Polymer electrolyte membranes from Cloisite 30B-based solid proton conductor and sulfonated polyether ether ketone/polyvinylidene fluoride-co-hexafluoro propylene blends for direct methanol fuel cells
M. Prasada,
Smita Mohantyab and
Sanjay K. Nayak*ab
aCentral Institute of Plastics Engineering & Technology (CIPET), Chennai, India. E-mail: papers.journal@gmail.com; Fax: +91 674 2740463; Tel: +91 674 2742852
bLaboratory for Advanced Research in Polymeric Materials (LARPM), Central Institute of Plastics Engineering and Technology, Bhubaneswar-751024, Odisha, India
First published on 30th October 2014
Abstract
Polymer electrolyte membranes (PEMs) were developed for direct methanol fuel cell (DMFC) applications, based on sulfonated polyether ether ketone (SPEEK) and polyvinylidene fluoride-co-hexafluoropropylene (PVDF-HFP) filled with Cloisite 30B (C 30B) as a solid hydrophilic proton conductor. PEMs were characterized using liquid uptake, oxidative stability, TGA, XRD, SEM, AFM and impedance analysis. The single cell DMFC performance reveals that the optimized PEM showed an open circuit voltage (OCV) of 0.79 V and a maximum power density value of 55 mW cm−2 with a proton conductivity value in the range of 10−2 S cm−1. The methanol permeability and selectivity ratio of the optimized membrane were 1.35 × 10−7 cm2 s−1 and 9.63 × 104 S s cm−3, respectively. As a result fabricated membranes could be seen as promising PEMs for DMFC applications.
1. Introduction
Among the various types of fuel cells, direct methanol fuel cells (DMFCs) are the best choice because of their easy handling and high energy density at very low operating temperatures.1 Polymer electrolyte membranes (PEMs), a major component used in DMFCs, are still Nafion® based perfluorinated membranes. The Nafion® membrane shows high proton conductivity at room temperature, as well as good mechanical and chemical stability. However, decreased proton conductivity at low humidity, high cost and high methanol fuel permeability are the main technical hitches for using these in DMFCs.2–5Sulfonated polyether ether ketone (SPEEK) is a promising alternative for PEMs, as these acquire good thermal and oxidative stability, good film forming properties and high proton conductivity. Conversely, there are disadvantages, such as higher swelling degree at high degrees of sulfonation, which leads to dimensional unsteadiness, methanol fuel permeability from anode to cathode during the operation of DMFCs and lower oxidative stability. Additionally, SPEEK membranes are known to have poor compatibility with the electrode-containing Nafion® binder and this results in difficulties during the membrane electrode assembly (MEA) preparation.6 Hence filled counterparts of these sulfonated polymers, such as nanocomposites or blend nanocomposites of SPEEK, have been reported to be used for a wide array of fuel cell applications under variable conditions to achieve improved DMFC performances.7–16
The addition of hygroscopic solid inorganic proton conductors such as nanoclays to the polymeric matrix enhances its ability to maintain sufficient humidity inside the membranes, and thus improves proton conductivity.12–16 Moreover, by the choice of suitable hydrophilic nanoclays, the possibility of a decrease in methanol permeability by means of the formation of tortuous pathways occurs, and this improves the performances of PEMs in DMFCs.11,15 In this regard, it seems possible to improve the performance of PEMs by choosing suitable nanofillers.
Further, to improve the dimensional stability and compatibility of SPEEK membranes to the Nafion® electrode binder solution, a very cost-effective blending technique with a hydrophobic polymer gives better control in swelling degree and dimensional stability without much loss of proton conductivity.17–24 Song et al.17 prepared a cost-effective blend nanocomposite membrane of SPEEK with PVDF and discussed the effect of PVDF content on water uptake, swelling ratio, and proton conductivity. Also, Jung et al.18 have developed a blend membrane with partially sulfonated polystyrene (SPS) and partially sulfonated poly(2,6-dimethyl-1,4-phenylene oxide) (SPPO), which exhibits relatively higher proton conductivity and methanol permeability than single-component SPS and SPPO membranes. Inan et al.22 fabricated a SPEEK/PVDF blend and have observed from their study that the properties of fuel cell blend membranes depend not only on finding suitable polymer combinations but also on the molecular weights of the polymers.
In this current study, we prepared blend nanocomposite PEMs, in which hydrophilic C 30B nanoclays are dispersed in a SPEEK/polyvinylidene fluoride-co-hexafluoro propylene (PVDF-HFP) blend system. We studied the capability of the prepared PEMs for DMFC applications by evaluating their liquid uptakes, proton conductivities, oxidative stabilities and thermal stabilities. The electrochemical performance of the superior membrane was investigated in the DMFC setup.
2. Experimental section
2.1. Materials
Victrex PEEK (grade 450 G), was supplied from Victrex PLC (England) and used as the base polymer. Kynar flex 2801, PVDF-HFP (11–12% hexafluoropropylene copolymer) was supplied by Arkema, India. Organically modified nanoclay C 30B, with a cation exchange capacity (CEC) of 90 meq. per 100 g, was procured from M/s Southern Clay Products. Conc. H2SO4 (98% pure), used as a sulfonating agent, was supplied by Merck (lab grade). The catalysts, Pt black for the cathode and Pt/Ru black for the anode, were purchased from Johnson-Matthey, USA. Nafion® 5 wt% binder solution from E.I. DuPont Company was used for MEA preparation. Nafion® 117 membrane, from E.I. DuPont Company, was chosen as the commercial sample to compare with the obtained results.2.2. Sulfonation of PEEK
The sulfonation of PEEK was carried out using conc. H2SO4 according to the procedure described elsewhere.12,16 In brief; 10 g of predried PEEK polymer was gradually added into 350 mL of vigorously stirred conc. H2SO4 at room temperature. After the desired reaction time of 70 h, the sulfonated PEEK (SPEEK) was recovered by precipitating it in excess ice cold water. Then the precipitate was repeatedly washed with deionised water until it reached pH 6–7. After neutralizing, the SPEEK precipitate was kept in a vacuum oven at 100 °C until it was completely dried. The obtained SPEEK, with a degree of sulfonation of 67%, was used for further experiments. The degree of sulfonation (DS) was determined via volumetric titration using standard sodium hydroxide (NaOH) solution with phenolphthalein as an indicator, using eqn (1).25 In this study, SPEEK with DS = 67% was used as a base matrix for membrane preparation.
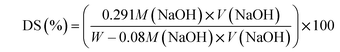 | (1) |
2.3. Preparation of blend nanocomposite membranes
The SPEEK blend nanocomposite membranes were prepared by a solution casting method according to scheduled formulation (Table 1). Dried SPEEK polymer was dissolved in DMAc for 6 h and then a further 5 wt% of C 30B was added to the SPEEK solution, and this was stirred for 3 h in an ultrasonic bath. Subsequently, different wt% of PVDF-HFP powder were added to the SPEEK/C 30B solution, and this was stirred for about 6 h followed by ultrasonication for 2 h. The resulting solution was casted onto a glass plate, dried under vacuum at 80 °C for 12 h, and peeled off from the glass plate.| Membrane code | Mass ratio SPEEK![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PVDF-HFP PVDF-HFP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C 30B C 30B |
Oxidative stability test (wt%) | Proton conductivity × 10−2 (S cm−1)a | Methanol permeability × 10−7 (cm2 s−1) | Selectivity × 104 (S s cm−3)b | |
|---|---|---|---|---|---|---|
| 25 °C | 70 °C | |||||
| a These results are mean values of at least three trials for each sample.b Proton conductivity to methanol permeability ratio. | ||||||
| SPEEK | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
92.1 | 3.18 | 4.98 | 8.61 | 4.15 |
| SPEEK BNCM A-2 | 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
96.9 | 4.21 | 8.10 | 5.00 | 8.40 |
| SPEEK BNCM B-2 | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
97.8 | 3.81 | 6.32 | 4.10 | 9.27 |
| SPEEK BNCM C-2 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
98.3 | 2.71 | 4.96 | 2.85 | 9.47 |
| SPEEK BNCM D-2 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
99.1 | 1.31 | 2.14 | 1.35 | 9.63 |
| SPEEK BNCM E-2 | 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
99.3 | 0.08 | 1.09 | — | — |
| Nafion® 117 | — | 97.8 | 7.1 | 8.4 | 8.12 | 8.74 |
2.4. DMFC single cell performance test
All the membranes were pretreated with dilute sulfuric acid solution for 24 h. The MEA was prepared by hot pressing the anode and the cathode catalyst layers onto an optimized PEM. The active surface area of the MEA was 5 cm2 and this was composed of a Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ru alloy-supported carbon catalyst used for the anode and a Pt-black catalyst used for the cathode. The addition of Nafion® binder solution during the catalyst preparation bridged the active sites of the catalysts as well as bridging between catalysts and membranes in the interface. Then the slurry of catalyst was brushed directly (4 mg cm−2) onto the membranes, and these were hot pressed at 120 kg cm−2 pressure and 100 °C. A fuel cell test station, Won A Tech, Korea, was used for evaluating the performance by measuring the cell voltage versus current density at 70 °C. Air with a flow rate of 500 cc min−1 and methanol (2 M) with a flow rate of 10 cc min−1 were supplied to the cathode and anode respectively. Once the temperature reached 70 °C, the DMFC performance measurement was conducted three times, and the results are presented as the average of these.
Ru alloy-supported carbon catalyst used for the anode and a Pt-black catalyst used for the cathode. The addition of Nafion® binder solution during the catalyst preparation bridged the active sites of the catalysts as well as bridging between catalysts and membranes in the interface. Then the slurry of catalyst was brushed directly (4 mg cm−2) onto the membranes, and these were hot pressed at 120 kg cm−2 pressure and 100 °C. A fuel cell test station, Won A Tech, Korea, was used for evaluating the performance by measuring the cell voltage versus current density at 70 °C. Air with a flow rate of 500 cc min−1 and methanol (2 M) with a flow rate of 10 cc min−1 were supplied to the cathode and anode respectively. Once the temperature reached 70 °C, the DMFC performance measurement was conducted three times, and the results are presented as the average of these.
2.5. Membrane characterization
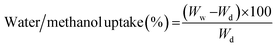 | (2) |
Similarly, the swelling ratios of the membranes were measured by immersing the various dried membranes in aqueous solutions at a predetermined temperature for 24 h and then at an elevated temperature, i.e. 70 °C, for 2 h. After a desired time, the length (Lw) and area (Aw) of the membranes were determined by wiping off the surface water with a piece of filter paper. The swelling ratio was calculated using eqn (3) and (4).
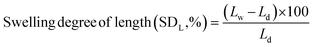 | (3) |
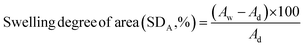 | (4) |
The phase morphologies of the membranes were studied using atomic force microscopy (M/s Park Autoprob, Korea) in non-contact mode.
| σ (S cm−1) = d/RS | (5) |
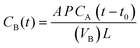 | (6) |
3. Results and discussion
3.1. Liquid uptakes and swelling ratios of membranes
Water content has a major effect on the proton conductivities of the PEMs. Generally proton transport through membranes requires a well-connected channel formed by ionic clusters of hydrophilic sulfonated functional groups. But too much water absorption by the membranes results in unstable membranes. Higher water uptake values of the membranes will enhance their swelling ratios and lead to a decrease in their mechanical stabilities.It was observed from Fig. 1 and Table 1 that the SPEEK blend nanocomposite membranes with 5 wt% of C 30B exhibit a decreasing tendency for water uptake, methanol uptake and swelling degree (both SDL and SDA) values with the incorporation of PVDF-HFP. At 25 °C, with the introduction of 25 wt% of PVDF-HFP, the water uptake values decreased from 25.10% to 11.36% and methanol uptake values decreased from 24.72% to 14.21%. Similarly, the blend nanocomposite PEM with 25 wt% of PVDF-HFP showed SDL and SDA values of about 12.32% and 8.31%, respectively, which is the most favourable swelling degree, with an excellent dimensional stability. Also, at higher temperature all the blend nanocomposite membranes showed comparatively low liquid uptake and swelling degree values with the addition of PVDF-HFP. It is expected that the lower swelling degree values of SPEEK blend nanocomposite membranes will improve their dimensional stabilities and maintain the contact between the PEMs and the catalysts, which can improve their DMFC performances.
The decrease in liquid uptake and swelling degree values can be explained as follows. The introduction of hydrophobic PVDF-HFP causes a larger average separation between the two neighbouring –SO3H groups, which causes a decrease in the formation of hydrophilic ionic clusters and therefore in water/methanol uptake values.19,20 Conversely, the addition of C 30B nanoclay facilitates the formation of intermolecular hydrogen bonds between the –SO3H group of SPEEK and the water and methanol molecules, and retains membrane hydrophilicity.11,12,26 Hence, the addition of C 30B nanoclay maintains the membrane liquid uptake characteristics and the hydrophobic PVDF-HFP polymer, controlling its dimensional stability. Also, it has been evident that the liquid uptake and swelling degree values of the SPEEK blend nanocomposite membranes were lower than those of the standard Nafion® 117 membrane.
3.2. Oxidative stability
The chemical/oxidative stability of a PEM under fuel cell operating conditions is a vital issue with a direct effect on the operation and durability of DMFCs. Peroxide radicals are responsible for chemical attack on the membrane material. In this work, the oxidative stabilities of the membranes were investigated in a 30% H2O2 aqueous solution at room temperature. From Table 1, it was noticed that the addition of 5 wt% C 30B and 25 wt% PVDF-HFP to the SPEEK system increases its weight residue from 92.10% to 99.30% after Fenton’s test. The weight loss of the pure SPEEK membrane after 5 h at room temperature was 7.9 wt%, while for Nafion® 117 it was found to be 2.2 wt%. As shown in Table 1, the blend nanocomposite membrane is considered to be more stable against peroxide radicals than the pure SPEEK membrane. The hydrophobicity and chemical stability of PVDF-HFP improves its resistance to attack towards free radical formation during the fuel cell operation reaction. This enhanced stability may be due to the restriction of movement of highly active free radicals within the blend nanocomposite membrane system for attack of the PEM.7,11,223.3. Proton conductivity
The proton conductivity values at 25 °C and 70 °C of all membranes are presented in Table 1. Prior to measurement, all membranes were fully hydrated in deionised water for 24 h at room temperature. Proton transport in membranes requires well-interconnected channels formed by clusters of hydrophilic sulfonated ionic functional groups. The diameter of the interconnected channels has a significant effect on the proton conduction rate in membranes. It can be observed from Table 1 that the proton conductivity values of Nafion® 117 and SPEEK at 25 °C are 7.10 and 3.18 × 10−2 S cm−1 respectively. Also it is evident that at 25 °C and 70 °C, with the addition of 20 wt% PVDF-HFP, percentage reductions of 72.87% and 74.88% were observed in the proton conductivity values, respectively. The introduction of hydrophobic PVDF-HFP reduces the density of the hydrophilic sulfonic acid groups on the SPEEK and the content of interconnected ionic channels perhaps decreases, thus reducing the proton conductivity values. At the same time, the addition of hydrophilic C 30B in the SPEEK/PVDF-HFP blend system forms a conducting ionic channel, which helps to retain water molecules within the membrane structure at higher temperature, maintaining the proton conductivity.27,28 The proton conductivity of Nafion® 117 was also measured under the same experimental conditions for comparison purposes. Further, the membranes with 25 wt% PVDF-HFP, i.e. SPEEK BNCM E-2 blend nanocomposite PEMs, showed lower proton conductivity values of the order of 10−3 S cm−1.Determination of the optimum loading of PVDF-HFP has been done by comparing the liquid uptake values, swelling degrees, and oxidative stabilities of all blend nanocomposite membranes. Also, apart from dimensional unsteadiness and the heterogeneous surface observed in the case of SPEEK BNCM E-2, beyond 20 wt% PVDF-HFP loading there was a considerable decrease in the values of proton conductivity. Since the primary objective of our study is to improve the dimensional and oxidative stability, the blend nanocomposite membrane with a composition with 20 wt% of PVDF-HFP, with a lower swelling degree and methanol permeability without much drop in the proton conductivity value, was optimized and used for further characterisation studies related to DMFC applications.
3.4. Wide angle X-ray diffraction (WXRD)
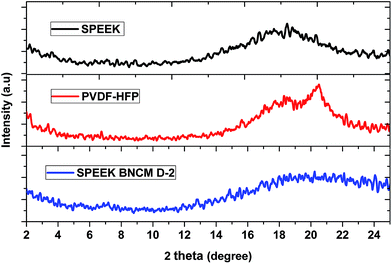
Generally the nanostructure of blend nanocomposites is typically characterized using XRD techniques. Fig. 2 illustrates the XRD patterns of the PVDF-HFP, SPEEK and SPEEK BNCM D-2 blend nanocomposite membranes. As seen in Fig. 2, two sharp diffraction peaks appear at 2 theta values of 18.4° and 20.18°, which correspond to the (1 0 0) and (0 2 0) reflections of the semicrystalline structure of PVDF-HFP.24,29 Due to the introduction of sulfonic acid groups in the structure, the SPEEK membrane shows amorphous characteristics by showing a wide diffraction peak. From the diffraction patterns, it was found that the sharp crystalline diffraction peaks of PVDF-HFP broadened in the presence of SPEEK and C 30B nanoclay. This may indicate that the addition of SPEEK induced significant disorder into the PVDF-HFP polymer. When C 30B was introduced to the SPEEK/PVDF-HFP blend system, there was no sharp deflection within the scattered angle limit, suggesting the possibility for extensive exfoliation of polymeric molecules within the C 30B layers.173.5. Thermal stability: TGA study
The thermal stabilities of the prepared SPEEK and SPEEK BNCM D-2 membranes were studied by TGA and the results are presented in Fig. 3. Both membranes showed multiple step degradation, in which the first degradation step below 200 °C is mainly attributed to the loss of moisture or residual solvent. In continuation, the major weight loss occurred in SPEEK and SPEEK BNCM D-2 between ∼200 °C and 400 °C, and is mainly associated with the desulfonation process of the sulfonic acid group in the SPEEK matrix, which involves the evolution of SO2 and SO gases.6,9,30 The transition observed in SPEEK membranes above 400 °C is mainly due to the degradation of the SPEEK polymeric main chain. The third weight loss beyond 400 °C is mainly attributed to the degradation of the polymeric chains of PVDF-HFP initially and the SPEEK chains secondly in the blend nanocomposite membrane system. Similar degradation characteristics have also been reported in the case of blend membranes of PVDF-HFP and SPEEK.19,22 Also, from the DTG thermogram, apart from the peak below 200 °C, which is associated with the evaporation of solvents and water molecules, a separate peak has been observed corresponding to a desulfonation process (200–400 °C) and degradation of the polymeric chains of PVDF-HFP and SPEEK (above 400 °C). Thus, from TGA analysis, it is evident that the SPEEK BNCM D-2 membrane shows sufficient thermal stability for DMFC applications.3.6. Morphological studies
SEM analysis of the membrane surface is an effective way to study the compatibility in polymer blend systems. The surface and cross-sectional morphology of SPEEK and its optimised blend nanocomposite membrane were studied by SEM. From Fig. 4(a) and (c), a clear dense morphology can be observed for SPEEK membranes.11 The cross-sectional morphology of the membranes seen in Fig. 4(d) revealed miscibility behaviour between SPEEK and PVDF-HFP. SPEEK blend nanocomposite membranes showed a comparatively good adhesion between SPEEK and the PVDF-HFP matrix. This might be due to the better interaction of C 30B nanoclay with the polymeric blend matrix due to polar–polar interactions. The improved homogeneous characteristics will help to retain water molecules within their ionic clusters, maintaining their hydrophilicity and thus proton conductivity values. Also, the most favourable proton conductivity values observed for the SPEEK blend nanocomposite membranes might be correlated with their better homogeneity observed from SEM images. | ||
| Fig. 4 SEM images of (a) SPEEK surface, (b) SPEEK BNCM D-2 surface, (c) SPEEK cross section, (d) SPEEK BNCM D-2 cross section. | ||
To further validate the result of the addition of hydrophobic PVDF-HFP on the microstructures of the membranes, non contact AFM images of SPEEK and SPEEK BNCM D-2 membranes were studied and their phase and three dimensional images are presented in Fig. 5(a–d). Generally in the AFM phase micrographs, dark regions are assigned to a hydrophilic region and light regions are assigned to a hydrophobic region.31,32 Hence, the distribution and connectivity of the hydrophilic conductive channels has a great influence on the proton conductivity values of the membranes. The three dimensional AFM images show a combination of peak- and valley-like structures. It is known that a relatively uniform nodular structure on the membrane surface improves the hydrophilicity and thus favors proton conduction. From Fig. 5, the SPEEK membrane showed a more continuous connectivity between hydrophilic channels, which creates a conductive pathway for the protons, i.e. the presence of more continuous dark regions in the AFM phase micrographs. Also, three dimensional images revealed the fact that SPEEK shows more continuous hydrophilic nodular channels as compared to SPEEK BNCM D-2. Thus the decrease in hydrophilic regions in the blend nanocomposite membranes, i.e. SPEEK BNCM D-2, may directly impact the reduction of liquid uptakes, swelling degrees, and proton conductivities of the membranes.
 | ||
| Fig. 5 AFM phase images of (a) SPEEK and (c) SPEEK BNCM D-2 membranes. Three dimensional images of (b) SPEEK and (d) SPEEK BNCM D-2 membranes. | ||
3.7. DMFC single cell performance test
Single cell DMFC performance curves for DMFCs, comprising Nafion® 117, SPEEK and SPEEK BNCM D-2 at an operating temperature of 70 °C, are shown in Fig. 6. Generally, the open circuit voltage (OCV) is directly interrelated to the fuel cross over, and generally, the higher the OCV, the lower the fuel crosses over.33,34 Moreover, a higher OCV of a membrane is due to better compatibility between the catalyst layer and the membrane.35 From our studied membranes, the OCV obtained for the SPEEK BNCM D-2 MEA is 0.79 V, which is a similar value to that of Nafion® 117 (0.78 V). Thus, the polarization curve clearly proves that the incorporation of PVDF-HFP and C 30B in the SPEEK matrix increases its DMFC performance, shown by higher OCV values. Also from Fig. 6, it has been observed that the current densities for SPEEK, SPEEK BNCM D-2 and Nafion® 117 were measured as 23, 39 and 36 mA cm−2 (at a potential of 0.4 V and 2 M methanol concentration), respectively. The maximum power density of the SPEEK BNCM D-2 membrane was measured as 55 mW cm−2 compared to 64 mW cm−2 for Nafion® 117 at 2 M methanol feed.Interestingly, the SPEEK BNCM D-2 blend nanocomposite PEM, even with a lower proton conductivity value than SPEEK, has provided a higher DMFC output in terms of power density and OCV value, which is a very clear indication for the leading role of reduced methanol permeability with the incorporation of PVDF-HFP and C 30B. On the other hand, the other possible reason for the better DMFC performance of SPEEK BNCM D-2 may be due to the stable MEA interface; it showed a lower activation resistance value, which improves the overall cell performance. Thus, lower methanol permeability and a stable MEA interface improves the performances of the SPEEK BNCM D-2 blend nanocomposite membrane. Also the improvement in the DMFC performance of SPEEK BNCM D-2 may be attributed to its lower swelling degree and better dimensional stability, as well as its oxidative stability.
3.8. Methanol permeability study
In addition to the proton conductivity and oxidative stability against peroxide radicals, the methanol permeability value has a significant effect on the performance of PEMs for DMFC applications. Methanol crossover through the PEM from anode to cathode is an undesirable situation because it leads to a voltage drop during the performance of the fuel cell. So methanol permeability should also be considered in the selection of a new membrane material for DMFCs. Membranes with lower methanol permeation are always preferable, as are those that give maximum power density values and maximum open circuit voltage (OCV) values. Fig. 7 shows the typical curve of methanol concentration vs. time for the investigated membranes, using a 2 M methanol solution. All values for methanol permeability tests for the PEMs were obtained from the slope of the straight line.Also, Fig. 8 and Table 1 describe the effect of addition of PVDF-HFP and C 30B particles, i.e. reduction in the methanol permeability values of SPEEK from 8.61 × 10−7 cm2 s−1 to 1.35 × 10−7 cm2 s−1 when the PVDF-HFP content is increased by approximately 5–20 wt%. The decrease in methanol permeability of the SPEEK BNCM D-2 blend nanocomposite membrane might be due the combined effect of the hydrophobic PVDF-HFP and C 30B clay layers, which creates a longer diffusive tortuous pathway for methanol, or the fact that some of the nanoscopic channels for passing methanol molecules are restricted.15,29 For ideal DMFC applications, the PEM should have the lowest methanol permeability and highest proton conductivity values with maximum power density. Hence the lower methanol permeability and average proton conductivity values of the SPEEK BNCM D-2 blend nanocomposite membrane is one of the significant advantages for its use as a PEM in DMFC systems.
 | ||
| Fig. 8 Methanol permeability and membrane selectivity values of fabricated blend nanocomposite membranes at various loadings of PVDF-HFP containing 5 wt% of C 30B. | ||
The term membrane selectivity, defined as the ratio of proton conductivity to methanol permeability, is commonly used to evaluate overall membrane performance. A membrane with a higher selectivity is desired for DMFCs. As shown in Fig. 8, the selectivities of the membranes increased with increasing PVDF-HFP content. The membrane SPEEK BNCM D-2 had a higher selectivity in comparison to the SPEEK membrane, primarily due to the enhanced methanol barrier property. Among all the membranes investigated in this study, the blend nanocomposite membrane SPEEK BNCM D-2 exhibited a higher selectivity value than Nafion® 117 membranes (only 8.74 × 104 S s cm−3), with a lower membrane thickness (100 μm). More importantly, SPEEK/PVDF-HFP/C 30B blend nanocomposite PEMs are cheap non-perfluorosulfonated polymer membranes, as compared to the standard Nafion® membranes.
4. Conclusions
SPEEK/PVDF-HFP/C 30B blend nanocomposite membranes with improved dimensional and oxidative stabilities and optimum proton conductivities were prepared by a solution casting method. Single cell DMFC performance reveals that the optimized SPEEK/PVDF-HFP/C 30B membrane showed an OCV of 0.79 V and a maximum power density value of 55 mW cm−2 at a 2 M methanol feed, which confirmed that the efficiency of the PEM was suitable for DMFC applications. In addition, the obtained methanol permeabilities of the PEMs were lower than those of Nafion® 117. The introduction of PVDF-HFP and C 30B collectively improved the oxidative and dimensional stabilities of PEMs. The liquid uptakes and the swelling ratios of the membranes decreased with the increase of PVDF-HFP content, which led to improved MEA interface stabilities. TGA studies revealed sufficient thermal stability for DMFC applications. Improvements in the OCV values, oxidative, thermal and liquid uptake stabilities and higher selectivity ratios of the membranes could be achieved without much loss in the proton conductivity values, making them suitable PEMs for DMFC applications.References
- J. Kerres, J. Membr. Sci., 2001, 185, 3–27 CrossRef CAS.
- L. Jorissen, V. Gogel, J. Kerres and J. Garche, J. Power Sources, 2002, 105, 267–273 CrossRef CAS.
- M. Amjadi, S. Rowshanzamir, S. J. Peighambardoust, M. G. Hosseini and M. H. Eikani, Int. J. Hydrogen Energy, 2010, 35, 9252–9260 CrossRef CAS PubMed.
- M. G. Casciola, M. Alberti, M. Sganappa and R. Narducci, J. Power Sources, 2006, 162, 141–145 CrossRef CAS PubMed.
- W. C. Choi, J. D. Kim and S. I. Woo, J. Power Sources, 2001, 96, 411–414 CrossRef CAS.
- S. M. J. Zaidi, S. D. Mikhailenko, G. P. Robertson, M. D. Guiver and S. J. Kaliaguine, J. Membr. Sci., 2000, 173, 17–34 CrossRef CAS.
- H. Y. Jung and J. K. Park, Int. J. Hydrogen Energy, 2009, 34, 3915–3921 CrossRef CAS PubMed.
- H. Heyi, Y. Wen, Z. Hui and L. Gregory, J. Polym. Sci., Part A: Polym. Chem., 2014, 52, 121–127 CrossRef.
- D. Gupta and V. Choudhary, React. Funct. Polym., 2013, 73, 1268–1280 CrossRef CAS PubMed.
- L. M. Carvalho, A. R. Tan and G. A. Souza, J. Appl. Polym. Sci., 2008, 110, 1690–1698 CrossRef.
- R. H. Varsha, K. R. Sangram, R. Swati and M. Patri, J. Membr. Sci., 2011, 372, 40–48 CrossRef PubMed.
- M. H. S. Mohammad, D. Erfan, S. Kaveh, S. M. Fatemeh and K. Ghader, J. Power Sources, 2010, 195, 2450–2456 CrossRef PubMed.
- M. M. Hasani, E. Dashtimoghadam, F. S. Majedi and K. K. Kabiri, J. Power Sources, 2009, 190, 318–321 CrossRef PubMed.
- M. L. Ponce, L. Prado, B. Ruffmann, K. Richau, R. Mohr and S. P. Nunes, J. Membr. Sci., 2003, 217, 5–15 CrossRef CAS.
- G. Rapee, C. Suwabun, P. N. Suzana and S. Sergey, J. Membr. Sci., 2008, 323, 337–346 CrossRef PubMed.
- M. Mohammad, S. Hasani, H. E. Sharriar, G. Reza and M. Homayoun, Energy Fuels, 2008, 22, 2539–2592 CrossRef.
- X. Song, Y. Geping, C. Kedi and S. Yuyan, J. Membr. Sci., 2007, 289, 51–57 CrossRef PubMed.
- B. Jung, B. Kim and J. M. Yang, J. Membr. Sci., 2004, 245, 61–69 CrossRef CAS PubMed.
- J. Wootthikanokkhan and N. Seeponkai, J. Appl. Polym. Sci., 2006, 102, 5941–5948 CrossRef CAS.
- H. Y. Jung and J. K. Park, Int. J. Hydrogen Energy, 2009, 34, 3915–3921 CrossRef CAS PubMed.
- S. Ren, G. Sun, C. Li, Z. Wu, W. Jin, W. Chen, Q. Xin and X. Yang, Mater. Lett., 2006, 60, 44–47 CrossRef CAS PubMed.
- T. Y. Inan, H. Dogan, E. E. Unveren and E. Eker, Int. J. Hydrogen Energy, 2010, 35, 12038–12053 CrossRef CAS PubMed.
- H. Y. Jung and J. K. Park, Electrochim. Acta, 2007, 52, 7464–7468 CrossRef CAS PubMed.
- S. Xue and G. Yin, Polymer, 2006, 47, 5044–5049 CrossRef CAS PubMed.
- L. Unnikrishnan, S. Mohanty and S. K. Nayak, High Perform. Polym., 2013, 25, 854–867 CrossRef PubMed.
- L. Unnikrishnan, M. Prasad, S. Mohanty and S. K. Nayak, Polym.-Plast. Technol. Eng., 2012, 51, 568–577 CrossRef CAS.
- B. Smitha, S. Sridhar and A. A. Khan, Macromolecules, 2004, 37, 2233–2239 CrossRef CAS.
- Y. F. Lin, C. Y. Yen, C. C. M. Ma, S. H. Liao, C. H. Hung and Y. H. Hsiao, J. Power Sources, 2007, 165, 692–700 CrossRef CAS PubMed.
- K. G. Gnana, K. Pillai, S. N. Kee and E. R. Nimma, J. Membr. Sci., 2007, 303, 126–131 CrossRef PubMed.
- Y. Luo, R. Huo, X. Jin and F. E. Karasz, J. Anal. Appl. Pyrolysis, 1995, 34, 229–242 CrossRef CAS.
- X. Li, C. Zhao, H. Lu, Z. Wang and H. Na, Polymer, 2005, 46, 5820–5827 CrossRef CAS PubMed.
- Y. S. Kim, M. A. Hickenr, L. M. Dong, B. S. Pivovar and J. E. McGrath, J. Membr. Sci., 2004, 243, 317–326 CrossRef CAS PubMed.
- H. L. Wu, C. C. M. Ma, H. C. Kuan, C. H. Wang, C. Y. Chen and C. L. Chiang, J. Polym. Sci., Part B: Polym. Phys., 2006, 44, 565–572 CrossRef CAS.
- J. Juhana, A. F. Ismail, T. Matsuura and K. Nagai, J. Membr. Sci., 2000, 173, 17–34 CrossRef.
- M. H. S. Mohammad, R. G. Seyed, M. D. Nassir, D. Erfan and S. M. Fatemeh, Solid State Ionics, 2009, 180, 1497–1504 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2014 |