avoided by using this internal standard method.
 | ||
| Fig. 8 (a) SERS spectra of a blank milk sample; (b) I701/IRaman peak ratio for four different Raman peaks of blank milk. | ||
To verify the feasibility of detecting melamine in milk without any pretreatment using this method, milk samples spiked with melamine at different concentrations were directly tested and the SERS spectra were shown in Fig. 9(a). A calibration curve for melamine in milk was shown in Fig. 9(b). It was found that the normalized Raman signal intensity of milk sample increases linearly with the increasing concentrations of the melamine. The limit of quantification (LOQ) using this approach can reach 0.05 ppm with a relative standard deviation (RSD) of 7.49% (n = 9). From the correlation plot of SERS intensity against the concentration of melamine, the linearity range is determined to be between 0.05 ppm and 10 ppm with a correlation coefficient of 0.9963. This method not only simplified the testing process but also displayed a comparable sensitivity for melamine sensing with previous SERS methods.
 | ||
| Fig. 9 (a) SERS spectra of melamine spiked milk sample with concentrations ranging from 0 to 10 ppm; (b) calibration curve for melamine spiked milk samples. | ||
Recovery experiments were carried out by spiking two milk samples with a standard melamine solution of 0.1, 0.5 and 1.0 ppm, and the results of analysis are shown in Table 1. As seen from Table 1, the recoveries of melamine are in the range of 99.8–108.9% with RSDs between 0.67% and 1.9%.
| Sample | Spiked (ppm) | Found (ppm) | RSD (%) | Recovery (%) |
|---|---|---|---|---|
| A | 1 | 1.085 | 1.9 | 108.5 |
| 0.5 | 0.5375 | 1.9 | 107.5 | |
| 0.1 | 0.09988 | 0.67 | 99.8 | |
| B | 1 | 1.081 | 0.9 | 108.1 |
| 0.5 | 0.5445 | 1.49 | 108.9 | |
| 0.1 | 0.10067 | 1.41 | 100.7 |
To evaluate the applicability of the proposed method for detecting melamine in other kinds of milk, a characterization in the variation of the 701/929 intensity ratio for three types of milk (pasteurized milk, skimmed milk, sweet milk) has been conducted (see ESI†). The 701/929 intensity ratio for three types of milk are almost at the same level. this indicates that the proposed method is applicable for detecting melamine in various kinds of liquid milk.
Conclusions
A new droplet detection configuration was developed to simplify and optimize the detection conditions using silver colloid as a SERS substrate. Aluminum tape, a hydrophobic material, was employed as the supporting platform and the influences of reflectivity of substrates and height of sample to a Raman signal have been studied systematically. The SERS response distributes regularly over the droplet area and represents good reproducibility compared with the dried droplet configurations. Based on this configuration, in a particular order of agent addition, melamine in milk was quantitatively detected without any sample pretreatment. Quantitative detection was realized through an internal standard method and the LOQ can reach 0.05 ppm. The linear coefficient is 0.9963 at a concentration range from 0.05 to 10 ppm. Therefore, this configuration based on aluminum tape and a particular order of agent addition provides a robust and versatile approach for the determination of melamine in various milk samples without the need of pretreatment.Acknowledgements
The work was supported by the International S&T Cooperation Program of China (Grant no. 2011DFA31770).Notes and references
- C. W. Chan, E. Y. Y. Chan and S. M. Griffiths, Lancet, 2008, 372, 1444–1445 CrossRef.
- R. L. M. Dobson, S. Motlagh, M. Quijano, R. T. Cambron, T. R. Baker, A. M. Pullen, B. T. Regg, A. S. Bigalow-Kern, T. Vennard, A. Fix, R. Reimschuessel, G. Overmann, Y. Shan and G. P. Daston, Toxicol. Sci., 2008, 106, 251–262 CrossRef CAS PubMed.
- P. Lutter, M.-C. Savoy-Perroud, E. Campos-Gimenez, L. Meyer, T. Goldmann, M.-C. Bertholet, P. Mottier, A. Desmarchelier, F. Monard, C. Perrin, F. Robert and T. Delatour, Food Control, 2011, 22, 903–913 CrossRef CAS.
- J. a. Campbell, D. S. Wunschel and C. E. Petersen, Anal. Lett., 2007, 40, 3107–3118 CrossRef CAS.
- K. Ai, Y. Liu and L. Lu, J. Am. Chem. Soc., 2009, 131, 9496–9497 CrossRef CAS PubMed.
- C. Han and H. Li, Analyst, 2010, 135, 583–588 RSC.
- X.-F. Zhang, M.-Q. Zou, X.-H. Qi, F. Liu, X.-H. Zhu and B.-H. Zhao, J. Raman Spectrosc., 2010, 41, 1655–1660 CrossRef.
- L. C. Mecker, K. M. Tyner, J. F. Kauffman, S. Arzhantsev, D. J. Mans and C. M. Gryniewicz-Ruzicka, Anal. Chim. Acta, 2012, 733, 48–55 CrossRef CAS PubMed.
- L.-M. Chen and Y.-N. Liu, J. Raman Spectrosc., 2012, 43, 986–991 CrossRef CAS.
- A. Kim, S. J. Barcelo, R. S. Williams and Z. Li, Anal. Chem., 2012, 84, 9303–9309 CAS.
- P. Ma, F. Liang, Y. Sun, Y. Jin, Y. Chen, X. Wang, H. Zhang, D. Gao and D. Song, Microchim. Acta, 2013, 180, 1173–1180 CrossRef CAS.
- E. Kämmer, T. Dörfer, A. Csáki, W. Schumacher, P. A. Da Costa Filho, N. Tarcea, W. Fritzsche, P. Rösch, M. Schmitt and J. Popp, J. Phys. Chem. C. Nanomater. Interfaces, 2012, 116, 6083–6091 CrossRef PubMed.
- C. Lofrumento, M. Ricci, E. Platania, M. Becucci and E. Castellucci, J. Raman Spectrosc., 2013, 44, 47–54 CrossRef CAS.
- C. M. Muntean, N. Leopold, A. Halmagyi and S. Valimareanu, J. Raman Spectrosc., 2013, 44, 817–822 CrossRef CAS.
- S. Nie, Science, 1997, 275, 1102–1106 CrossRef CAS PubMed.
- M. Kim and K. Itoh, J. Phys. Chem., 1987, 91, 126–131 CrossRef CAS.
- S. R. Emory, W. E. Haskins and S. Nie, J. Am. Chem. Soc., 1998, 7863, 8009–8010 CrossRef.
- J. Zhang, X. Li, X. Sun and Y. Li, J. Phys. Chem. B, 2005, 109, 12544–12548 CrossRef CAS PubMed.
- S. Han, S. Hong and X. Li, J. Colloid Interface Sci., 2013, 410, 74–80 CrossRef CAS PubMed.
- E. L. Doctor and B. McCord, Analyst, 2013, 138, 5926–5932 RSC.
- X. Lin, W.-L.-J. Hasi, X.-T. Lou, S. Lin, F. Yang, B.-S. Jia, Y. Cui, D.-X. Ba, D.-Y. Lin and Z.-W. Lu, J. Raman Spectrosc., 2014, 45, 162–167 CrossRef CAS.
- R. A. Alvarez-Puebla, E. Arceo, P. J. G. Goulet, J. J. Garrido and R. F. Aroca, J. Phys. Chem. B, 2005, 109, 3787–3792 CrossRef CAS PubMed.
- S. Keskin and M. Culha, Analyst, 2012, 137, 2651–2657 RSC.
- E. Avci and M. Culha, RSC Adv., 2013, 3, 17829 RSC.
- C. Lee and D. Meisel, J. Phys. Chem., 1982, 60439, 3391–3395 CrossRef.
- J. Xu, J. Du, C. Jing, Y. Zhang and J. Cui, ACS Appl. Mater. Interfaces, 2014, 6, 6891–6897 CAS.
- Y. Xie, X. Zhu, Y. Sun, H. Wang, H. Qian and W. Yao, Eur. Food Res. Technol., 2012, 235, 555–561 CrossRef CAS.
- L.-L. Qu, Y.-T. Li, D.-W. Li, J.-Q. Xue, J. S. Fossey and Y.-T. Long, Analyst, 2013, 138, 1523–1528 RSC.
- Y. Chenga, Y. Donga, J. Wub, X. Yanga, H. Bai, H. Zhenga, D. Rena, Y. Zoua and M. Li, J. Food Compos. Anal., 2010, 23, 199–202 CrossRef.
- H. Li, Y. Gu, H. Guo, X. Wang, Y. Liu, W. Xu and S. Xu, J. Phys. Chem. C, 2012, 116, 23608–23615 CAS.
- S. Sivapalan, B. DeVetter and T. Yang, ACS Nano, 2013, 3, 2099–2105 CrossRef PubMed.
- A. M. Michaels and L. Brus, J. Phys. Chem. B, 2000, 104, 11965–11971 CrossRef CAS.
- W. Hill, B. Wehling, A. Spektroskopie and P. O. Box, J. Phys. Chem., 1993, 97, 9451–9455 CrossRef CAS.
- P. Cao, J. Yao, B. Ren, R. Gu and Z. Tian, J. Phys. Chem. B, 2002, 106, 7283–7285 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra07764d |
| This journal is © The Royal Society of Chemistry 2014 |